UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
|
| |
ý | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2017
OR
|
| |
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the transition period from to
Commission file number: 001-37471
PIERIS PHARMACEUTICALS, INC.
(Exact name of registrant as specified in its charter)
|
| | |
Nevada | | EIN 30-0784346 |
(State or other jurisdiction of incorporation or organization) | | (I.R.S. Employer Identification No.) |
| | |
255 State Street, 9th Floor Boston, MA United States | | 02109 |
(Address of principal executive offices) | | (Zip Code) |
Registrant’s telephone number, including area code
857-246-8998
Securities registered pursuant to Section 12(b) of the Exchange Act:
|
| | |
Title of each class | | Name of each exchange on which registered |
Common Stock, par value $0.001 per share | | The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Exchange Act:
None
(Title of class)
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ý
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Exchange Act. Yes ☐ No ý
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ý No ☐
Indicate by check mark whether the registrant has submitted electronically and posted on its corporate Web site, if any, every Interactive Data File required to be submitted and posted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes ý No ☐
Indicate by check mark if disclosure of delinquent filers pursuant to Item 405 of Regulation S-K is not contained herein, and will not be contained, to the best of registrant’s knowledge, in definitive proxy or information statements incorporated by reference in Part III of this Form 10-K or any amendment to this Form 10-K. ý
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company. See the definitions of “large accelerated filer,” “accelerated filer” and “smaller reporting company” in Rule 12b-2 of the Exchange Act. (Check one):
|
| | | | | | |
Large accelerated filer | | ☐ | | Accelerated filer | | ☐ |
| | | |
Non-accelerated filer | | ☐ [Do not check if a smaller reporting company] | | Smaller reporting company | | ý
|
| | | | | | |
Emerging growth company ý | | | | |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ý
The aggregate market value of Common Stock held by non-affiliates of the registrant on June 30, 2017, the last business day of the registrant’s most recently completed second fiscal quarter, based on the closing price on that date of $5.06, was $184,955,062.
As of March 12, 2018, the registrant had 53,855,371 shares of common stock outstanding.
DOCUMENTS INCORPORATED BY REFERENCE
None.
TABLE OF CONTENTS
Forward Looking Statements
This Annual Report on Form 10-K contains forward looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), that involve risks and uncertainties, principally in the sections entitled “Business,” “Risk Factors,” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations.” All statements other than statements of historical fact contained in this Annual Report on Form 10-K, including statements regarding future events, our future financial performance, business strategy and plans and objectives of management for future operations, are forward-looking statements. We have attempted to identify forward-looking statements by terminology including “anticipates,” “believes,” “can,” “continue,” “ongoing,” “could,” “estimates,” “expects,” “intends,” “may,” “appears,” “suggests,” “future,” “likely,” “goal,” “plans,” “potential,” “projects,” “predicts,” “should,” “would,” or “will” or the negative of these terms or other comparable terminology. Although we do not make forward-looking statements unless we believe we have a reasonable basis for doing so, we caution you that these statements are based on our projections of the future that are subject to known and unknown risks and uncertainties and other factors that may cause our actual results, level of activity, performance or achievements expressed or implied by these forward-looking statements, to differ. The description of our Business set forth in Item 1, the Risk Factors set forth in Item 1A and our Management’s Discussion and Analysis of Financial Condition and Results of Operations set forth in Item 7 as well as other sections in this report, discuss some of the factors that could contribute to these differences. These forward-looking statements include, among other things, statements about:
| |
• | the accuracy of our estimates regarding expenses, future revenues, uses of cash, capital requirements and the need for additional financing; |
| |
• | the initiation, cost, timing, progress and results of our development activities, preclinical studies and clinical trials; |
| |
• | the timing of and our ability to obtain and maintain regulatory approval of our existing product candidates, any product candidates that we may develop, and any related restrictions, limitations, and/or warnings in the label of any approved product candidates; |
| |
• | our plans to research, develop and commercialize our current and future product candidates; |
| |
• | our collaborators’ election to pursue research, development and commercialization activities; |
| |
• | our ability to obtain future reimbursement and/or milestone payments from our collaborators; |
| |
• | our ability to attract collaborators with development, regulatory and commercialization expertise; |
| |
• | our ability to obtain and maintain intellectual property protection for our product candidates; |
| |
• | our ability to successfully commercialize our product candidates; |
| |
• | the size and growth of the markets for our product candidates and our ability to serve those markets; |
| |
• | the rate and degree of market acceptance of any future products; |
| |
• | the success of competing drugs that are or become available; |
| |
• | regulatory developments in the United States and other countries; |
| |
• | the performance of our third-party suppliers and manufacturers and our ability to obtain alternative sources of raw materials; |
| |
• | our ability to obtain additional financing; |
| |
• | our use of the proceeds from our securities offerings; |
| |
• | any restrictions on our ability to use our net operating loss carryforwards; and |
| |
• | our ability to attract and retain key personnel. |
Moreover, we operate in a very competitive and rapidly changing environment. New risks emerge from time to time and it is not possible for us to predict all risk factors, nor can we address the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause our actual results to differ materially from those contained in any forward-looking statements.
You should not place undue reliance on any forward-looking statement, each of which applies only as of the date of this Annual Report on Form 10-K. Before you invest in our securities, you should be aware that the occurrence of the events described in the section entitled “Risk Factors” and elsewhere in this Annual Report on Form 10-K could negatively affect our business, operating results, financial condition and stock price. All forward-looking statements included in this document are based on information available to us on the date hereof, and except as required by law, we undertake no obligation to update or revise publicly any of the forward-looking statements after the date of this Annual Report on Form 10-K to conform our statements to actual results or changed expectations.
We have registered trademarks for Pieris® and Anticalin®. All other trademarks, trade names and service marks included in this Annual Report on Form 10-K are the property of their respective owners. Use or display by us of other parties’ trademarks, trade dress or products is not intended to and does not imply a relationship with, or endorsements or sponsorship of, us by the trademark or trade dress owner.
As used in this Annual Report on Form 10-K, unless the context indicates or otherwise requires, “our Company”, “the Company”, “Pieris”, “we”, “us”, and “our” refer to Pieris Pharmaceuticals, Inc., a Nevada corporation, and its consolidated subsidiary, Pieris Pharmaceuticals GmbH (formerly known as Pieris AG), a company organized under the laws of Germany, Pieris Australia Pty Ltd., a company organized under the laws of Australia that is a consolidated subsidiary of Pieris Pharmaceuticals GmbH, and Pieris Securities Corporation, a Massachusetts securities corporation, a consolidated subsidiary of Pieris Pharmaceuticals, Inc. Effective as of August 26, 2015 and with notification from the Amtsgericht München as of September 29, 2015, Pieris AG was transformed to Pieris Pharmaceuticals GmbH as a result of a change in the legal entity.
Currency Presentation and Currency Translation
Unless otherwise indicated, all references to “dollars,” “$,” “U.S. $,” or “U.S. dollars” are to the lawful currency of the United States. All references in this Report to “euro” or “€” are to the currency introduced at the start of the third stage of the European Economic and Monetary Union pursuant to the Treaty establishing the European Community, as amended. We prepare our financial statements in U.S. dollars.
The functional currency for our operations is primarily the euro. With respect to our financial statements, the translation from the euro to U.S. dollars is performed for balance sheet accounts using exchange rates in effect at the balance sheet date and for revenue and expense accounts using a weighted average exchange rate during the period. The resulting translation adjustments are recorded as a component of accumulated other comprehensive loss.
Where in this Report we refer to amounts in euros, we have for your convenience also, in certain cases, provided a conversion of those amounts to U.S. dollars in parentheses. Where the numbers refer to a specific balance sheet account date or financial statement account period, we have used the exchange rate that was used to perform the conversions in connection with the applicable financial statement. In all other instances, unless otherwise indicated, the conversions have been made using the noon buying rate of €1.00 to U.S. $1.1996 based on Thomson Reuters as of December 31, 2017.
PART I
Corporate History
General
Pieris Pharmaceuticals, Inc. was incorporated in the State of Nevada in May 2013 under the name “Marika Inc.”. Pieris Pharmaceuticals, Inc. began operating the business of Pieris Pharmaceuticals GmbH, or Pieris GmbH, through a reverse acquisition on December 17, 2014. Pieris GmbH (formerly Pieris AG, a German company which was founded in 2001) continues as an operating subsidiary of the Company.
Pieris Pharmaceuticals, Inc. is the sole stockholder of Pieris GmbH with its corporate headquarters located at 255 State Street, 9th Floor, Boston, Massachusetts 02109. The research facilities of Pieris GmbH are located in Freising, Germany. Pieris Australia Pty Ltd., a wholly owned subsidiary of Pieris GmbH, was formed on February 14, 2014 to conduct research and development in Australia. Pieris Pharmaceuticals Securities Corporation, a wholly owned subsidiary of Pieris Pharmaceuticals, Inc. was formed on December 14, 2016 to buy, sell, deal in, or hold securities on its own behalf and not as a broker, and will engage in its activities exclusively for investment purposes.
Business Overview
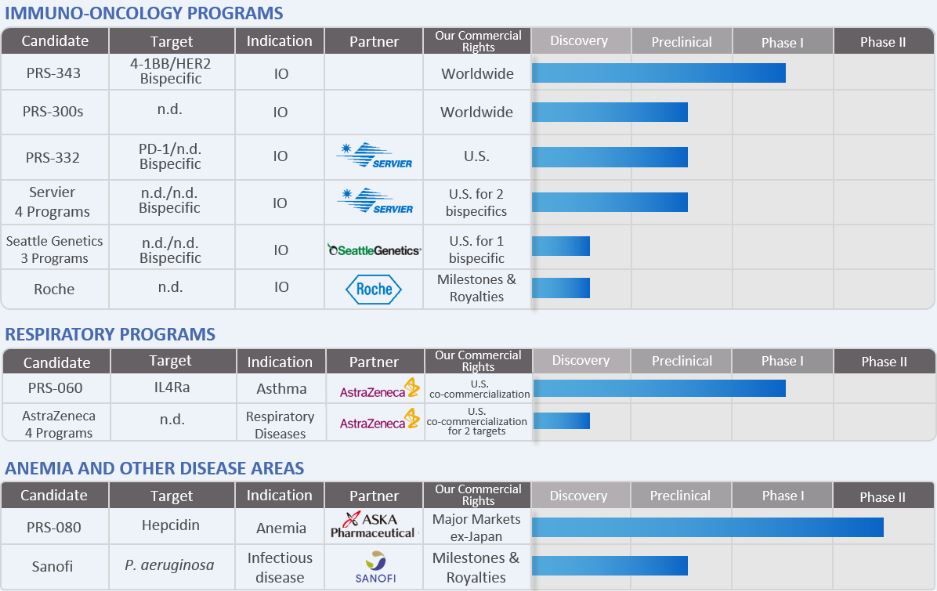
We are a clinical stage biotechnology company that discovers and develops Anticalin-based drugs to target validated disease pathways in a unique and transformative way. Our clinical pipeline includes an immuno-oncology (IO) bispecific targeting HER2 and 4-1BB, an inhaled IL-4 receptor alpha antagonist Anticalin protein to treat uncontrolled asthma and a half-life-optimized, hepcidin antagonizing Anticalin protein to treat anemia. Our proprietary Anticalin proteins are a novel class of therapeutics validated in the clinic and by partnerships with leading pharmaceutical companies.
Anticalin proteins are a class of low molecular-weight therapeutic proteins derived from lipocalins, which are naturally occurring proteins typically found in human blood plasma and other bodily fluids. Anticalin-branded proteins function similarly to monoclonal antibodies, or mAbs, by binding tightly and specifically to a diverse range of targets. An antibody is a large protein used by the immune system that recognizes a unique part of a foreign target molecule, called an antigen. We believe Anticalin proteins possess numerous advantages over antibodies in certain applications. For example, Anticalin proteins are small in size and are monomeric, meaning they are comprised of a single protein units rather than a multi-protein complex. Therefore, we believe Anticalin proteins are generally more stable biophysically than tetrameric monoclonal antibodies, composed of four protein subunits, potentially enabling unique routes of drug administration such as pulmonary delivery. Higher-molecular-weight entities, such as antibodies, are often too large to be delivered effectively through these methods. Our Anticalin technology is modular, which allows us to design Anticalin proteins to bind with specificity to one or multiple targets at the same time. This multispecificity offers advantages in biological settings where binding to multiple targets can enhance the ability of a drug to achieve its desired effects, such as killing cancer cells. Moreover, unlike antibodies, the pharmacokinetic, or PK, profile of Anticalin proteins can be adjusted to potentially enable program-specific optimal drug exposure. Such differentiating characteristics suggest that Anticalin proteins have the potential, in certain cases, to become first-in-class drugs.
We have access to intellectual property rights directed to various aspects of our Anticalin technology platform, allowing for development and advancement of our platform and drug candidates. We believe our ownership and/or license of our Anticalin platform provides us with a strong intellectual property position, particularly where we are seeking to address targets and diseases in a novel way and for which there is existing monoclonal antibody intellectual property.
Our core Anticalin technology and platform were developed in Germany, and we have collaboration arrangements with major multi-national pharmaceutical companies headquartered in the U.S., Europe, and Japan. We also established a collaboration with Les Laboratoires Servier and Institut de Recherches Internationales Servier, or Servier, in January 2017 and entered into an exclusive license agreement with Aska Pharmaceutical Co., Ltd., or Aska, in February 2017. In May 2017, we entered into a License and Collaboration Agreement, and a Non-Exclusive Anticalin Platform Technology License Agreement with AstraZeneca AB, or AstraZeneca. In February 2018 we entered into a collaboration and license agreement with Seattle Genetics Inc. ("Seattle Genetics"). We have also in-licensed different antibodies to drive the advancement of our multispecific immuno-oncology pipeline. These include existing agreements with Sanofi Group, or Sanofi, and F.Hoffmann—La Roche Ltd. and Hoffmann—La Roche Inc., or Roche.
In connection with our efforts to develop multispecific Anticalin-based proteins designed to engage immunomodulatory targets, during the second quarter of 2017, the Company entered into a License and Transfer Agreement with Sichuan Kelun-Biotech Biopharmaceutical Co. Ltd. (“Kelun”). Under that Agreement, Kelun has granted to us a non-exclusive worldwide license (with the right to sublicense) under certain intellectual property owned or controlled by Kelun to research, develop, manufacture, and commercialize bi- and multi- specific fusion proteins that include a monoclonal antibody developed by Kelun specific for an undisclosed target and one or more Anticalin proteins. We believe that the drug-like properties of the Anticalin drug class were demonstrated in various clinical trials with different Anticalin-based drug candidates, including PRS-080, PRS-060 and others.
Our current development plans focus on two core pillars, immuno-oncology, or IO, and respiratory diseases. In addition, we are developing PRS-080, a non-core asset, for the treatment of functional iron deficiency anemia.
The first bispecific Anticalin-based drug candidate in our IO pipeline, PRS-343, was designed to target the immune receptor 4-1BB and the tumor target HER2. PRS-343 is the result of a genetic fusion of a variant of the HER2-targeting antibody trastuzumab with an Anticalin protein specific for 4-1BB. The mode of action of this 4-1BB/ HER2 bispecific is to promote 4-1BB clustering by bridging 4-1BB-positive T cells with HER2-positive tumor cells, and to thereby provide a potent costimulatory signal to tumor antigen-specific T cells. PRS-343 is intended to localize 4-1BB activation in the tumor, and to thereby both increase efficacy and reduce systemic toxicity compared to 4-1BB-targeting antibodies being developed by third parties in clinical trials. Patient dosing in a multicenter, open-label, Phase I dose escalation study that will include expansion cohorts, commenced in September 2017. The study is designed to determine the safety, tolerability, and potential anti-cancer activity of PRS-343 in patients with advanced or metastatic HER2-positive solid tumors for which standard treatment options are not available, are no longer effective, are not tolerated, or the patient has refused standard therapy. Elevated HER2 expression is associated with multiple cancers, including gastroesophageal, bladder, breast, and a range of other tumor types.
In January 2017 Pieris initiated a strategic collaboration with Servier to discover and develop five Anticalin-based bispecific therapeutics in immuno-oncology. Three programs are currently undergoing preclinical evaluation together with Servier.
Pieris continues to explore opportunities to develop additional differentiated Anticalin-based multispecific therapeutics in IO. To this end, discovery campaigns are ongoing to generate functionally active Anticalin proteins against immunomodulatory targets. We are carrying out proof of concept and proof of mechanism studies will be carried out on several additional fully proprietary programs to support drug candidate nomination.
The second Anticalin drug candidate, PRS-060, binds to the IL-4 receptor alpha-chain (IL-4Ra), thereby inhibiting the actions of IL-4 and IL-13, two cytokines (small proteins mediating signaling between cells within the human body) known to be key mediators in the inflammatory cascade that causes asthma and other inflammatory diseases. The small size and biophysical stability of PRS-060 could enable direct delivery to the lungs, through the use of an inhaler, which we believe may enable high pulmonary concentrations of the drug candidate to be achieved at lower doses than would be reached with antibodies that are systemically delivered. Further, an inhaled drug may be expected to be substantially better tolerated than systemically administered antibodies. We initiated a single ascending dose study of PRS-060 in December 2017.
PRS-060 is being developed in partnership with AstraZeneca under our strategic alliance initiated in May 2017. Pieris is sponsoring the Phase I clinical study for PRS-060, after which AstraZeneca will be responsible for further clinical development. Pieris has the right to opt-into further co-development and co-commercialization of PRS-060 with AstraZeneca after completion of the Phase IIa clinical study.
The third Anticalin drug candidate, PRS-080, is a polyethylene glycol (PEG)-conjugated Anticalin protein that binds to hepcidin, a natural regulator of iron levels in the blood. An excess amount of hepcidin can cause functional iron deficiency, or FID, which often cannot be treated adequately with iron supplements and can lead to anemia. PRS-080 has been designed to target hepcidin for the treatment of FID in anemic patients with chronic kidney disease, or CKD, particularly in end-stage renal disease patients requiring dialysis. We believe that by blocking the actions of hepcidin, PRS-080 may serve to address anemia by mobilizing iron from the endogenous iron stores in the body for incorporation into red blood cells. With a serum half-life of several days, PRS-080 was designed to inhibit hepcidin sufficiently to mobilize functional serum iron for erythropoiesis, followed by recovery of blood hepcidin levels to prevent iron overload.
PRS-080 has been investigated in two single-ascending dose Phase I/Ib trials, first in healthy subjects, then in stage 5 CKD patients requiring hemodialysis, both under governance by the German Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, or BfArM). Single intravenous PRS-080 administrations were safe and well tolerated up to the tested dose of 16 mg/kg in healthy volunteers and up to the tested dose of 8 mg/kg in end-stage CKD patients. The studies were completed in 2015 and 2017, respectively. Both trials demonstrated dose proportional elevations in both serum iron and transferrin saturation. Based on the Phase I results, a multicenter, randomized, double-blind, placebo-
controlled, multiple ascending dose (two cohorts of 4mg/kg and 8 mg/kg, respectively) pilot Phase IIa study in anemic hemodialysis dependent CKD patients commenced in the third quarter of 2017. This study is intended primarily to obtain initial results on the safety, tolerability, and pharmacological activity of 5 once weekly doses of PRS-080, and secondarily to evaluate the effect of repeated PRS-080 administration on hemoglobin levels in this patient population. Completion of dosing of all patients is expected in mid-2018.
Strategy
Our goal is to become a fully integrated biotechnology company by discovering and developing Anticalin-based therapeutics to target validated disease pathways in a unique and transformative way, and later developing and commercializing our products. We intend to engage with partners for many of our programs in a combination of geographic and indication-based arrangements to maximize our business opportunities. We also intend to retain certain development and commercial rights on selected products as our experience in drug development grows. Key elements of our strategy include:
| |
• | Advancing PRS-343 through Phase I dose escalation first-in-patient trial followed by expansion studies and combination regimens in selected HER2 positive tumor patient populations with major unmet needs. |
A multicenter Phase I study with PRS-343 commenced in September 2017. The study will aim to assess safety and tolerability of PRS-343.
| |
• | Advancing PRS-060 through first-in-human trial. Our first-in-human clinical trial for PRS-060 commenced in December 2017 and we intend to start a follow up study that demonstrates Proof of Mechanism for PRS-060 in the second half of 2018. |
| |
• | Advancing PRS-332 to development candidate nomination and initiate IND-enabling activities. PRS-332 is currently undergoing preclinical evaluation together with Servier. |
| |
• | Advancing PRS-080 through a multi-ascending dose study |
In the fourth quarter of 2017 we initiated a Phase IIa study of PRS-080 in anemic hemodialysis dependent CKD patients. This study is intended primarily to obtain initial results on the safety, tolerability, and pharmacological activity of 5 once weekly doses of PRS-080, and secondarily to evaluate the effect of repeated PRS-080 administration on hemoglobin levels in this patient population. Completion of dosing of all patients is expected in the second half of 2018.
| |
• | Continuing to build our platform by entering into new partnerships and license and collaborative arrangements and advancing our currently partnered programs. We have entered into partnership and collaborative arrangements with pharmaceutical companies in a diverse range of therapeutic areas and geographies. We have active partnerships with global pharmaceutical companies, such as Servier, AstraZeneca, Seattle Genetics, Sanofi, Roche, and Aska. Together with our partners, we intend to advance multiple drug candidates through preclinical studies and to select further drug candidates for clinical development in the future. We will also continue to seek to engage with new pharmaceutical partners that can contribute funding, experience and marketing ability for the successful development and commercialization of our current and future drug candidates. |
| |
• | Pursuing additional opportunities for our Anticalin technology. We intend to continue to identify, vet and pursue opportunities to develop novel Anticalin therapeutics for oncology, respiratory diseases, and additional diseases, as we continue to improve on the Anticalin platform technology. |
Anticalin platform technology
Our platform technology focuses on low molecular-weight Anticalin proteins that bind tightly and specifically to a diverse range of targets. Anticalin proteins are derived from human proteins called lipocalins, which are naturally occurring low-molecular weight human proteins of approximately 18 to 20kDA molecular mass typically found in blood plasma and other bodily fluids. The lipocalin class of proteins defines a group of extracellular specific-binding proteins that, collectively, exhibit extremely high structural homology, yet have an uncharacteristically low amino acid sequence identity (less than 20%), making them attractive “templates” for amino acid diversification. Lipocalins naturally bind to, store and transport a wide spectrum of molecules. The defining attributes of the 12-member human lipocalin class and, by extension, Anticalin proteins, engineered from the lipocalin class of proteins, are a four-loop variable region and a rigidly conserved beta-barrel backbone, which, together, form a cup-like binding pocket. The graphic below shows both tear (left) and NGAL (right) lipocalins.
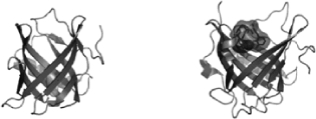
We currently develop our Anticalin proteins from either tear lipocalin, found primarily in human tear fluid as well as the lung epithelium, or neutrophil gelatinase-associated lipocalin (NGAL), a protein involved in the innate immune system, by making discreet mutations in the genetic code for the binding regions. These mutations have the potential to lead to highly specific, high-affinity binding for both small and large molecular targets. Random mutations are introduced at pre-defined positions involved in endogenous ligand engagement, creating exponentially diverse pools of Anticalin proteins, the most potent and well behaved of which are selected and optimized in a customized manner through in vitro selection. Using techniques such as phage display, a successful technique in antibody-based drug discovery, to build and refine antibody libraries, the ability to introduce diversity and then select for the best binders among a large pool of Anticalin proteins gives us the opportunity to select Anticalin proteins for a wide variety of targets. The flexibility inherent in the Anticalin proteins’ cup-like structure allows us to choose both small-molecule targets that fit inside the ‘cup’ as well as larger protein targets that can be bound by the Anticalin proteins’ outward-facing arms. Our Phase Ia trial for PRS-080, our prior Phase I study of PRS-050 as well as Daiichi's Phase I study of a PCSK9-specific Anticalin protein indicates that these proteins may be non-immunogenic and thereby have the potential to exhibit a favorable safety profile.
The below graphic demonstrates Anticalin drug candidates binding to a small molecule (left), a small protein target (hepcidin, center) and a large protein target (CTLA4, right):
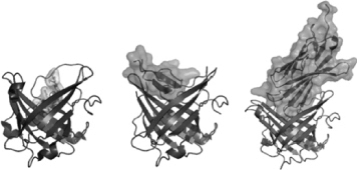
To obtain a specific Anticalin protein, we take advantage of the breadth of our proprietary Anticalin libraries, generated through our protein engineering expertise. We have created, and will continue to create, proprietary Anticalin libraries by rationally diversifying the lipocalin regions that are responsible for ligand binding, applying different libraries to different types of targets. By utilizing bacterial production from the earliest stages of drug discovery through Current Good Manufacturing Practice, or cGMP, manufacturing, we have created a seamless platform that improves the quality, yield and cost-effectiveness of our drug candidates. However, Anticalin protein manufacturing is not limited to bacterial systems, with the underlying expression system being driven on a program-by-program basis. See “—Manufacturing” below.
As targeted, protein-based molecules, Anticalin proteins also function similarly to monoclonal antibodies, thereby offering many of the same favorable qualities, including:
| |
• | High specificity to their targets. Like monoclonal antibodies, Anticalin proteins can bind their targets without binding other molecules, even molecules with very similar chemical structures or amino acid sequences, allowing for more effective treatments through, for example, minimizing off-target effects. |
| |
• | Tight binding and effective biological activity at their targets. Like monoclonal antibodies, Anticalin proteins are able to bind their targets at subnanomolar affinities. Anticalin proteins can potentially achieve desirable biological effects by inhibiting an undesired or inducing a desired cell activity by binding to cell-surface receptors or their ligands. |
| |
• | Human origin. Like many monoclonal antibodies in development and marketed today, Anticalin proteins are derived from a natural class of circulating human proteins. Their human origin increases the likelihood that Anticalin proteins will not be recognized as foreign by the immune system and subsequently rejected. |
| |
• | Scalability for large-scale production. Like monoclonal antibodies, Anticalin proteins lend themselves to large-scale production, yet can also be produced in a range of expression systems ranging from prokaryotic (bacterial) to eukaryotic (animal, plant, fungal) cells. Anticalin proteins can take advantage of several well-understood and widely |
practiced methods of protein production both in small amounts for preclinical testing and at larger scale for clinical trials and commercial production.
While often compared to monoclonal antibodies, Anticalin proteins, we believe, offer several advantages over antibodies, including:
| |
• | Small size and biophysical stability. Anticalin proteins are small in size and are monomeric. Therefore, we believe Anticalin proteins are generally more stable biophysically than tetrameric monoclonal antibodies, which will potentially enable unique routes of administration to target diseases, such as pulmonary delivery. Higher-molecular-weight entities such as antibodies are often too large to be delivered effectively through these methods. We believe Anticalin proteins will also be less expensive to manufacture than antibodies due to their lower molecular weight and less bulky structure as well as the ability to use the prokaryotic-based manufacturing systems, a less costly manufacturing system than mammalian cell-based manufacturing systems, to create them. |
| |
• | Optimization of half-life. Anticalin proteins can be engineered to have a half-life that is optimal for the indication area and a desired dosing schedule. Antibodies typically have half-lives of two weeks or longer, whereas Anticalin proteins can be engineered to have half-lives from hours to weeks, depending on the half-life extension technology employed, if any. This optionality allows us to exert greater control over the amount of circulating Anticalin protein in the blood and the amount of time such Anticalin proteins circulate in the blood, depending on the underlying biology we are trying to address. |
| |
• | Modular platform for higher-order multispecificity and avoidance of cross-linking. Our Anticalin technology is modular, allowing for monovalent or multivalent target engagement, including multispecificity within a single protein. We believe that a monovalent “backbone” is an advantage in situations where pure antagonism of certain cellular receptors is desired. The dual-binding nature of monoclonal antibodies, which have two “arms,” can be a disadvantage in cases when the antibodies bind to and cross-link cell-surface receptors. Such cross-linking often leads to undesirable activation of the cells bearing those receptors. Single-action (monovalent) Anticalin proteins have only a single binding site and are thus not subject to cross-linking. Further, when it is called for by the biology we are addressing, we can create multispecific Anticalin proteins that can simultaneously bind (i) two or more different targets or (ii) different epitopes, the specific piece of an antigen to which an antibody binds, on the same target by genetically linking Anticalin proteins with distinct specificities on a common cDNA strand. We believe this multispecificity offers advantages in biological settings where binding to multiple targets can enhance the ability of a drug to achieve its desired effects, such as killing cancer cells. Unique Anticalin proteins can be pieced together and undergo simultaneous target engagement as a single fusion protein, without generally compromising on manufacturability. |
| |
• | Flexible formatting facilitates selection of potent T cell engagers. The molecular architecture of Anticalin proteins as a single polypeptide chain that folds into a stable eight-stranded β-barrel with exposed N- and C-termini, both not part of the binding site, makes them ideal building blocks to generate bispecific and even multispecific fusion proteins offering novel therapeutic modalities. Multispecific Anticalin-based fusion proteins can be used to pursue innovative therapeutic strategies in IO, particularly by addressing the “immunological synapse” that forms at the interface upon contact between an immune cell and a cancer cell. This can drive an efficient activation of tumor-specific T cells in the vicinity of the tumor, thereby avoiding some of the toxicities observed with peripheral T cell activation in healthy tissues. Generally, the formatting flexibility of Anticalin-based biologics offers the ability of modulating valency and geometry of the multispecific compound according to biological needs. For example, Anticalin proteins can be genetically fused to either the N- or C-terminus of the antibody heavy or light chain, thereby resulting in different geometries of the fusion protein with the antibody as well as Anticalin binding sites covering a range of distances with regard to the T cell target on the one hand and the tumor antigen on the other. |
Implementation of our Anticalin Platform Technology: Our Drug Candidates Pipeline
Each of our drug candidates is in the early stage of development, and we anticipate that it will likely be several years before any of our drug candidates could be commercialized. The following table summarizes the status of our current drug candidates and programs:
PRS-343 targeting 4-1BB (CD-137) in oncology
PRS-343 is a bispecific protein targeting the immune receptor 4-1BB and the tumor target HER2. It is generated by genetic fusion of an Anticalin protein specific for 4-1BB to each heavy chain of a variant of the HER2-targeting antibody trastuzumab. The mode of action of this 4-1BB/HER2 bispecific is to promote 4-1BB clustering by bridging 4-1BB-positive T cells with HER2-positive tumor cells, and to thereby provide a potent costimulatory signal to tumor antigen-specific T cells. PRS-343 is intended to localize 4-1BB activation in the tumor, and to thereby both increase efficacy and reduce systemic toxicity compared to 4-1BB-targeting antibodies being developed by third parties in clinical trials. We initiated a Phase I dose-escalation study of PRS-343 in HER2 positive patients in September 2017.
Biology of the costimulatory immune receptor 4-1BB
4-1BB, is a co-stimulatory immune receptor and a member of the tumor necrosis factor receptor, or TNFR, super-family. It is mainly expressed on activated CD4+ and CD8+ T cells, activated B cells, and natural killer, or NK, cells. 4-1BB plays an important role in the regulation of immune responses and thus is a target for cancer immunotherapy. 4-1BB ligand (4-1BBL) is the only known natural ligand of 4-1BB, and is constitutively expressed on several types of antigen-presenting cells, or APC. 4-1BB-positive T cells are activated by engaging a 4-1BBL-positive cell. The induced 4-1BB clustering leads to activation of the receptor and downstream signaling. Note that the trimeric 4-1BBL as a soluble molecule is not an effective 4-1BB agonist, providing evidence that larger scale clustering is required for activation. In a T cell pre-stimulated by the T cell receptor binding to a cognate Major histocompatibility complex, or MHC, target, costimulation via 4-1BB leads to further enhanced activation, survival and proliferation, as well as the production of pro-inflammatory cytokines and an improved capacity to kill.
Validation of 4-1BB as a therapeutic target in cancer
The benefit of 4-1BB costimulation for the elimination of cancerous tumors has been demonstrated in a number of in vivo models in the mouse. The forced expression of 4-1BBL on a tumor, for example, leads to tumor rejection. Likewise, the forced expression of an anti-4-1BB single chain antibody fragment (scFv) on a tumor leads to a CD4+ T cell and NK-cell dependent elimination of the tumor. A systemically administered anti-4-1BB antibody has also been demonstrated to lead to retardation of tumor growth.
Human ex vivo data support the extraordinary potential of 4-1BB as a costimulatory receptor in cancer therapy: It has been reported that for T cells isolated from human tumors, 4-1BB is an effective marker for those that are tumor-reactive. Based on this observation, we believe anti-4-1BB antibodies can be utilized to improve adoptive T cell therapy (ACT) by augmenting the expansion and activity of CD8+ melanoma tumor-infiltrating lymphocytes.
Finally, the potential of 4-1BB targeting has also been shown in nonclinical combination therapy studies, where an additional benefit was demonstrated by combination of 4-1BB agonism with checkpoint blockade or NK cell-targeting antibodies.
Current approaches to clinical 4-1BB targeting
The preclinical demonstration of the potential therapeutic benefit of 4-1BB costimulation has spurred the development of therapeutic antibodies targeting 4-1BB, PF-05082566 (22, 23) and BMS-663513, which are currently in early phase clinical trials.
PF-05082566 is a fully humanized IgG2 monoclonal antibody that binds 4-1BB in a manner that blocks the binding of endogenous 4-1BBL to 4-1BB, and that according to publicly available data is well tolerated as a monotherapy and in combination with rituximab.
BMS-663513 is an IgG4 monoclonal antibody that, in contrast to PF-05082566, binds 4-1BB in a manner that does not interfere with the 4-1BB / 4-1BBL interaction. While an initial trial reported manageable toxicity with doses up to 10mg/kg, a follow-up monotherapy Phase II trial was reported to have been stopped due to an “unusually high incidence of grade 4 hepatitis”. Current clinical trials with BMS-663513 are focusing on safety and efficacy at lower doses as monotherapy or in combination e.g. with Rituximab (NCT01775631).
Rationale for bispecific targeting of 4-1BB
We believe that the natural mode of activation of 4-1BB, which requires receptor clustering, demonstrates that an ideal 4-1BB-targeting agent should firstly lead to clustering of 4-1BB, and secondly do so in a tumor-localized fashion on tumor-infiltrating lymphocytes or TILs. The antibodies currently in clinical development are not ideal in that respect, as 4-1BB clustering can only be induced by binding to Fcg receptor-positive cells, which are not selectively tumor-localized but distributed throughout the body for Fcg-dependence of TNFR targeting. The toxicity data of BMS-663513 indicates that such a non-selective activation leads to unacceptable toxicity, potentially making it impossible to find a therapeutic window for such 4-1BB-targeting antibodies.
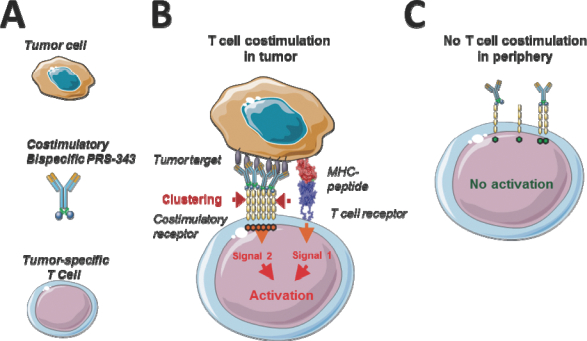
We therefore hypothesized that to obtain an ideal 4-1BB-targeting agent, a bispecific molecule should be designed that targets 4-1BB on one end and a differentially expressed tumor target on the other end. A visualization of the general concept is provided in Figure 1, below. HER2/4-1BB bispecific is envisioned to promote 4-1BB clustering by bridging T cells with HER2-positive tumor cells, and to thereby provide a potent costimulatory signal to tumor antigen-specific T cells, further enhancing its T cell receptor, or TCR, -mediated activity and leading to tumor destruction.
|
| |
Figure 1 | Concept of costimulatory T cell engagement. (A) The elements of the system are a target-positive tumor cell, a T cell with a TCR that is specific for an HLA/peptide combination on the tumor, and a costimulatory bispecific. (B) Within a patient´s tumor, tumor-specific T cells are bridged with tumor cells by a costimulatory bispecific. The resulting clustering of the costimulatory T cell receptor provides a local co-activating signal to the T cell, further enhancing its TCR-mediated activity and leading to tumor destruction. (C) Toxic side effects are expected to be manageable, as target-negative cells do not lead to costimulation of T cells due to a lack of target-mediated receptor clustering, and healthy tissue is spared by tumor-costimulated T cells due to the absence of a primary, TCR-mediated signal. Design and Generation of HER2/4-1BB bispecific PRS-343 |
To obtain a molecule that would work by the mode of action of costimulatory T cell engagement, we generated the HER2/4-1BB bispecific PRS-343. The molecule consists of two different building blocks binding to the two targets HER2 and 4-1BB. To generate the 4-1BB-specific building block of PRS-343, we utilized Anticalin technology. A 4-1BB-binding Anticalin protein was generated based on a re-design of the natural binding pocket of NGAL using mutant Anticalin libraries and a selection and screening process. The lead 4-1BB-binding Anticalin Protein binds human 4-1BB with an affinity of 2 nM as determined by SPR, and is capable of costimulating human T cells when immobilized on a plastic dish together with an anti-CD3 antibody.
To generate the bivalent HER2/4-1BB bispecific PRS-343, we constructed a genetic fusion of a 4-1BB-specific Anticalin protein to the C-terminus of each heavy chain of the trastuzumab IgG4 variant, connected by a flexible, non-immunogenic 15 amino acid linker sequence.
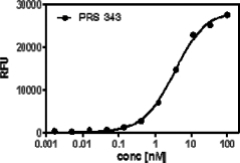
We utilized a Sandwich ELISA experiment to investigate whether PRS-343 can bind both targets at the same time, which is a necessary prerequisite for the envisioned mode of action of PRS-343. Figure below shows that a sigmoid binding curve results from this titration, proving that both targets can indeed be engaged at the same time, fulfilling the key requirement for simultaneous costimulatory engagement of T cells by HER2-positive target cells.
|
| |
Figure 2 | PRS-343 simultaneous binding to targets HER2 and 4-1BB. Recombinant Her2 was coated on a microtiter plate, followed by titration of PRS-343. Subsequently, a constant concentration of biotinylated human 4-1BB was added, which was detected via a peroxidase-conjugated avidin variant, ExtrAvidin®. |
Mode of action – costimulatory T cell activation
We developed a novel T cell activation assay format to investigate whether PRS-343 is capable of costimulating T cells that have received a basic stimulus via the TCR. The assay, visualized in Figure 3 below, is based upon providing the T cell receptor stimulus via an anti-CD3 antibody coated onto the plastic culture dish, while 4-1BB costimulation is achieved by tumor-target dependent clustering of 4-1BB on purified T cells.
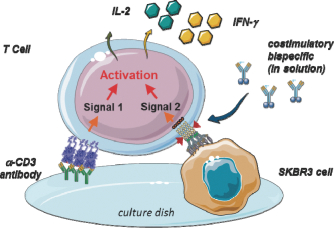
|
| |
Figure 3 | Visualization of costimulatory T cell activation assay. HER2-positive tumor cells are grown overnight on cell culture plates that have been precoated with low amounts of an anti-CD3 antibody to provide a limited primary activation of T cells via the T cell receptor. T cells are added to the wells together with the titrated 4-1BB/HER2 bispecific PRS-343, leading to clustering of the costimulatory 4-1BB receptor, which in turn results in T cell costimulation. T cell costimulation is detected by increased supernatant IL-2 and IFN-g levels in the culture supernatants after continued culture. |
There is a clear induction of IL-2 (Figure A) and IFN-γ (Figure C) with increasing concentrations of PRS-343. The fitted EC50 of this effect is similar for both proinflammatory cytokines, with 0.7 nM for IL-2 induction and 0.3 nM for IFN-ã
induction, respectively. That T cell costimulation is indeed, due to the bispecific engagement of T cells and SKBR3 cells, shown by two observations: firstly, the monospecific antibody trastuzumab does not lead to enhanced T cell activation (average shown as dotted line in Figure A and Figure C), and secondly, disrupting the bispecific interaction with an excess of trastuzumab abolishes the effect of IL-2 and INF-g induction almost completely, except at the highest concentrations of PRS-343 employed (Figure B and Figure D).
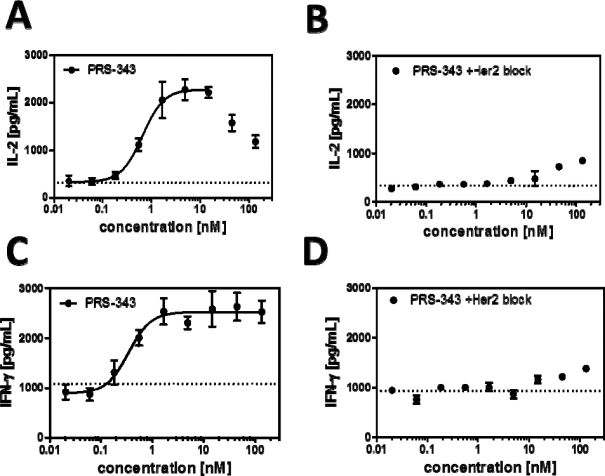
|
| |
Figure 4 | Experimental result of costimulatory T cell activation assay. HER2-positive SKBR3 tumor cells were grown overnight on 96-well plates that had been precoated with 0.25 µg/mL anti-CD3 antibody for 1 h at 37°C. The next day, T cells purified from healthy donor PBMC were added to the wells together with the titrated 4-1BB/HER2 bispecific PRS-343 (filled circle) or trastuzumab as a control (dotted line). After three days in culture, IL-2 (A) and IFN-g, levels in the culture supernatants were measured by an Electrochemoluminescence immunoassay. In parallel, the experiment was performed in the presence of an excess of trastuzumab (340 nM) to inhibit the binding of PRS-343 to the SKBR3 cells, and IL-2 (C) and IFN-g (D) levels were measured. |
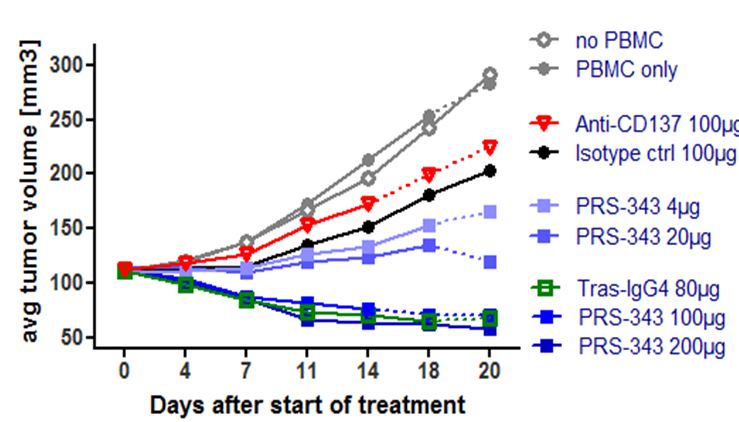
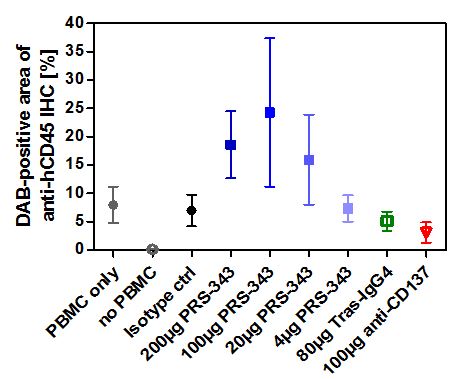
In vivo proof of concept data utilizing a humanized SK-OV-3 mouse model demonstrated dose-dependent tumor growth inhibition compared to treatment with the isotype control (Figure 5). It is anticipated that the TGI in this model is predominantly caused by the anti-HER2. The anti-tumor response observed with PRS-343 was accompanied by a significantly higher tumor infiltration with human lymphocytes (hCD45+). Interestingly, the anti-4-1BB benchmark neither displayed tumor growth inhibition nor enhanced lymphocyte infiltration into tumors compared to isotype. The tras-IgG4 control was also devoid of lymphocyte infiltration into the tumor, but displayed a tumor growth inhibition comparable to PRS-343. Taken together, these data show that PRS-343 provided dual activity by both increasing the frequency of tumor-infiltrating lymphocytes by bispecific targeting of CD137 and HER2 as well as mediating direct tumor growth inhibition by the direct, monospecific targeting of HER2.
|
| |
Figure 5 | PRS-343 activity in NOG mice engrafted with HER2-positive SK-OV-3 cell line and human PBMC. (A) Median of tumor growth. (B) Frequency of CD45+ cells determined by immunohistochemistry of tumors after study end. |
PRS-332 targeting an undisclosed checkpoint target
PRS-332 is a bispecific Anticalin-antibody fusion protein comprising an anti-PD-1 antibody genetically fused to an Anticalin molecule targeting an undisclosed checkpoint target. Other drug candidates targeting the checkpoint molecule PD-1 include nivolumab, marketed by Bristol Myers Squibb (trade name Opdivo), and pembrolizumab, marketed by Merck, (trade name Keytruda). There are currently a number of approved PD-1 or PD-L1 inhibitors approve in the US. Anti-PD-1 antibodies have demonstrated great clinical benefit in several cancers, including melanoma, non-small cell lung cancer, renal cell carcinoma, Hodgkin lymphoma, head and neck carcinomas, bladder cancer and others. However, there are many patients who do not respond, relapse or acquire resistance to PD-1 treatment. In order to improve on existing PD-1 therapies, Pieris in collaboration with Servier is developing PRS-332 with the intent to simultaneously block PD-1 and another immune checkpoint co-expressed on exhausted T cells.
Pipeline products: 300 Series
Current antibody-based therapies targeting tumor cell destruction or immune activation are hampered by, among other factors, low response rates and the induction of immune-related adverse events. The 300-Series Anticalin proteins are designed to target checkpoint proteins or, like PRS-343, immune-stimulatory proteins and consist of a variety of multifunctional biotherapeutics that can combine, via a genetic fusion, antibodies with Anticalin proteins or two or more Anticalin proteins to each other. These combined molecules have the potential to build upon current therapies through the capability of modifying or regulating one or more immune functions on a single fusion protein, thereby having the potential to elevate immune responses within a tumor microenvironment. We believe that a tethered Anticalin protein directed at checkpoint or co-stimulatory targets can preferentially activate the immune system at the site of the tumor microenvironment thus providing efficacy with enhanced therapeutic index. We believe that the 300-Series Anticalin proteins represent a “platform within a product” opportunity in IO since it may be possible to apply a single combined Anticalin-antibody molecule in a number of different cancers. This is based
on the shared underlying biology such as checkpoint and costimulatory biology found within tumors arising in different organs. Pieris intends to expand and accelerate its IO pipeline, including anticipated IND filings in 2019.
AstraZeneca Collaboration:
PRS-060 targeting IL-4Rα in asthma
PRS-060 is an Anticalin drug candidate targeting IL-4Rα, a cell surface receptor expressed on immune cells in the lung epithelium and sub-mucosal layer. IL-4Rα is specific to the circulating cytokine IL-4 and the closely related cytokine IL-13, both key drivers of the immune system. PRS-060 is derived from human tear lipocalin, has picomolar affinity for human IL-4Rα (20 pM) and has a favorable stability profile. We showed in vitro that PRS-060 can inhibit the activity of both IL-4 and IL-13. We have developed a bioprocess that has generated GMP material for use in preclinical safety studies and first in human clinical studies; demonstrated that PRS-060 is sufficiently safe and well tolerated in preclinical safety studies to allow progression to first-in-human-clinical evaluation and that PRS-060 can be formulated for pulmonary delivery by inhalation. Our first-in-human clinical trial for PRS-060 was initiated in December 2017. We believe PRS-060 represents a first in class inhaled biologic targeting IL-4Rα for the treatment of asthma.
Asthma market
Asthma is a very common chronic airway disorder affecting approximately 300 million people worldwide according to the Global Initiative for Asthma and approximately 26 million Americans according to the U.S. Centers for Disease Control. Of these 26 million, about 7 million are children. Asthma is responsible for 13 million physician visits a year including about 2 million emergency visits in the United States, according to the American Lung Association. In the United States between 2008-2013, asthma was responsible for $3 billion in losses from missed work and school days, $29 billion from asthma-related mortality, and $50.3 billion in medical costs. This resulted in a total cost of asthma in the U.S. of $81.9 billion in 2013 (Nurmagambetov, Kuwahara and Garbe, 2018, Annals of the American Thoracic Society, http://www.atsjournals.org/doi/pdf/10.1513/AnnalsATS.201703-259OC, In Press).
Challenges in using conventional therapy
According to a 2016 Artisan Health Care Consulting analysis, asthma affects approximately 80 million people in the U.S. and Europe. The analysis determined that approximately 15%, or 12 million, of the group studied were considered >12 years old and have moderate to severe uncontrolled asthma.
The current standard of care for persistent, moderate to severe allergic asthma is high dose inhaled corticosteroids or ICS often in combination with inhaled long-acting beta-adrenergic agonists, or LABA. In very severe allergic asthma, omalizumab (Xolair from Roche) is given to patients in addition to ICS/LABA combinations. Omalizumab (Xolar®) was approved for this condition in the United States in 2003. Outside of the United States, omalizumab is approved for severe asthma. Omalizumab works by binding to the immune mediator immunoglobulin E, or IgE, and inhibiting IgE-mediated activation of mast cells and basophils, types of white blood cells. It has also been shown to impact some diseases, such as asthma, that are driven by eosinophils, another important class of immune cells. However, patient response to omalizumab has been shown to be inconsistent, as reported in a publication by McNicholl and Heaney in 2008 in the journal Core Evidence, which explained that in only some studies did omalizumab improve lung function. Furthermore, general asthma symptoms are also typically unaffected by omalizumab. Finally, in 2007, the U.S. Food and Drug Administration, or the FDA, issued a black box warning for omalizumab due to reported cases of anaphylaxis, a potentially life-threatening allergic reaction suffered by some patients who had taken the drug. Despite these shortcomings worldwide sales of omalizumab in 2015 were, according to a report by Datamonitor Healthcare published on September 19, 2016, $1.9 billion.
The next generation of therapies beyond omalizumab targets a broader range than just IgE mediated mechanisms. These approaches target other immune mediators, including IL-5, IL-4, IL-13, thymic stromal lymphopoietin, or TSLP, IL-33 (which act in concert on eosinophils, B cells, epithelial cells, goblet cells and others) and PGD2 (through stimulation of CRTH2 receptors). Asthma is associated with high levels of eosinophils, immune cells that play a role in protecting the body against infection. The creation of eosinophils can be interrupted at the early stages, while the cells are still maturing. Multiple products are approved that target eosinophils through IL-5 or its receptor IL-5RA. GlaxoSmithKline’s, or GSK's, mepolizumab (Nucala) which targets IL-5 was approved for severe eosinophilic asthma in adults and children older than 12 in 2015. Teva’s Cinqair also targeting IL-5 was approved in 2016 and more recently AstraZeneca’s Fansera, which targets the IL-5RA, was approved in November 2017. However, eosinophils are only one of many cell types and immune system components that are involved with the body’s exaggerated inflammation response in asthma. Mast cells, basophils, goblet cells and other cells also play a role. These cells can be seen infiltrating the airways along with eosinophils, leading to the conclusion that more cell types are
involved in asthma pathogenesis. We believe that targeting just one of these components unlikely to be as effective in treating severe asthma as an approach that targets the broader Th2 (cell-mediated) pathway.
In 2017, Regeneron and its partner Sanofi reported that they had successfully completed a phase 3 study for dupilumab in severe, steroid-dependent asthma. Dupilumab (Dupixent), is a monoclonal antibody that targets IL-4Rα that is delivered subcutaneously and was approved for the treatment of moderate to severe atopic dermatitis in March 2017. In a 2013 paper in the New England Journal of Medicine, Wenzel et al. reported that dupilumab showed a benefit on the asthma control questionnaire 5 (ACQ5) symptom score, a widely accepted measure for classifying the ability of a medication to control asthma. Patients dosed with dupilumab had fewer asthma attacks compared to placebo-treated patients when standard therapies, such as long-acting beta-agonists and inhaled glucocorticoids, were withdrawn, demonstrating the efficacy of dupilumab. Patients also showed improved lung function and reduced levels of Th2-associated inflammatory markers. Dupilumab is administered systemically through injection. In November 2014, Regeneron and Sanofi announced that in a Phase IIb study, dupilumab also demonstrated improved lung function and reduced exacerbations when administered together with standard of care. These effects were observed in both unselected severe asthma patients and selected patients presenting elevated Th2 responses. In their October 2017 press release Regeneron and Sanofi announced that dupilumab had met its primary endpoint and key secondary endpoints the Liberty Asthma Venture phase 3 study in adults and adolescents with severe, steroid dependent asthma. In the overall population, dupilumab reduced the use of oral corticosteroids (OCS) by 70% compared to 42% for patients on placebo. For patients with a high baseline eosinophilia (300 or greater eosinophilic cells/microliter) the reduction in OCS use was 80% compared to 43% for placebo. Dupilumab also reduced the number of exacerbations in the overall population by 59% and this reduction was greater (71%) in patients with high eosinophil counts. In addition, dupilumab compared to placebo also improved lung function, Forced Expiratory Volume in 1 Second (FEV1), by 220ml in the overall population and 320ml in patients with high eosinophils. This data has not yet been published in a peer reviewed journal. The Liberty Asthma Quest phase 3 study showed that dupilumab, when added to the standard of care, reduced severe asthma attacks (exacerbations) by 46 percent in the overall population and 67 percent in patients with high eosinophils (300 or greater eosinophilic cells/microliter. The companies submitted a Supplemental Biologics License Application (sBLA) for dupilumab, for the treatment of asthma in patients 12 and over, to the FDA during the fourth quarter of 2017.
Advantages to inhalation as a route of administration for PRS-060
We have demonstrated that PRS-060 is sufficiently safe and well tolerated in inhalation toxicology studies in mice to allow progression to first-in-human-clinical evaluations, and we believe that overall it may have a better tolerability profile than systemically administered monoclonal antibodies. Furthermore, we believe that PRS-060 could be produced at a lower cost of goods than mAbs because we will use manufacturing procedures that employ bacterial expression systems, which generally provides a cost advantage over mammalian production systems, typically used for mAbs. Since dosing by inhalation is a common route of administration in asthma patients, it could represent a more convenient dosage regimen for patients than dosing of antibodies by injection. PRS-060 could therefore be self-administered using a standard device rather than requiring a visit to a physician for the drug to be given.
Preclinical data
In in vitro assays, PRS-060 specifically bound to immobilized targets such as human IL-4Rα in a concentration-dependent manner. We tested the binding of PRS-060 to various targets in an enzyme-linked immunosorbent assay, (ELISA) a standard in vitro assay platform. In these tests, PRS-060 bound to IL-4Rα with subnanomolar affinity and it did not bind to three other human cell-surface interleukin receptors (IL-6R, IL-18RA, IL-23RA). Furthermore, the activity of IL-4 and IL-13 was inhibited by PRS-060 in a dose-dependent manner. The below charts show the inhibition of IL-4 (left) or IL-13 (right) induced proliferation in human TF-1 cells in vitro by PRS-060.
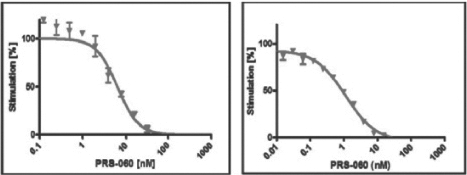
In in vivo assays in mice genetically altered to express human IL-4Rα, human IL-4 and IL-13, low doses of PRS-060 inhibited the induction of eotaxin protein, a marker of airway inflammation, in lung tissue following pulmonary delivery. We observed this inhibition at both the RNA and protein levels compared both to buffer and to wild type tear lipocalin (control).
The below chart shows the duration of PRS-060-mediated inhibition of eotaxin gene expression, a marker of airway inflammation, in lung tissue by a single pulmonary dose in mice:
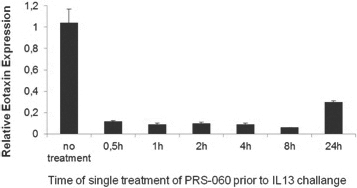
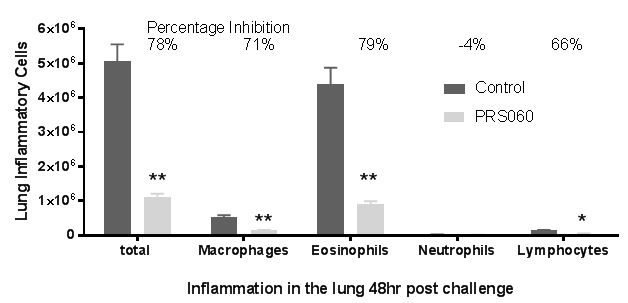
When we administered IL-13 into the lung of humanized mice (that express human IL-4, IL-13 and IL-4RA), inflammation was induced as determined by eotaxin expression, which was not inhibited when phosphate buffered saline, (PBS) or human wild type lipocalin was administered into the lung. In contrast to the PBS administration, increases in eotaxin expression were prevented when PRS-060 was administered into the lung before IL-13. As demonstrated in the above chart, the model showed the inhibitory potential lasts for up to 24 hours after PRS-060 administration. We have also demonstrated that PRS-060 reduces the inflammation associated with antigen challenge in a mouse asthma model. The chart below shows that pre-treatment with PRS-060 reduces the lung levels of the key inflammatory cells eosinophils and lymphocytes, a profile that supports the hypothesis that lung delivery of an IL-4Rα antagonist to asthmatics may be viable approach to the treatment of asthma.
PRS-080 targeting hepcidin in CKD-related FID-anemia
PRS-080 is an Anticalin drug candidate targeting hepcidin, a peptide mediator that is an important negative regulator of iron absorption and storage, derived from the naturally occurring human lipocalin known as NGAL. The normal function of hepcidin is to maintain equilibrium in iron supply for red blood cell production by binding to ferroportin, the protein that transports iron from the inside of a cell to the outside, inducing its internalization and subsequent degradation. The binding of hepcidin to ferroportin reduces the iron uptake from the intestine into the body and inhibits iron mobilization from cellular stores into red blood cells. An excess amount of hepcidin can cause functional iron deficiency (FID), which often cannot be treated adequately with iron supplements and can lead to anemia. According to a 2009 publication by Young and Zaritsky in the Clinical Journal of the American Society of Nephrology, lowering hepcidin levels or antagonizing its actions would reverse the negative effects of inflammation on red blood cell formation by allowing mobilization of stored iron and improved iron absorption.
PRS-080 has been designed to target hepcidin for the treatment of FID in anemic patients with CKD, particularly in end-stage renal disease patients requiring dialysis, to allow them to mobilize iron that is trapped in iron storage cells for use in the creation of red blood cells. We have also engineered PRS-080 to have a half-life of less than a week, so that following administration, it is expected to clear from the human body in a much shorter timeframe than antibodies, which typically have a
half-life of two weeks or greater. This half-life was achieved by covalently linking PRS-080 to a specific polyethylene glycol, or PEG, in order to extend the serum half-life of the combined molecule to desirable levels. Since hepcidin is constantly produced by the body, we believe that a frequent, e.g. once per week, dosing interval will be optimally suited to interfere with hepcidin function. A shorter half-life than mAbs may be more compatible with this dosing schedule. A longer mAb-like residence time is not seen as advantageous, but rather could lead to the accumulation of both the drug and the target beyond the typical residence time of hepcidin, resulting in large quantities of hepcidin bound to mAbs. We completed a Phase Ia single-ascending dose clinical trial with PRS-080 in healthy volunteers in 2015. The trial was conducted in accordance with German law at a clinical site in Neu-Ulm, Germany, that belongs to Nuvisan GmbH, our contract research organization, or CRO. Results from this trial were presented at the 2015 Annual Conference of the American Society of Hematology (http://www.bloodjournal.org/content/126/23/536). Based on the data obtained. we initiated a Phase Ib clinical study in CKD 5 patients requiring hemodialysis which we completed in February 2017. The company initiated a multi-dose clinical study in CKD patients requiring hemodialysis in the third quarter of 2017, which is assessing the safety and tolerability of multi-dose administration of PRS-080, as well as a secondary assessment of repeated doses of PRS-080 on hemoglobin levels over a period of approximately one month.
Anemia and functional iron deficiency in the CKD population
Anemia is a serious medical condition in which blood is deficient in red blood cells, and hemoglobin, leading to inadequate oxygen delivery to tissues and cells throughout the body. Anemia is generally said to exist when hemoglobin is less than 13 g/dL in men and 12 g/dL in women. Anemia has a number of potential causes, including nutritional deficiencies, iron deficiency, bone marrow disease, medications, and abnormalities in production of or sensitivity to erythropoietin, a hormone that controls red blood cell production. Anemia is a frequent and severe consequence of CKD. In addition, within the CKD population, anemia may be caused by FID. FID exists when, despite adequate stores, iron cannot be mobilized for erythropoiesis. In this case, despite treatment with exogenous erythropoietin and iron supplements, “functional” iron is still deficient. FID-anemic patients can be identified and selected for therapy using marketed laboratory tests for iron metabolism. According to the results of a 2017 research analysis conducted for us by Artisan Healthcare Consulting, there were an estimated 505,000 individuals with ESRD that were on hemodialysis in the US in 2017 and approximately 90% of these patients are treated with ESA. Up to 50% of the ESRD patients on hemodialysis that are currently treated with ESA have FID. Approximately 15% of the FID patients are still anemic despite ESA treatment. Based on the estimated 505,000 individuals with ESRD on hemodialysis, we believe that approximately 34,000 individuals are FID-anemic despite ESA treatment in the US.
Challenges in using conventional therapy
We believe CKD patients with FID-anemia are especially poorly served. These patients have adequate stores of iron but this iron is not efficiently incorporated into red blood cell precursors through rESAs and iron supplements. According to the 2009 publication by Young and Zaritsky in the Clinical Journal of the American Society of Nephrology, this imbalance in iron metabolism is a result of a high level of circulating hepcidin in the blood stream. We believe existing therapies are limited in that they do not have an impact on hepcidin or, in the case of rESAs, patients often become resistant to the therapy.
Our potential solution: binding hepcidin with PRS-080
We have engineered PRS-080 so that it binds to hepcidin and reduces the impact of hepcidin’s negative regulation on iron mobilization. We believe that by blocking the actions of hepcidin, PRS-080 will serve to address anemia by mobilizing iron for incorporation into red blood cells.
In patients suffering from anemia of CKD, and specifically in patients with FID, hepcidin is chronically produced by the body in abnormally large amounts. Therefore, we believe that the best way to inhibit its function is to administer an inhibitor on a repeated basis, such as once a week. Our approach will use PRS-080 in connection with a conjugated PEG30 molecule, a well-known half-life extender, in order to allow the drug sufficient residence time in the body. Once coupled to PEG30, PRS-080 is intended to have a half-life that will be optimally suited for dosing anemic patients with CKD. In contrast, antibodies typically have a half-life of two to three weeks. Such a long half-life renders antibodies unsuitable for frequent administration and elimination of a circulating target protein like hepcidin because such antibodies tend to accumulate the target after binding due to their own long residence time in the body with the associated risk of bound hepcidin being released by antibodies that are still circulating in the blood.
Preclinical data
Hepcidin binds to ferroportin and induces its internalization and subsequent degradation, thus disabling iron mobilization from cells. PRS-080 binds strongly to hepcidin and inhibits its activity in a dose dependent manner as shown in in vitro potency assays. PRS-080 is able to completely inhibit the internalization of FPN above a concentration of 20 nM.
Our preclinical studies targeted the cynomolgus monkey orthologue of hepcidin, which has a high degree of similarity (96% identity) with human hepcidin. PRS-080 was found to bind with high affinity to the cynomolgus monkey version of hepcidin.
We performed a dose finding study in cynomolgus monkeys, testing intravenous 30-minute infusions as well as subcutaneous injections of PRS-080 to study PK properties of PRS-080 and the functional consequences of hepcidin inhibition on iron mobilization. A dose of 1 mg/kg PRS-080 produced a robust, transient and reversible increase in total iron levels from approximately 36 µM at baseline to 52 µM after 8 hours. Doses higher than 1 mg/kg elevated serum iron concentrations to comparable levels and, in a dose-dependent manner, prolonged the response. A linear correlation was observed over time between the PRS-080 dose and increase of serum iron concentrations. The PK properties of PRS-080 were investigated after a single administration at doses ranging from 20 mg/kg to 150 mg/kg. The concentration over time profiles of PRS-080 showed standard drug-like properties, as the kinetics were dose proportional and there was a low volume of distribution. Elimination of PRS-080 occurred with a terminal half-life of about 2 days, which can be extrapolated to translate to 3 days in humans.
We also carried out a 4-week repeated dose toxicology study with intravenous infusions of PRS-080 for 30 minutes every other day. Our work included toxicokinetic and ADA measurements. During the study, safety pharmacology parameters on the cardiovascular system and respiration were monitored and all safety endpoints were met. Our preclinical studies also examined a different NGAL-derived Anticalin, or surrogate molecule, which targets rat hepcidin in a rat model of inflammation-induced anemia. In these studies, administration of the surrogate molecule once per day or every other day inhibited the manifestation of anemia in the rats over the course of a three-week period.
The 4-week repeated PRS-080 dosing to cynomolgus monkeys was well tolerated up to the highest tested dose of 120 mg/kg. This dose was classified as producing no adverse events: routine laboratory tests and blood cell examinations did not demonstrate any adverse findings and safety pharmacology investigations were also without adverse events. As a result of the hepcidin inhibition, the study showed increased iron uptake and storage, for example in the liver, and mobilization.
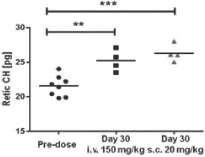
The functional consequence of PRS-080 treatment on bone marrow activity and red blood cell production, or hematopoiesis, by means of hemoglobin (an oxygen transporting protein contained in red blood cells) concentration in reticulocytes, a precursor of red blood cells, was investigated in cynomolgus monkeys following repeated administration. As shown in the below chart, after administration of PRS-080 either intravenously (i.v. 150 mg/kg) or subcutaneously (s.c. 20 mg/kg), elevated hemoglobin concentrations in reticulocytes (Retic CH) were observed on day 30 compared to pre-treatment (pre-dose).
Phase I trial design and results
The Phase Ia trial of PRS-080 was conducted in healthy volunteers at a clinical site in Neu-Ulm, Germany by Nuvisan GmbH, a CRO. The study was a single dose escalating, blinded, placebo controlled study at a dose range from 0.2 to 40 mg/kg (equivalent to 0.08 to 16.0 mg/kg based on protein content). Forty-eight subjects were dosed with PRS-080 or a placebo. This study was governed and approved by the German Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, or BfArM) and the local Ethics Committee. Treatment of subjects began at the end of 2014 and was completed in June 2015, followed by evaluation of the data.
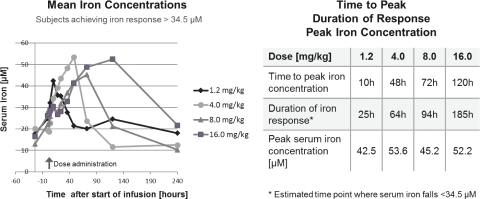
PRS-080 was well tolerated. All treatment emergent Adverse Events, or AEs, were either mild or moderate and no Serious AEs were observed. No association of AEs to specific organs and no apparent dose dependency or difference between placebo and active treatment was observed. Notably, no hypersensitivity or infusion reactions were noted and vital signs, body temperature and electrocardiograms were unchanged. Pharmacokinetics of PRS-080 were dose-proportional with a half-life of approximately 3 days. PRS-080 administration resulted in an immediate decrease in plasma hepcidin concentration, which was followed by an increase in serum iron concentration and transferrin saturation. As shown in the figure below, the duration of
this response in iron and transferrin saturation increased dose-dependently from about 25 hours at the lower dose to about 185 hours at the highest dose.
Phase Ib trial in anemic CKD 5 patient
Based on this positive safety and pharmacological activity we initiated a Phase lb study in CKD 5 patients undergoing hemodialysis and suffering from FID-anemia earlier in 2016. This study was governed and approved by the German Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, or BfArM). It was a multi-center, randomized and double blinded Phase Ib trial studying single PRS-080 administrations at 3 dose levels, 2, 4, and 8 mg/kg bw. This study was completed in the first quarter of 2017. In each dose group 6 patients were treated with PRS-080 and 2 patients with placebo. PRS-080 was well tolerated and shown to be safe in all dose groups. There was only one SAE (worsening of dry gangrene in a patient with long standing diabetic foot syndrome) Vital signs, physical examination, and electrocardiograms were not affected by the treatment with PRS-080. No differences in the safety analysis were observed between placebo and drug. There was no evidence of immunogenicity in the single ascending dose study.
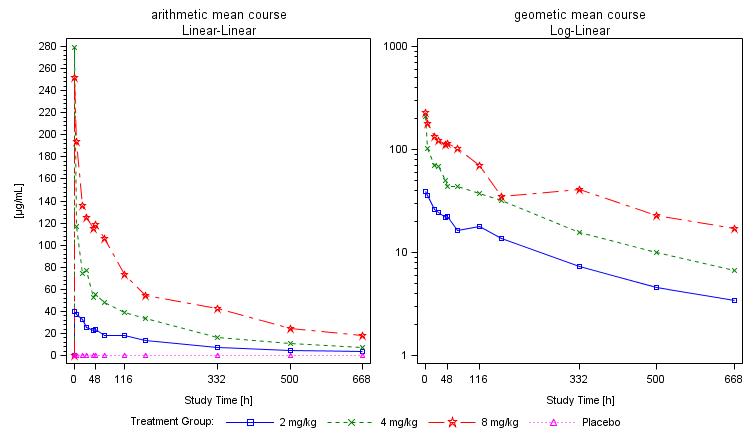
Pharmacokinetics shows a dose-proportional increase for free and total PRS-080 with a dose peak plasma concentration at 1 hour (tmax) and a rapid decrease over the following 19 hours.
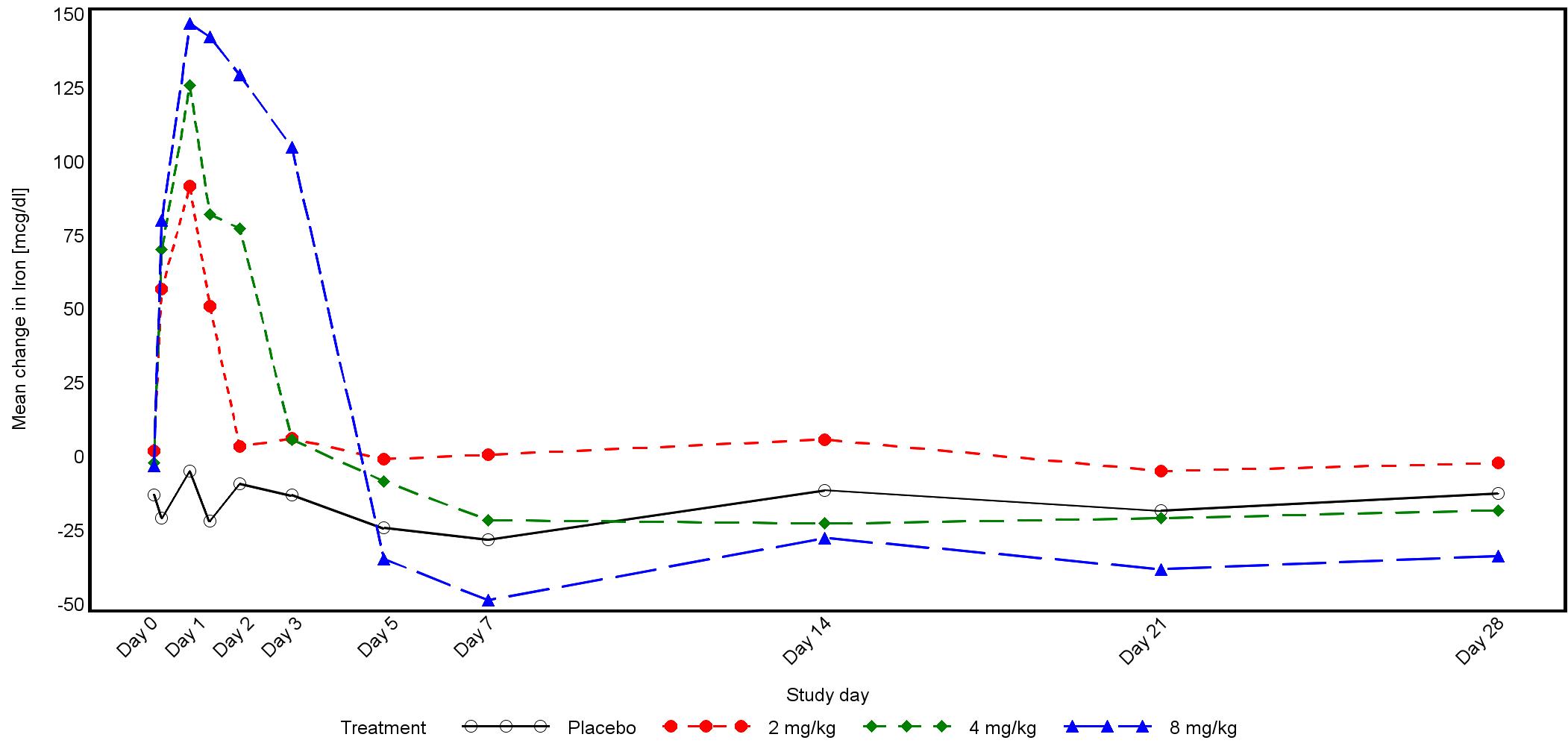
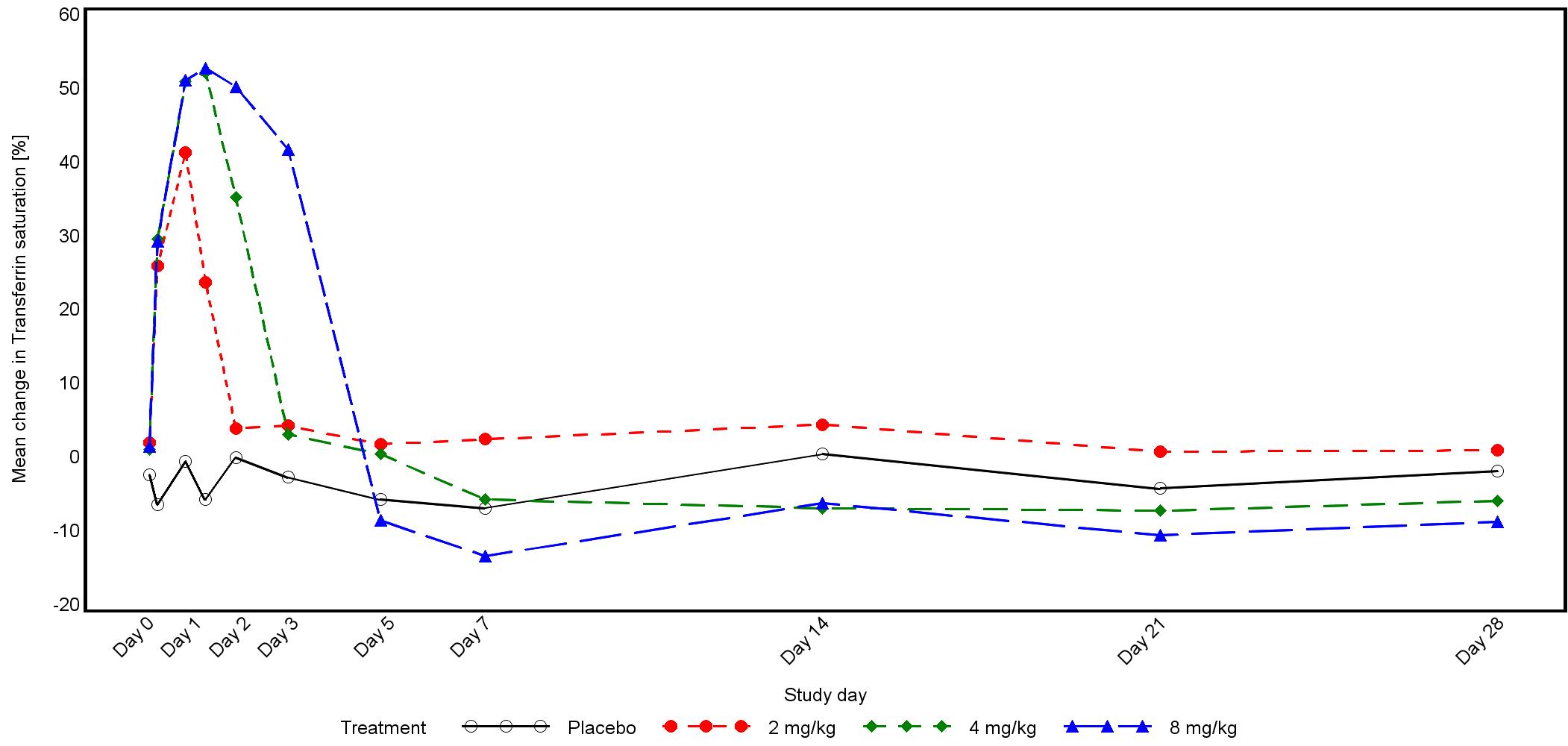
The magnitude of iron mobilization by PRS-080 was dose-dependent with increases in both serum iron concentration and transferrin saturation (TSAT) following treatment. The maximal concentrations (Cmax) of iron and TSAT were reached within 19 hours in all three dose groups. A return to baseline values were reached after 2, 3, and 5 days after the end of infusions of 2, 4, and 8 mg/kg PRS-080, respectively.
The figure below shows mean serum concentration of iron during the study:
The figure below shows mean TSAT during the study:
Subsequently and due to the good safety profile and iron mobilization of PRS-080, a multi-center, placebo controlled ascending dose pilot Phase IIa trial has been initiated to study the safety, pharmacokinetics and effects of hepcidin inhibition on iron mobilization by 5 weekly doses of PRS-080 in 4 and 8 mg/kg patient cohorts. In addition first results on hemoglobin levels in CKD patients will be examined as a secondary outcome measure. In total 12 patients will be included; in each group 4 patients will be treated with PRS-080 and 2 patients with placebo. This Phase IIa study started in September 2017 and is ongoing in clinical centers in Germany and Czech Republic. The German Federal Institute for Drugs and Medical Devices (Bundesinstitut für Arzneimittel und Medizinprodukte, or BfArM) and the Czech State Institute of Drug Control (SUKL) approve and govern the study. First results should be available in the second half of 2018.
Competition
The pharmaceutical and biotechnology industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. While we believe that our technology, development experience, scientific knowledge and strategies provide us with competitive advantages, we face and will continue to face intense competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions and governmental agencies and public and private research institutions, both in the United States and abroad.
We compete, or will compete, with existing and new therapies that may become available in the future. Some of these competitors are pursuing the development of pharmaceuticals that target the same diseases and conditions that our drug candidates target. Any drug candidates that we are able to develop and commercialize will compete with existing and new drugs being developed by our competitors. Our competitors may develop or market products or other novel technologies that are more effective, safer, more convenient or less costly than any that may be commercialized by us, or may obtain regulatory approval for their products more rapidly than we may obtain approval for ours.
The acquisition or licensing of pharmaceutical products is also very competitive, and a number of more established companies, some of which have acknowledged strategies to license or acquire products and many of which are bigger than us and have more institutional experience and greater cash flows than we have, may have competitive advantages over us, as may other emerging companies taking similar or different approaches to product licenses and/or acquisitions. In addition, a number of established research-based pharmaceutical and biotechnology companies may acquire products in late stages of development to augment their internal product lines, which may provide those companies with an even greater competitive advantage.
There are a number of other companies presently working to develop therapies for anemia, asthma, and oncology, including divisions of large pharmaceutical companies and biotechnology companies of various sizes. There are also a variety of available drug therapies marketed for these diseases. Our drug candidates, if any are approved, may compete with these existing drug and other therapies, and to the extent they are ultimately used in combination with or as an adjunct to these therapies, our drug candidates may not be competitive with them. Some of the currently approved drug therapies are branded and subject to patent protection, and others are available on a generic basis. Many of these approved drugs are well established therapies and are widely accepted by physicians, patients and third-party payors. As a result, market acceptance of, and a significant share of the market for, any of our drug candidates that we successfully introduce to the market will pose challenges.
In addition to currently marketed therapies, there are also a number of drugs in clinical development to treat anemia, asthma, or cancer. These medicines in development may provide efficacy, safety, convenience, and other benefits that are not provided by currently marketed therapies and may not be provided by any of our current or future product candidates. As a result, they may provide significant competition for any of our product candidates.
Many of our competitors will have substantially greater financial, technical and human resources than we have. Additional mergers and acquisitions in the pharmaceutical industry may result in even more resources being concentrated in some of our competitors. Competition may increase further as a result of advances made in the commercial applicability of technologies and greater availability of capital for investment in these fields. Our success will be based in part on our ability to build, obtain regulatory approval for and market acceptance of, and actively manage a portfolio of drugs that addresses unmet medical needs and creates value in patient therapy.
In addition, our competitors may have a variety of drugs in development or awaiting market approval that could reach the market and become established before we have a product to sell. Our competitors may also develop alternative therapies that could further limit the market for any drugs that we may develop. Many of our competitors are using technologies or methods different or similar to ours to identify and validate drug targets and to discover novel small molecule drugs. Many of our competitors and their collaborators have significantly greater experience than we do in the following:
| |
• | identifying and validating targets; |
| |
• | screening compounds against targets; |
| |
• | preclinical and clinical trials of potential pharmaceutical products; and |
| |
• | obtaining regulatory clearances. |
In addition, many of our competitors and their collaborators have substantially greater advantages in the following areas:
| |
• | research and development resources; |
| |
• | manufacturing capabilities; and |
Smaller companies also may prove to be significant competitors, particularly through proprietary research discoveries and collaborative arrangements with large pharmaceutical and established biotechnology companies. Many of our competitors have products that have been approved by the FDA, or its foreign counterparts, or are in advanced development. We face competition from other companies, academic institutions, governmental agencies, and other public and private research organizations for collaborative arrangements with pharmaceutical and biotechnology companies, in recruiting and retaining highly qualified scientific and management personnel and for licenses to additional technologies. Developments by others may render our product candidates or our technologies obsolete. Our failure to compete effectively could have a material adverse effect on our business.
PRS-300 series
Under the 300-series, Pieris is developing multispecific immuno-oncology drug candidates. This includes tumor-targeted co-stimulatory molecules, such as PRS-343, and multispecific checkpoint inhibitors, such as PRS-332.
The rationale behind the multispecific tumor-targeted co-stimulatory molecules is to activate the immune system in the tumor microenvironment. Other companies that also develop multispecific drug candidates designed to activate the immune system in a tumor dependent manner by targeting a co-stimulatory receptor, such as 4-1BB, include Roche, Molecular Partners, Alligator Biosciences, and Aptevo Therapeutics. Additionally, there are multiple of drug candidates in clinical trials solely targeting a co-stimulatory receptors including 4-1BB, OX40, GITR, and CD27.
The first checkpoint inhibitor, Ipilimumab, targeting CTLA-4 was approved for the treatment of melanoma patients in 2011 and is being marketed by Bristol-Myers Squibb. Nivolumab from Bristol-Myers Squibb was approved for the treatment of melanoma in 2014 as the first PD-1 inhibitor. Pembrolizumab from Merck & Co was the second PD-1 inhibitor to be approved and the first one in the US. In addition to Nivolumab and Prembrolizumab there are multiple approved checkpoint inhibitors targeting the PD-1/PD-L1 pathway e.g. from Roche, AstraZeneca, Pfizer, and Merck KGaA.
There are also drug candidates in preclinical or clinical testing for other checkpoint targets such as LAG3, TIM3 and B7-H3. Companies developing either dual-checkpoint inhibitors or combinations of two or more monospecific checkpoint inhibitors includes Xencor, F-star, Bristol-Myers Squibb, Merus, Tesaro, Novartis, Merck & Co among others.
Additionally, a number of other companies, such as Amgen, Affimed, Macrogenics, F-Star Molecular Partners, Xencor, Immunocore and Zymeworks, also pursue other multispecific approaches in oncology, which therapies are in clinical or preclinical development.
PRS-343
PRS-343 is bispecific Anticalin-antibody fusion protein targeting 4-1BB and HER2. The mode of action of PRS-343 is to promote 4-1BB clustering by bridging 4-1BB-positive T cells with HER2-positive tumor cells, and to thereby provide a potent costimulatory signal to tumor antigen-specific T cells. Other drug candidates targeting the co-stimulatory receptor 4-1BB include urelumab, which is being developed by Bristol Myers Squibb, and Utomilumab (PF-05082566), which is being developed by Pfizer, both of which are currently in clinical development (Sitetrove, January 13, 2018). In the HER2-positive space, several companies are active with approved, clinical and preclinical drugs candidates. The most prominent company is Roche, having three approved drugs on the market through its subsidiary Genentech. The first drug from Roche targeting HER2 is Trastuzumab, which has been marketed for treatment of breast cancer patients since 1998 and for gastric cancer patients since 2010. The two other drugs are Pertuzumab and Ado-trastuzumab Emtansine which both are marketed for breast cancer patients. In addition to PRS-343, there are also other HER2 targeting drug candidates in clinical development designed to induce an immune response by bridging HER2-postive tumor cell with immune cells e.g. GBR 1302, a bispecific antibody targeting HER2 and CD3, from Glenmark (Pharmaprojects, January 13, 2018).
One company has publicly disclosed a competitor program to PRS-343. MacroGenics presented data on a HER2 and 4-1BB (41BB) bispecific during their R&D day on December 13th, 2016. In addition to MacroGenics other companies have also disclosed 4-1BB-based bispecific drug candidates. Roche and Molecular Partners have both presented data on bispecific drug candidates targeting Fibroblast activation protein (FAP) and 4-1BB. Alligator Bioscience together with Aptevo Therapeutics have disclosed a 4-1BB-based bispecific drug candidate targeting 5T4. However, the 4-1BB-based bispecific drug candidates targeting FAP or 5T4 does not constitute direct competition to PRS-343 since they are targeting a different tumor target and will, thus, likely be developed in different indications or patient populations.
Additionally, other companies such as AstraZeneca, Novartis, Merck, Agenus, Five Prime Therapeutics, Seattle Genetics, GlaxoSmithKline and Celldex have preclinical and clinical development programs focusing on other co-stimulatory targets which include OX40, CD40, GITR, CD27 and ICOS.
PRS-332
PRS-332 is a bispecific Anticalin-antibody fusion protein comprising an anti-PD-1 antibody genetically fused to an Anticalin protein targeting an undisclosed checkpoint target. Other drug candidates targeting the checkpoint molecule PD-1 include Nivolumab, marketed by Bristol Myers Squibb, and Pembrolizumab, marketed by Merck & Co. Anti-PD-1 antibodies have demonstrated great clinical benefit in several cancers, including melanoma, non-small cell lung cancer, renal cell carcinoma, Hodgkin lymphoma, head and neck carcinomas. However, there are many patients who do not respond, relapse or acquire resistance to PD-1 treatment. In order to improve on existing PD-1 therapies, Pieris is developing PRS-332 with the intent to simultaneously block PD-1 and another immune checkpoint co-expressed on exhausted T cells.
Additionally, other companies such as Merck & Co, Bristol-Meyer Squibb, Xencor, Tesaro, and Novartis are developing drugs targeting PD-1 or PD-L1 an additionally checkpoint target either as a combination product or as a bispecific dual checkpoint inhibitor. The other checkpoint target in such approaches include CTLA-4, LAG-3 and TIM-3.
PRS-060
Like PRS-060, new developments for the treatment of uncontrolled moderate to severe asthma patients mainly include drug candidates targeting the Th2 pathway by interfering with IL-4/IL-13, IL-5 actions or thymic stromal lymphopoietin (TSLP). Such agents include dupilumab (Sanofi/Regeneron, IL-4RA), mepolizumab (Nucala) (GSK, IL-5), reslizumab (Cinqair) (Teva, IL-5), benralizumab (Fasenra) (Astra Zeneca, IL-5RA) and tezepelumab (Amgen/AstraZeneca, TSLP),). These drugs are in later clinical development (Phase II) than PRS-060, or have been approved (mepolizumab, reslizumab, benralizumab) for severe eosinophilic asthma; the mAb omalizumab, directed against IgE, is also approved and marketed for the treatment of uncontrolled, moderate to severe asthma patients. However, in contrast to PRS-060, these mAbs are given to patients through injection and distribute systemically through the blood stream. CSJ117 (Novartis), an inhaled fAb fragment that targets TSLP, is currently in Phase I clinical development. There are a number of other companies presently marketing or developing other therapies for asthmatic patients.
PRS-080
There are very few other drug candidates in development that interfere with hepcidin function or expression. Nucleic acid based approaches that were in preclinical development by IONIS/Xenon and Alnylam have been suspended for unknown reasons. Noxxon’s RNA aptamer NOX-H94 has completed Phase II clinical studies in cancer and ESRD patients. While an increase of Hb values was seen in cancer patients, no such effect could be confirmed in the ESRD population. We are not aware of any ongoing development for NOX-H94. PRS-080 is significantly more potent and has a longer half-life than NOX-H94. We therefore believe that Noxxon’s results are not predictive for efficacy of PRS-080. Lilly has been developing a mAb against hepcidin in cancer as well as chronic kidney disease patients as well as a mAb against the ferroportin transporter. The latter has been suspended for unknown reasons and there has been no update on the anti-hepcidin mAb from Lilly since 2015. Ferrumax develops a soluble form of hemojuvelin, a protein that regulates hepcidin expression and iron metabolism that aims to suppress the production rate of hepcidin.
There are also a number of companies which are focused on treating anemia in CKD patients under alternative approaches. Fibrogen (in partnership with Astellas and AstraZeneca), Akebia Therapeutics (in partnership with Mitsubishi Tanabe and Otsuka), GSK, Bayer, Daiichi Sankyo, Zydus Cadila and Japan Tobacco have hypoxia-inducible-factor prolyl hydroxylase (HIF-PH) inhibitors in clinical development that target stimulation of bone marrow activity. Fibrogen reported positive topline results from phase 3 trials in China with its HIF-PH inhibitor Roxadustat (in January 2017). Akebia recently reported positive results from a Phase II trial in Asia with its HIF-PH inhibitor Vadadustat (January 2018). Multiple phase 3 trials are currently ongoing with different HIF-PH inhibitors in both dialysis-dependent as well as dialysis-independent CKD patients. Acceleron is targeting the sequestration of Activin A, a natural inhibitor of hematopoiesis, is in a Phase II clinical study. Auryxia by Keryx, which is an oral, absorbable, iron-based phosphate binder, completed a phase 3 in non-dialysis dependent CKD 3-5 patients in 2016 and announced topline results showing that the study met the primary and all pre-specified secondary endpoints including change in Hb values. Keryx filed a New Drug Application, or NDA for the CKD indication with the FDA in November 2017. There are also various companies conducting late-stage development of erythropoietin biosimilars.
Manufacturing
We do not own or operate, and currently have no plans to establish, any manufacturing facilities. We currently rely, and expect to continue to rely on third-party contract manufacturers, or CMOs, for the manufacture of our drug candidates for larger scale preclinical and clinical testing, as well as for commercial quantities of any drug candidates that are approved.
We currently rely on multiple CMOs for all of our clinical supplies, including active pharmaceutical ingredients (APIs), drug substances and finished drug products, and label & packaging for our preclinical research and clinical trials, including the Phase IIa trial for PRS-080, the Phase I trial for PRS-343 and the Phase I trial for PRS-060
We believe that we will be able to contract with other CMOs to obtain APIs if our existing sources of APIs were no longer available or sufficient, but there is no assurance that APIs would be available from other third-party manufacturers on acceptable terms, on the timeframe that our business would require, or at all. We do not have supply commitments or other arrangements in place with our existing CMOs. We also do not currently have arrangements in place for redundant supply of bulk drug substance.
We do not have any current contractual relationships for the manufacture of commercial supplies of any of our drug candidates if they are approved, and we intend to enter into agreements with a third-party contract manufacturer and one or more back-up manufacturers for the commercial production of our product candidates as they near potential approval.
Any drug products to be used in clinical trials and any approved product that we may commercialize will need to be manufactured in facilities, and by processes, that comply with the FDA’s current good manufacturing practice requirements and comparable requirements of the regulatory agencies of other jurisdictions in which we are seeking approval. We currently employ internal resources to manage our manufacturing contractors.
We believe that PRS-080, PRS-060, and PRS 343 and our other Anticalin®-branded drug candidates can be manufactured in reliable and reproducible biologic processes from readily available starting materials. PRS-080 and PRS-060 are produced using bacterial expression systems similar to those that have been used in the past for the production of other proteins and which systems are widely used in the industry. PRS-343 is produced using a mammalian expression system similar to those systems which are widely used in the industry for the production of antibodies. We believe that the manufacturing process is amenable to scale-up and will not require unusual or expensive equipment. We expect to continue to develop, on our own or with our collaborators, drug candidates that can be produced cost-effectively at contract manufacturing facilities.
Intellectual Property and Exclusivity
Our commercial success depends in part on our ability to obtain and maintain exclusivity of our proprietary Anticalin technologies through intellectual property protection for our drug candidates, libraries of different protein scaffolds and consensus sequences, and the fundamental Anticalin platform technology, including novel therapeutic and diagnostic discoveries, as well as other proprietary know-how, and to operate without infringing on the intellectual property rights of others.
We seek to protect our exclusive position of Anticalin technologies by, among other means, prosecuting our own international, U.S., and foreign patent applications related to our proprietary technology, inventions, and improvements that are important to the development and implementation of our business. We have established intellectual property protection in relation to our Anticalin technologies in key global markets, including in North America, Europe and Asia. We believe we have patent exclusivity relating to drug candidates derived from lipocalin proteins that runs until at least 2018 in the United States. We also rely on trade secrets for confidential know-how, which we generally seek to protect through contractual (e.g. confidentiality) agreements with employees and third parties.
We have protected the goodwill of our Company and our drug candidates, created through innovation and development, by putting in place trademark registrations of the Pieris and Anticalin marks as well as several defensive registrations.
We currently, and expect that we will continue to, file patent applications and maintain granted patents directed to our key drug candidates in an effort to establish intellectual property positions relating to new compositions of matter for these drug candidates, as well as novel medical applications of these compounds in the treatment, prevention or diagnosis of various indications. We also intend to seek patent protection, if available, with respect to biomarkers that may contribute to selecting the right patient population for use of any of our drug candidates, or with respect to pharmaceutical formulations that may be useful to produce final medicinal products.
We own or are the exclusive licensee of a patent portfolio consisting of several issued U.S. patents, and their respective counterparts in a number of foreign jurisdictions, several pending applications under the Patent Cooperation Treaty, multiple pending U.S. patent applications and corresponding pending patent applications in a number of foreign jurisdictions as well as three pending provisional patent applications, as described in further detail below.
In applicable jurisdictions, we will seek patent term extensions for certain patents of ours, including the patent term adjustment period in the United States. If we obtain marketing approval for our drug candidates in the United States or in certain jurisdictions outside of the United States, we may be eligible for regulatory protection, such as twelve years of data exclusivity for new biological entities in the United States and as mentioned below, up to five years of patent term extension potentially available in the United States, eight to eleven years of data and marketing exclusivity potentially available for new drugs in the European Union, up to five years of patent extension in Europe (supplemental protection certificate), and eight years of data exclusivity potentially available in Japan. There can be no assurance that we will qualify for any such regulatory exclusivity, or that any such exclusivity will prevent competitors from seeking approval solely on the basis of their own studies. See “—Government Regulation.”
Certain patents we own are directed to the basic Anticalin protein concept and platform technology (i.e. antagonist or agonist compounds derived from a natural lipocalin protein) and are expected to expire in 2018, subject to any patent term adjustments and terminal disclaimers in the United States. In addition, we hold issued patents and patent applications in the United States and other foreign jurisdictions, which patents are related to libraries of different scaffolds and consensus sequences such as human apolipoprotein D, human neutrophil gelatinase-associated lipocalin, or hNGAL, and human tear lipocalin, and are expected to expire between 2020 and 2030, subject to any patent term adjustments and terminal disclaimers in the United States. We also own a number of patents and patent applications at various stages of prosecution directed towards compositions of mater and in some cases, formulations or methods of use, of our preclinical and clinical drug candidates. Where possible, we will pursue patent term adjustments in the United States and any applicable foreign jurisdictions.
As a result of research efforts to date under the Research and License Agreement with Technische Universität München, or TUM, we hold a worldwide exclusive license to multiple patents and patent applications. These patents and patent applications relate to Anticalin proteins specific for the T cell co-receptor CD4and hNGAL lipocalin muteins and/or a library of an hNGAL scaffold of a certain consensus sequence, which patent is expected to expire in 2029, subject to any patent term adjustments or terminal disclaimers in the United States. We also hold an exclusive license to patents or patent applications related to bacterial lipocalin muteins.
As of the date of this report, a significant portion of our pending U.S. patent applications and pending patent applications in foreign jurisdictions were directed to newly-discovered or improved scaffold libraries of lipocalin muteins, compounds derived therefrom (i.e., specific drug candidates), or the uses of such compounds to treat, prevent and mitigate certain diseases and conditions whose pathological development involve the targets of interest as well as to diagnose, prognose and select treatments for the diseases and conditions. We would expect that any patents that may issue from the pending U.S. patent applications would likely expire between 2029 and 2038 without taking into account possible patent term adjustments or other extensions, however, any and all of these patent applications may not result in issued patents, and not all issued patents may be maintained in force for their entire term. We are actively pursuing intellectual property protection for our immuno-oncology drug candidates in key global markets that, if granted, could expire as late as 2038 or later depending on the date of the filing of such patent applications.
In addition to patents, we hold trademarks in the United States for the Anticalin and Pieris marks. Similarly, we hold their respective counterparts, either as registered trademarks or as pending applications, in a number of foreign jurisdictions. We expect that we will continue to look for trademark protection for the goodwill associated with our Company and our drug candidates in the countries or regions where we will have investment, research and development, sales or other activities.
We also rely upon unpatented trade secrets and know-how and continuing technological innovation to develop and maintain our competitive advantage. We strive to protect our proprietary information, in part, by using confidentiality agreements and/or invention assignment agreements with our collaborators, scientific advisors, employees and consultants. The confidentiality agreements are designed to protect our proprietary information and, in the case of agreements requiring invention assignment, to grant us ownership of technologies that are developed through a relationship with a third party. We also actively manage our publication and patent applications in that we only disclose information necessary to stir scientific interest or demonstrate patentability without materially compromising the secrecy of our valuable trade secrets and know-how. While we consider trade secrets and know-how to be a critical component of our intellectual property, trade secrets and know-how can be difficult to protect. In particular, with respect to our technology platform, we anticipate that these trade secrets and know-how will, over the course of time, be disseminated within the industry through independent development, the publication of journal articles describing the methodology and the movement of personnel skilled in the technology from academic to industry positions and
vice versa. As a result, those proprietary trade secrets and know-how may lose their value to us over a period of time, and we may lose any competitive advantage afforded by them, as they become public knowledge.
Strategic Partnerships and Other License Agreements
Since 2007, we have entered into several strategic partnerships and other license or option agreements to complement our drug discovery and development. Specifically, we have entered into strategic partnerships with Servier, AstraZeneca and Seattle Genetics (collectively, the "Strategic Partnerships") and other, non-strategic license or option agreements with Aska, Roche, Sanofi, Allergan Sales, LLC, Allergan, Inc., and Allergan Pharmaceuticals Holdings (Ireland) Unlimited Company, or (collectively) Allergan, Cadila Healthcare Limited, or Zydus, and Strides Arcolab Limited, or Stelis (collectively, the "Other License Agreements"), Under the Strategic Partnerships and License Agreements, we have developed and conducted or will develop and conduct selection and screening of drug candidates, as well as in vitro potency and efficacy testing, using our Anticalin®-brand drug discovery platform, our Anticalin libraries, and other proprietary methods to generate, identify, and characterize drug candidates against certain biological targets associated with several diseases. The Strategic Partnerships and License Agreements have provided us with approximately €60.1 million ($67.4 million) in revenue to date. With respect to discontinued agreements, we have no ongoing performance obligations, and do not expect to receive any significant additional consideration pursuant to those agreements.
Our Strategic Partnerships and License Agreements are ongoing and, under which, our partners are obligated to use commercially reasonable efforts to develop and commercialize drug candidates identified in the course of the collaboration. We are entitled to receive from our partners’ research, development and regulatory milestone payments and, in some cases, including the Servier and AstraZeneca collaborations, royalties on net sales for products developed and commercialized under these collaborations. With respect to our Strategic Partnerships, we have commercial rights, including the option to co-develop or co-commercialize one or more therapeutic programs with the applicable partners. We plan to continue to actively seek out additional collaboration partners that fit within our corporate development strategy.
The Strategic Partnerships represent the Company's cornerstone collaborations in its key therapeutic areas of immuno-oncology and respiratory diseases and include co-development and co-commercialization options. Certain terms and conditions of these Strategic Partnerships are summarized below:
Our collaboration with Servier
On January 4, 2017, we, along with Pieris GmbH, entered into a License and Collaboration Agreement and a Non-Exclusive Platform License Agreement with Servier. Pursuant to the terms of the agreements, we, along with Servier, will initially pursue five bispecific therapeutic programs, including our PRS-332 program. We will jointly develop PRS-332 and split commercial rights geographically, with Pieris retaining all commercial rights in the U.S. and Servier having commercial rights in the rest of the world. The four additional committed programs, which have been defined, may combine antibodies from the Servier portfolio with one or more Anticalin proteins based on our proprietary platform to generate innovative IO bispecific drug candidates. The collaboration may be expanded by up to three additional therapeutic programs. We also have the option to co-develop and retain commercial rights in the U.S. for up to three programs beyond PRS-332, while Servier will be responsible for development and commercialization of the other programs worldwide.
Under the agreements, we received an upfront payment of €30 million (approximately $32.0 million). We may also receive funding for full time staff for specific projects, as well as development-dependent and commercial milestone payments for PRS-332 and each additional program. The total development, regulatory and sales-based milestone payments to us could exceed €1.7 billion (approximately $1.8 billion) over the life of the collaboration and are dependent on the final number of projects pursued and the number of co-development options exercised by us. We will share preclinical and clinical development costs for each co-developed program with Servier. In addition, we will be entitled to receive tiered royalties up to low double digits on the sales of commercialized products in the Servier territories.
The term of each agreement ends upon the expiration of all of Servier’s payment obligations under such agreement. The agreements may be terminated by Servier for convenience beginning 12 months after their effective date upon 180 days’ notice. The agreements may also be terminated by either of us for material breach upon 90 days’ or 120 days’ notice of a material breach, with respect to the collaboration agreement and license agreement, respectively, provided that the applicable party has not cured such breach by the applicable 90-day or 120-day permitted cure period, and dispute resolution procedures specified in the applicable Agreement have been followed. The agreements may also be terminated due to the other party’s insolvency or for a safety issue and may in certain instances be terminated on a product-by-product and/or country-by-country basis. The license agreement will terminate upon termination of the collaboration agreement, on a product-by-product and/or country-by-country basis.
Our Collaboration with AstraZeneca
On May 2, 2017, we entered into a License and Collaboration Agreement, and a Non-Exclusive Anticalin Platform Technology License Agreement with AstraZeneca (the "AstraZeneca Agreements"), which became effective on June 10, 2017, following expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976. Under the AstraZeneca Agreements the parties will advance several novel inhaled Anticalin proteins.
Under the AstraZeneca Agreements, we received an upfront, non-refundable payment of $45.0 million. In addition, we have initiated a Phase I trial for the AstraZeneca Lead Product PRS-060 (AZD1402) for which we will receive a $12.5 million upfront payment. We are also eligible to receive research, development, commercial, sales milestone payments, and royalty payments. The total potential milestones are categorized as follows: research, development, and commercial milestones - up to $1.1 billion; and sales milestones - up to $1.0 billion. We may receive tiered royalties on sales of potential products commercialized by AstraZeneca and for co-developed products, gross margin share on worldwide sales equal dependent on our level of committed investment.
The term of the AstraZeneca Agreements ends upon the expiration of all of AstraZeneca’s payment obligations under such AstraZeneca Agreements. The AstraZeneca Collaboration Agreement may be terminated by AstraZeneca in its entirety for convenience beginning 12 months after its effective date upon 90 days’ notice or, if we have obtained marketing approval for the marketing and sale of a product, a 180 days’ notice. Each program may be terminated at AstraZeneca’s option; if any program is terminated by AstraZeneca, we will have full rights to such program. The AstraZeneca Collaboration Agreement may also be terminated by AstraZeneca or us for material breach upon 180 days’ notice of a material breach (or 30 days with respect to payment breach), provided that the applicable party has not cured such breach by the permitted cure period (including an additional 180 days if the breach is not susceptible to cure during the initial 180-day period) and dispute resolution procedures specified in the applicable AstraZeneca Agreement have been followed. The AstraZeneca Collaboration Agreement may also be terminated due to the other party’s insolvency and may in certain instances be terminated on a product-by-product and/or country-by-country basis. Each party may also terminate the agreement if the other party challenges the validity of patents related to certain intellectual property licensed under the AstraZeneca Agreement, subject to certain exceptions for infringement suits, acquisitions and newly-acquired licenses. The License Agreement will terminate upon termination of the AstraZeneca Collaboration Agreement, on a product-by-product and/or country-by-country basis.
Our Collaboration with Seattle Genetics
On February 8, 2018, we entered into a License and Collaboration Agreement and a Non-Exclusive Anticalin® Platform Technology License Agreement (collectively the “Seattle Genetics Agreements”) with Seattle Genetics, pursuant to which the parties will develop multiple targeted bispecific immuno-oncology treatments for solid tumors and blood cancers.
Under the terms of the Seattle Genetics Agreements, Seattle Genetics paid us a $30 million upfront fee, tiered royalties on net sales up to the low double-digits, and will pay us up to $1.2 billion in total success-based payments across three product candidates. The companies will pursue multiple antibody-Anticalin fusion proteins during a research phase, and Seattle Genetics has the option to select up to three therapeutic programs for further development. Prior to the initiation of a pivotal trial, we may opt into global co-development and U.S. commercialization of the second program and share in global costs and profits on a 50/50 basis. Seattle Genetics will solely develop, fund and commercialize the other two programs. Seattle Genetics may also decide to select additional candidates from the initial research phase for further development in return for the payment to us of additional fees, milestone payments, and royalties.
The term of each of the Seattle Genetics Agreements ends upon the expiration of all of Seattle Genetics’ payment obligations under such Agreement. The License and Collaboration Agreement may be terminated by Seattle Genetics in its entirety for convenience beginning 12 months after its effective date upon 90 days’ notice or, for any program where a pivotal study has been initiated, upon 180 days’ notice. Any program may be terminated at Seattle Genetics’ option. If any program is terminated by Seattle Genetics after a pre-defined pre-clinical stage, we will have full rights to continue such program. If any program is terminated by Seattle Genetics prior to such pre-defined pre-clinical stage, we will have the right to continue to develop such program, but will be obligated to offer a co-development option to Seattle Genetics for such program. The License and Collaboration Agreement may also be terminated by Seattle Genetics or us for an uncured material breach by the other party upon 90 days’ notice, subject to extension for an additional 90 days if the material breach relates to diligence obligations and subject, in all cases, to dispute resolution procedures. The License and Collaboration Agreement may also be terminated due to the other party’s insolvency and may in certain instances, including for reasons of safety, be terminated on a product-by-product basis. Each party may also terminate the Agreements if the other party challenges the validity of any patents licensed under the Agreements, subject to certain exceptions. The Non-Exclusive Anticalin® Platform Technology License Agreement will
terminate upon termination of the License and Collaboration Agreement, whether in its entirety or on a product-by-product basis.
Our License Agreements are older than our Strategic Partnerships and relate to non-strategic therapeutic areas, or do not provide us with co-development and co-commercialization rights. A brief summary of certain terms of selected License Agreements are provided below:
Our option agreement with Aska
On February 27, 2017, we entered into an Exclusive Option Agreement with Aska, granting Aska an exclusive option to license development and commercial rights to Pieris’ anemia drug, PRS-080, in Japan, South Korea and certain other Asian markets following completion of a multi-dose Phase IIa study to be conducted by Pieris in dialysis-dependent anemia patients.
Under the terms of the option agreement, we received an option payment of $2.75 million from Aska. Following an analysis period after the completion of the ongoing Phase IIa study conducted by us, Aska may exercise its option to obtain an exclusive license to develop and commercialize PRS-080 in Japan, South Korea and certain other Asian markets (excluding China). Should Aska exercise the option, we would be eligible for more than $80 million in combined option exercise fee and milestones associated with development and commercialization of PRS-080 in the first indication in Japan. We may receive further development milestones in additional indications, as well as in other countries within the Aska territory. We may also receive double-digit royalties on net sales of PRS-080 up to the mid- to high-teens.
The term of the Exclusive Option Agreement, including the option rights granted therein, ends on the earlier of (i) Aska’s written notice to us of Aska’s decision not to exercise the option rights granted under the Exclusive Option Agreement, (ii) Aska’s failure to exercise its option rights within sixty (60) days after the final results of the Phase IIa study are made available to Aska, (iii) three (3) months from date on which we deliver to Aska the final results of the Phase IIa study in the European Union, or (iv) our and Aska’s execution of the definitive agreements granting Aska licenses to develop and commercialize PRS-080 in Japan, South Korea and certain other Asian countries (excluding China) as contemplated under the Exclusive Option Agreement.
Our collaboration with Roche
In December 2015, we entered into a Research Collaboration and License Agreement with Roche, in cancer immune therapy for the research, development, and commercialization of Anticalin-based drug candidates against a predefined, undisclosed target selected by Roche. The parties will jointly pursue a preclinical research program with respect to the identification and generation of Anticalin proteins and during an extendable research term, Roche will fund the work to be performed by us pursuant to the research plan. Following the research program, Roche will be responsible for subsequent pre-clinical and clinical development of any product and will have worldwide commercialization rights.
On February 28, 2018, pursuant to Pieris' license agreement with Roche, as amended, Roche provided notice to Pieris of its extension of the research term of the Research Collaboration and License Agreement to continue until August 31, 2018.
Our collaboration with Sanofi
In September 2010, we entered into a collaboration and license agreement with Sanofi, which was subsequently amended in February 2013. Under the terms of the agreement, we have agreed to use our proprietary Anticalin technologies to identify drug candidates against certain targets, with further development and commercialization activities conducted by Sanofi. The collaboration started with two targets under two separate collaboration projects and was extended by an additional multispecific Anticalin program in 2013. Sanofi has terminated two of the three programs (one program was terminated for internal strategic reasons and the other program was terminated following in vivo studies, as in vitro functionality did not fully translate into in vivo functionality for this first in class program), and we have the right to develop and commercialize drug candidates of the terminated programs on a royalty-free basis. The remaining active collaboration project was handed over to Sanofi for further development in the fourth quarter of 2014.
Our agreement with Allergan
In August 2009, we entered into an agreement with Allergan, under which we provided drug candidates for the treatment of ocular diseases, and Allergan is responsible for the further development and commercialization of products based on those candidates and bearing related costs.
Our collaboration with Daiichi Sankyo
Daiichi Sankyo Company Limited (“Daiichi Sankyo” or "Daiichi") partnered with Pieris to research, develop and commercialize two Anticalin therapeutics pursuant to a May 31, 2011 Collaboration Research and Technology Licensing Agreement (the “DS Collaboration Agreement”).
On May 8, 2017, Daiichi Sankyo discontinued the development of one of those two therapeutics, a PSCK9 program (DS-9001), after having advanced this program to first-in-human studies for strategic reasons. Consequentially, this Anticalin program, including respective intellectual property rights, were transferred back to Pieris.
The second of these two therapeutic programs pursued under the DS Collaboration Agreement is an Anticalin protein directed to calcitonin gene-related peptide (“CGRP”) for therapeutic use in the treatment of migraine headaches. The CGRP program is in the IND-enabling stage and Pieris previously reported Daiichi Sankyo’s decision to initiate a GLP toxicity study in connection with this program.
Due to Daiichi Sankyo’s strategic prioritization and commercial reasons related to competing CGRP inhibitors in advanced stages of development, Daiichi Sankyo provided notice to Pieris on March 1, 2018 of its termination of the CGRP program. Pre-clinical data regarding this CGRP antagonizing Anticalin protein indicate that it has good drug-like properties, including strong target affinity, the ability to neutralize the biological activity of CGRP in vitro, and the ability to inhibit vasodilation, a cause of migraine pain, in rat skin following subcutaneous administration of the antagonist. In connection with the termination of the development of the CGRP program, and under the terms of the DS Collaboration Agreement, Daiichi Sankyo is obligated to carry out certain activities to facilitate transfer of activities, regulatory filings, materials, data, agreements and other matters to Pieris in connection with the return to Pieris of the development program related to the CGRP program. Pieris intends to diligently review the data associated with the program and consider its strategic options thereafter.
The foregoing summary of the terms relating to the DS Collaboration Agreement, including with respect to Daiichi Sankyo’s termination of such Agreement with respect to the CGRP program, is qualified in its entirety by the terms of the DS Collaboration Agreement, which were filed with the SEC as exhibit 10.7 to the Form 10-K filed on March 30, 2017. The contents of such exhibit are hereby incorporated by reference.
TUM License Agreement
On July 4, 2003, we entered into a Research and Licensing agreement with TUM, which was subsequently renewed and amended, on July 26, 2007. The agreement established a joint research effort led by Prof. Arne Skerra, Chair of Biological Chemistry of TUM, to optimize Anticalin technologies for use in therapeutic, prophylactic and diagnostic applications and as research reagents, and to gain fundamental insights in lipocalin scaffolds. We provided certain funding for TUM research efforts performed under the agreement. The research phase of this collaboration ended on February 28, 2013.
Under the terms of the agreement, TUM assigned to us certain materials and records resulting from the research. We retained rights to inventions made by our employees, and TUM assigned to us all inventions made under the agreement jointly by our employees and TUM personnel, provided that our employees made certain inventive contributions. With respect to all other inventions made in the course of the research, TUM granted to us worldwide exclusive license rights under patents and patent applications claiming such inventions. TUM retained rights to practice these inventions for research and teaching purposes.
As a result of research efforts to date under the agreement, we hold a worldwide exclusive license under our license agreement with TUM to multiple patents and patent applications. In the United States, we hold an exclusive license to an issued U.S. patent No. 8,598,317 for the composition of matter of mutein of human tear lipocalin binding to the extracellular region of the T cell co-receptor CD4 with detectable affinity, which patent will expire in 2027 (subject to a patent term adjustment period which is expected to be at least 742 days), as well as to its counterpart in the European Union. We also hold an exclusive license to an issued U.S. patent No. 8,420,051 directed to library of hNGAL scaffold of certain consensus sequence, which patent is expected to expire in 2029 (subject to a patent term adjustment period of 109 days), as well as to its counterparts in the European Union and in a number of foreign jurisdictions. Moreover, we hold an exclusive license to an issued U.S. patent No. 8,987,415 claiming isolated crystalline form of monomeric bacterial lipocalin. We bear the costs of filing, prosecution and maintenance of patents assigned or licensed to us under the agreement.
As consideration for the assignments and licenses, we are obliged to pay to TUM license payments on development of our proprietary products claimed by patents assigned or licensed to us by TUM. For each of such proprietary products developed by us, we could be required to pay up to an aggregate of approximately €0.2 million ($0.2 million) in license payments to TUM under the agreement.
We also are obliged to pay low single digit royalties, including annual minimum royalties, on sales of such products. Should we grant licenses or sublicenses to those patents to third parties, we are obliged to share a percentage of resulting revenue with TUM, which percentage of resulting revenue is creditable against our annual license payments to TUM. Our payment obligations are reduced by our proportionate contribution to a joint invention. Payment obligations terminate on expiration or annulment of the last patent covered by the agreement.
We can terminate the licenses to any or all licensed patents upon specified advance notice to TUM. TUM may terminate the license provisions of the agreement only for cause. Termination of the agreement does not terminate our rights in patents assigned to us.
Enumeral License Agreement
On April 18, 2016, we entered into a License Agreement with Enumeral Biomedical Holdings, Inc., or Enumeral, pursuant to which we acquired a non-exclusive (except in the exclusive field described below) worldwide license to use specified patent rights and know-how owned by Enumeral to research, develop and market fusion proteins consisting of PD-1 antibodies linked to one or more Anticalin proteins for use in the oncology area. Enumeral also agreed not to practice or assist third parties in practicing in the exclusive field, consisting of licensed antibodies fused to Anticalin proteins in the oncology area.
On June 6, 2016, we entered into a Definitive License Agreement to fully set forth the terms of Enumeral’s license of PD-1 antibodies and grant of options to license additional antibodies to us. Under the Definitive License and Transfer Agreement, we in-licensed intellectual property related to an Enumeral-generated antibody against PD-1 and an option to in-license up to two additional antibodies against undisclosed targets. Under the terms of the agreement, we acquired a non-exclusive worldwide license under the applicable Enumeral patents and know-how to research, develop and commercialize fusion proteins incorporating Enumeral’s PD-1 antibody and one or more Anticalin proteins, or the Subsequent Options, which was an expansion of the scope of the original License Agreement.
The Subsequent Options expired on May 31, 2017. We are also obliged to pay to Enumeral development and sales milestones on development of products incorporating the Enumeral antibody, as well as low to lower-middle single-digit royalties as a percentage of net sales depending on the amount of net sales in the applicable years. In the event that we are required to pay a license fee or royalty to any third party related to the licensed products, our royalty payment obligations to Enumeral are reduced by the amount of such third-party fees or payments, up to 50% of the royalty payment for each calendar year due to Enumeral. Payment obligations terminate on a product-by-product and country-by-country basis on the later of ten years from the first commercial sale of a product incorporating the Enumeral antibody or the last to expire, lapse or be abandoned of a claim from the licensed Enumeral patents filed as of the effective date of the agreement that cover the manufacture, use, offer for sale, sale or import of a product incorporating the Enumeral antibody.
The term of the Definitive License Agreement ends upon the expiration of the last to expire patent covered under the license. The Agreement may be terminated by us on 30 days’ notice and by Enumeral upon 60 days’ notice of a material breach by us (or 30 days with respect to a breach of payment obligations by us), provided that we have not cured such breach and dispute resolution procedures specified in the Definitive License Agreement have been followed.
On January 29, 2018, Enumeral filed a voluntary petition for relief under Chapter 11 of the United States Bankruptcy Code in the Bankruptcy Court for the District of Massachusetts (the “Bankruptcy Court”). Enumeral continues to operate its business as a debtor in possession under the jurisdiction of the Bankruptcy Court.
The Bankruptcy Court has approved a process pursuant to which Enumeral will solicit higher or better offers for the PD-1 Assets and conduct an auction process to determine the ultimate purchaser. The process contemplates that the winning bidder will purchase the PD-1 Assets, including the Definitive License Agreement, with the sale contemplated to close in early April 2018. We will have an opportunity to vet the proposed purchaser to ensure that they (i) can continue to perform Enumeral’s obligations under the Definitive License Agreement, and (ii) will not interfere with our use of the rights we currently enjoy under the Definitive License Agreement.
Kelun License Agreement
In connection with our efforts to develop multispecific Anticalin-based proteins designed to engage immunomodulatory targets, during the second quarter of 2017, the Company entered into a License and Transfer Agreement with Sichuan Kelun-Biotech Biopharmaceutical Co. Ltd. (“Kelun”). Under that Agreement, Kelun has granted to the Company a non-exclusive worldwide license (with the right to sublicense) under certain intellectual property owned or controlled by Kelun to research, develop,
manufacture, and commercialize bi- and multi- specific fusion proteins that include a monoclonal antibody developed by Kelun specific for an undisclosed target and one or more Anticalin proteins. We believe that the drug-like properties of the Anticalin drug class were demonstrated in various clinical trials with different Anticalin-based drug candidates, including PRS-080, PRS-060 and others.
Government Regulation
United States – FDA Process
The research, development, testing, manufacture, quality control, approval, labeling, packaging, storage, record-keeping, promotion, advertising, distribution, marketing, among other things, of drug products are extensively regulated by governmental authorities in the United States and other countries.
U.S. Drug Development Process
In the U.S., the Food and Drug Administration, or FDA, regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and in the case of biologics, also under the Public Health Service Act, or PHSA, and implementing regulations. Failure to comply with the applicable U.S. requirements may subject us to administrative or judicial sanctions, such as FDA refusal to approve pending NDAs, or their issuance of warning
letters, or the imposition of fines, civil penalties, product recalls, product seizures, total or partial suspension of production or distribution, injunctions and/or criminal prosecution. The process required by the FDA before a drug or biologic may be marketed in the U.S. generally involves the following:
| |
• | completion of preclinical laboratory tests, animal studies and formulation studies according to Good Laboratory Practices or other applicable regulations; |
| |
• | submission to the FDA of an Investigational New Drug application, or IND which must become effective before human clinical trials may begin; |
| |
• | performance of adequate and well-controlled human clinical trials according to Good Clinical Practices to establish the safety and efficacy of the proposed drug for its intended use; |
| |
• | submission to the FDA of an New Drug Application, NDA or Biologic License Application, or BLA; |
| |
• | satisfactory completion of an FDA inspection of the manufacturing facility or facilities at which the drug is produced to assess compliance with current good manufacturing practice, or cGMP, to assure that the facilities, methods and controls are adequate to preserve the drug’s identity, strength, quality and purity; and |
| |
• | FDA review and approval of the NDA or BLA. |
Once a pharmaceutical candidate is identified for development, it enters the preclinical testing stage. Preclinical tests include laboratory evaluations of product chemistry, toxicity and formulation, as well as animal studies. An IND sponsor must submit the results of the preclinical tests, together with manufacturing information and analytical data, to the FDA as part of the IND, which must become effective before human clinical trials may begin. An IND automatically becomes effective 30 days after receipt by the FDA, unless the FDA, within the 30-day time period, places the clinical trial on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. Clinical holds also may be imposed by the FDA at any time before or during studies due to safety concerns or non-compliance.
All clinical trials must be conducted under the supervision of qualified investigators. Clinical trials are conducted under protocols detailing the objectives of the study, the parameters to be used in monitoring the safety and effectiveness criteria to be evaluated. Each protocol must be submitted to the FDA as part of the IND. Further, an Institutional Review Board, or IRB, for each medical center proposing to conduct the clinical trial must review and approve the study protocol and informed consent information for study subjects for any clinical trial before it commences at that center, and the IRB must monitor the study until it is completed. Study subjects must sign an informed consent form before participating in a clinical trial. There are also requirements governing the reporting of on-going clinical trials and clinical trial results to public registries.
Human clinical trials are typically conducted in three sequential phases that may overlap or be combined:
| |
• | Phase I: The product candidate is initially introduced into healthy human subjects and tested for safety, dosage tolerance, absorption, metabolism, distribution and excretion. In the case of some products for severe or life- |
threatening diseases, such as cancer, especially when the product may be too inherently toxic to ethically administer to healthy volunteers, the initial human testing is often conducted in patients.
| |
• | Phase II: This phase involves studies in a limited patient population to identify possible adverse effects and safety risks, to preliminarily evaluate the efficacy of the product for specific targeted diseases and to determine dosage tolerance and optimal dosage. |
| |
• | Phase 3: Clinical trials are undertaken to further evaluate dosage, clinical efficacy and safety in an expanded patient population at geographically dispersed clinical study sites. These studies are intended to establish the overall risk-benefit ratio of the product candidate and provide, if appropriate, an adequate basis for product labeling. |
The FDA or an IRB may suspend clinical trials at any time on various grounds, including a finding that the research subjects or patients are being exposed to an unacceptable health risk. The FDA may approve an NDA for a product candidate, but require that the sponsor conduct additional clinical trials to further assess the drug after NDA approval under a post-approval commitment. Post-approval trials are typically referred to as Phase 4 clinical trials. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients.
During the development of a new drug, sponsors are given opportunities to meet with the FDA at certain points. Including prior to submission of an IND, at the end of Phase II, and before an NDA or BLA is submitted. These meetings can provide an opportunity for the sponsor to share information about the data gathered to date, for the FDA to provide advice, and for the sponsor and the FDA to reach agreement on the next phase of development. Sponsors typically use the end of Phase II meeting to discuss their Phase II clinical results and present their plans for the pivotal phase 3 clinical trial that they believe will support approval of the new drug.
Concurrent with clinical trials, companies usually complete additional animal studies and must also develop additional information about the chemistry and physical characteristics of the drug and finalize a process for manufacturing the product in commercial quantities in accordance with cGMP requirements. The manufacturing process must be capable of consistently producing quality batches of the product candidate and, among other things, the manufacturer must develop methods for testing the identity, strength, quality and purity of the final drug. Additionally, appropriate packaging must be selected and tested, and stability studies must be conducted to demonstrate that the product candidate does not undergo unacceptable deterioration over its shelf life.
U.S. Review and Approval Processes
The results of product development, preclinical studies and clinical trials, along with descriptions of the manufacturing process, analytical tests conducted on the chemistry of the drug, proposed labeling, and other relevant information are submitted to the FDA as part of an NDA or BLA requesting approval to market the product. The submission of an NDA or BLA is subject to the payment of user fees; a waiver of such fees may be obtained under certain limited circumstances. The FDA reviews all NDAs and BLAs submitted to ensure that they are sufficiently complete for substantive review before it accepts them for filing. The FDA may request additional information rather than accept an NDA or BLA for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA may refer the NDA or BLA to an advisory committee for review, evaluation and recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. The approval process is lengthy and often difficult, and the FDA may refuse to approve an NDA or BLA if the applicable regulatory criteria are not satisfied or may require additional clinical or other data and information. The FDA may issue a complete response letter, which may require additional clinical or other data or impose other conditions that must be met in order to secure final approval of the NDA or BLA, or an approved letter following satisfactory completion of all aspects of the review process. The FDA reviews an NDA to determine, among other things, whether a product is safe and effective for its intended use and whether its manufacturing is cGMP-compliant to assure and preserve the product’s identity, strength, quality and purity. The FDA reviews a BLA to determine, among other things whether the product is safe, pure and potent and the facility in which it is manufactured, processed, packed or held meets standards designed to assure the product’s continued safety, purity and potency.
NDAs or BLAs receive either standard or priority review. A drug representing a significant improvement in treatment, prevention or diagnosis of disease may receive priority review. Priority review for an NDA for a new molecular entity and original BLAs will be six months from the date that the NDA or BLA is filed. In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful therapeutic benefit over existing treatments may receive accelerated approval and may be approved on the basis of adequate and well-controlled clinical trials establishing that the drug product has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit or on
the basis of an effect on a clinical endpoint other than survival or irreversible morbidity. As a condition of approval, the FDA may require that a sponsor of a drug receiving accelerated approval perform adequate and well-controlled post-marketing clinical trials. Priority review and accelerated approval do not change the standards for approval, but may expedite the approval process.
If a product receives regulatory approval, the approval may be significantly limited to specific diseases and dosages or the indications for use may otherwise be limited, which could restrict the commercial value of the product. In addition, the FDA may require a sponsor to conduct Phase IV testing which involves clinical trials designed to further assess a drug’s safety and effectiveness after NDA or BLA approval, and may require testing and surveillance programs to monitor the safety of approved products which have been commercialized.
The Food and Drug Administration Safety and Innovation Act, or FDASIA, enacted in 2012, made permanent the Pediatric Research Equity Act, or PREA, which requires a sponsor to conduct pediatric studies for most drugs and biologicals, for a new active ingredient, new indication, new dosage form, new dosing regimen or new route of administration. Under PREA, original NDAs, BLAs and supplements thereto, must contain a pediatric assessment unless the sponsor has received a deferral or waiver. The required assessment must assess the safety and effectiveness of the product for the claimed indications in all relevant pediatric subpopulations and support dosing and administration for each pediatric subpopulation for which the product is safe and effective.
Patent Term Restoration and Marketing Exclusivity
Depending upon the timing, duration and specifics of FDA approval of our drugs, some of our U.S. patents may be eligible for limited patent term extension. These patent term extensions permit a patent restoration term of up to five years as compensation for any patent term lost during product development and the FDA regulatory review process. However, patent term restoration cannot extend the remaining term of a patent beyond a total of fourteen years from the product’s approval date. The patent term restoration period is generally one-half the time between the effective date of an IND, and the submission date of an NDA or BLA, plus the time between the submission date of an NDA or BLA and the approval of that application. Only one patent applicable to an approved drug is eligible for the extension, and the extension must be applied for prior to expiration of the patent. The United States Patent and Trademark Office, in consultation with the FDA, reviews and approves the application for any patent term extension or restoration.
Pediatric exclusivity is another type of marketing exclusivity available in the U.S. The FDASIA made permanent the Best Pharmaceuticals for Children Act, or BPCA, which provides for an additional six months of marketing exclusivity if a sponsor conducts clinical trials in children in response to a written request from the FDA, or a Written Request. If a Written Request does not include studies in neonates, the FDA is required to include its rationale for not requesting those studies. The FDA may request studies on approved or unapproved indications in separate Written Requests. The issuance of a Written Request does not require the sponsor to undertake the described studies.
Biologics Price Competition and Innovation Act of 2009
In March 2010, the Patient Protection and Affordable Care Act was enacted in the U.S. and included the Biologics Price Competition and Innovation Act of 2009, or BPCIA. The BPCIA amended the Public Health Service Act, or PHSA, to create an abbreviated approval pathway for two types of “generic” biologics—biosimilars and interchangeable biologic products, and provides for a twelve-year exclusivity period for the first approved biological product, or reference product, against which a biosimilar or interchangeable application is evaluated; however if pediatric studies are performed and accepted by the FDA, the twelve-year exclusivity period will be extended for an additional six months A biosimilar product is defined as one that is highly similar to a reference product notwithstanding minor differences in clinically inactive components and for which there are no clinically meaningful differences between the biological product and the reference product in terms of the safety, purity and potency of the product. An interchangeable product is a biosimilar product that may be substituted for the reference product without the intervention of the health care provider who prescribed the reference product.
The biosimilar applicant must demonstrate that the product is biosimilar based on data from (1) analytical studies showing that the biosimilar product is highly similar to the reference product; (2) animal studies (including toxicity); and (3) one or more clinical studies to demonstrate safety, purity and potency in one or more appropriate conditions of use for which the reference product is approved. In addition, the applicant must show that the biosimilar and reference products have the same mechanism of action for the conditions of use on the label, route of administration, dosage and strength, and the production facility must meet standards designed to assure product safety, purity and potency.
An application for a biosimilar product may not be submitted until four years after the date on which the reference product was first approved. The first approved interchangeable biologic product will be granted an exclusivity period of up to one year after it is first commercially marketed, but the exclusivity period may be shortened under certain circumstances.
Orphan Drug Designation
Under the Orphan Drug Act, the FDA may grant orphan drug designation to a drug intended to treat a rare disease or condition, which is generally a disease or condition that affects fewer than 200,000 individuals in the U.S., or more than 200,000 individuals in the U.S. and for which there is no reasonable expectation that the cost of developing and making available in the U.S. a drug for this type of disease or condition will be recovered from sales in the U.S. for that drug. Orphan drug designation must be requested before submitting an NDA or BLA. After the FDA grants orphan drug designation, the identity of the therapeutic agent and its potential orphan use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in or shorten the duration of the regulatory review and approval process. If a product that has orphan drug designation subsequently receives the first FDA approval for the disease for which it has such designation, the product is entitled to orphan product exclusivity, which means that the FDA may not approve any other applications to market the same drug for the same indication, except in very limited circumstances, for seven years. Orphan drug exclusivity, however, also could block the approval of one of our products for seven years if a competitor obtains approval of the same drug as defined by the FDA or if our product candidate is determined to be contained within the competitor’s product for the same indication or disease.
The FDA also administers a clinical research grants program, whereby researchers may compete for funding to conduct clinical trials to support the approval of drugs, biologics, medical devices, and medical foods for rare diseases and conditions. A product does not have to be designated as an orphan drug to be eligible for the grant program. An application for an orphan grant should propose one discrete clinical study to facilitate FDA approval of the product for a rare disease or condition. The study may address an unapproved new product or an unapproved new use for a product already on the market.
Fast Track Designation and Accelerated Approval
The FDA is required to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. Under the fast track program, the sponsor of a new drug candidate may request that the FDA designate the drug candidate for a specific indication as a fast track drug concurrent with, or after, the filing of the IND for the drug candidate. The FDA must determine if the drug candidate qualifies for fast track designation within 60 days of receipt of the sponsor’s request.
Under the fast track program and the FDA’s accelerated approval regulations, the FDA may approve a drug for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity, or prevalence of the condition and the availability or lack of alternative treatments. In clinical trials, a surrogate endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of how a patient feels, functions, or survives. A drug candidate approved on this basis is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post- approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug from the market on an expedited basis. All promotional materials for drug candidates approved under accelerated regulations are subject to prior review by the FDA.
In addition to other benefits such as the ability to use surrogate endpoints and engage in more frequent interactions with the FDA, the FDA may initiate review of sections of a fast track drug’s BLA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the fast track designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
Post-Approval Requirements
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product may result in restrictions on the product or even complete withdrawal of the product from the market. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes and additional labeling claims, are
subject to further FDA review and approval. Drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and certain state agencies, and are subject to periodic unannounced inspections by the FDA and certain state agencies for compliance with cGMP and other laws and regulations. We rely, and expect to continue to rely, on third parties for the production of clinical and commercial quantities of our products. Future inspections by the FDA and other regulatory agencies may identify compliance issues at the facilities of our contract manufacturers that may disrupt production or distribution, or require substantial resources to correct.
Any drug products manufactured or distributed by us pursuant to FDA approvals are subject to continuing regulation by the FDA, including, among other things, record-keeping requirements, reporting of adverse experiences with the drug, providing the FDA with updated safety and efficacy information, drug sampling and distribution requirements, complying with certain electronic records and signature requirements, and complying with FDA promotion and advertising requirements. The FDA strictly regulates labeling, advertising, promotion and other types of information on products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label.
From time to time, new legislation and regulations may be implemented that could significantly change the statutory provisions governing the approval, manufacturing and marketing of products regulated by the FDA. It is impossible to predict whether further legislative or regulatory changes will be enacted, or FDA regulations, guidance or interpretations changed or what the impact of such changes, if any, may be.
Foreign Regulation
In addition to regulations in the United States, we will be subject to a variety of foreign regulations governing clinical trials and commercial sales and distribution of our products. Whether or not we obtain FDA approval for a product, we must obtain approval by the comparable regulatory authorities of foreign countries or economic areas, such as the 28-member European Union, before we may commence clinical trials or market products in those countries or areas. The approval process and requirements governing the conduct of clinical trials, product licensing, pricing and reimbursement vary greatly from place to place, and the time may be longer or shorter than that required for FDA approval.
Under European Union regulatory systems, a company may submit marketing authorization applications either under a centralized or decentralized procedure. The centralized procedure, which is compulsory for medicinal products produced by biotechnology or those medicinal products containing new active substances for specific indications such as the treatment of AIDS, cancer, neurodegenerative disorders, diabetes, viral diseases and designated orphan medicines, and optional for other medicines which are highly innovative. Under the centralized procedure, a marketing application is submitted to the European Medicines Agency where it will be evaluated by the Committee for Medicinal Products for Human Use and a favorable opinion typically results in the grant by the European Commission of a single marketing authorization that is valid for all European Union member states. The initial marketing authorization is valid for five years, but once renewed is usually valid for an unlimited period.
When conducting clinical trials in the EU, we must adhere to the provisions of the EU Clinical Trials Directive and the laws and regulations of the EU Member States implementing them. These provisions require, among other things, that the prior authorization of an Ethics Committee and the submission and approval of a clinical trial authorization application be obtained in each Member State be obtained before commencing a clinical trial in that Member State.
As in the United States, it may be possible in foreign countries to obtain a period of market and/or data exclusivity that would have the effect of postponing the entry into the marketplace of a competitor’s generic product. As in the United States, a sponsor may apply for designation of a product as an orphan drug for the treatment of a specific indication in the EU before the application for marketing authorization is made. Orphan drugs in Europe enjoy economic and marketing benefits, including up to 10 years of market exclusivity for the approved indication unless another applicant can show that its product is safer, more effective or otherwise clinically superior to the orphan-designated product.
Reimbursement
Sales of pharmaceutical products depend in significant part on the availability of third-party reimbursement. Third-party payors include government healthcare programs, managed care providers, private health insurers and other organizations. These third-party payors are increasingly challenging the price and examining the cost-effectiveness of medical products and services. In addition, significant uncertainty exists as to the reimbursement status of newly approved healthcare products. We may need to conduct expensive pharmacoeconomic studies in order to demonstrate the cost-effectiveness of our products. Our product candidates may not be considered cost-effective. It is time consuming and expensive to seek reimbursement from third-party payors. Reimbursement may not be available or sufficient to allow us to sell our products on a competitive and profitable basis.
In addition, in some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, the European Union provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European Union do not follow price structures of the United States and generally tend to by significantly lower.
Description of the Acquisition
On December 17, 2014, Pieris Pharmaceuticals, Inc., Pieris GmbH and the former stockholders of Pieris GmbH entered into an Acquisition Agreement, or the Acquisition Agreement. Pursuant to the Acquisition Agreement, the stockholders of Pieris GmbH contributed all of their equity interests in Pieris GmbH to Pieris in exchange for shares of Pieris common stock, which resulted in Pieris GmbH becoming a wholly owned subsidiary of Pieris, which we refer to as the Acquisition. Prior to the Acquisition, as defined below, Pieris pursued a business of an errand concierge service online marketplace.
Prior to the closing of the Acquisition, on December 5, 2014, Pieris completed a 2.272727-for-1 forward split of its common stock in the form of a share dividend, with the result that 6,100,000 shares of common stock outstanding immediately prior to the stock split became 13,863,647 shares of common stock outstanding immediately thereafter. On December 16, 2014, Pieris amended and restated its Articles of Incorporation to, among other things, change its name from Marika Inc. to “Pieris Pharmaceuticals, Inc.,” and increase its authorized capital stock from 75,000,000 shares of common stock, par value $0.001 per share, to 300,000,000 shares of common stock, par value $0.001 per share, and 10,000,000 shares of “blank check” preferred stock, par value $0.001 per share.
On December 17, 2014, Pieris, Pieris GmbH and the former stockholders of Pieris GmbH entered into an Acquisition Agreement, or the Acquisition Agreement. Pursuant to the Acquisition Agreement, the stockholders of Pieris GmbH contributed all of their equity interests in Pieris GmbH to Pieris in exchange for shares of Pieris common stock, which resulted in Pieris GmbH becoming a wholly owned subsidiary of Pieris, which we refer to as the Acquisition. The Acquisition closed on December 17, 2014.
In connection with the Acquisition and pursuant to a Split-Off Agreement, dated December 17, 2014 among Pieris, Marika Enterprises Inc. and Aleksandrs Sviks, or the Split-Off Agreement, and a general release agreement, Pieris transferred its pre-Acquisition assets and liabilities to its former majority stockholder, Aleksandrs Sviks, in exchange for the surrender by him and cancellation of 11,363,635 shares of Pieris common stock, or the Split-Off. Upon the closing of the Acquisition and the Split-Off, Pieris discontinued its pre-Acquisition business plans and is now pursuing only the business of Pieris GmbH.
Upon the closing of the Acquisition, Pieris ceased to be a “shell company” under applicable rules of the SEC. On December 17, 2014, in connection with the Acquisition, our Board of Directors changed our fiscal year from a fiscal year ending on June 30 to one ending on December 31 of each year, which was the fiscal year of Pieris GmbH.
Emerging Growth Company and Smaller Reporting Company Status
The Jumpstart Our Business Startups Act of 2012, or the JOBS Act, establishes a class of company called an “emerging growth company,” which generally is a company whose initial public offering was completed after December 8, 2011 and had total annual gross revenues of less than $1 billion during its most recently completed fiscal year. Additionally, Section 12b-2 of the Securities Exchange Act of 1934, as amended, or the Exchange Act, establishes a class of company called a “smaller reporting company,” which generally is a company with a public float of less than $75 million as of the last business day of its most recently completed second fiscal quarter or, if such public float is $0, had annual revenues of less than $50 million during the most recently completed fiscal year for which audited financial statements are available. For the year ended December 31, 2017, we qualify as both an emerging growth company and a smaller reporting company.
As an emerging growth company and a smaller reporting company, we are eligible to take advantage of certain extended accounting standards and exemptions from various reporting requirements that are not available to public reporting companies that do not qualify for those classifications, including without limitation the following:
| |
• | An emerging growth company can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act of 1933, as amended, or the Securities Act, for complying with new or revised accounting standards. |
This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to avail ourselves of this extended transition period and, as a result, we will not be required to adopt new or revised accounting standards on the dates on which adoption of such standards is required for other public reporting companies.
| |
• | An emerging growth company is exempt from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and financial statements, commonly known as an “auditor discussion and analysis.” |
| |
• | An emerging growth company is not required to hold nonbinding advisory stockholder votes on executive compensation or any “golden parachute” payments not previously approved by stockholders. |
| |
• | Neither an emerging growth company nor a smaller reporting company is required to comply with the requirement of auditor attestation of internal controls over financial reporting, which is required for other public reporting companies by Section 404 of the Sarbanes-Oxley Act of 2002. |
| |
• | A company that is either an emerging growth company or a smaller reporting company is eligible for reduced disclosure obligations regarding executive compensation in its periodic and annual reports, including without limitation exemption from the requirement to provide a compensation discussion and analysis describing compensation practices and procedures. |
| |
• | A company that is either an emerging growth company or a smaller reporting company is eligible for reduced financial statement disclosure in its registration statements, which must include two years of audited financial statements rather than the three years of audited financial statements that are required for other public reporting companies. Smaller reporting companies are also eligible to provide such reduced financial statement disclosure in annual reports on Form 10-K. |
For as long as we continue to be an emerging growth company and/or a smaller reporting company, we expect that we will take advantage of the reduced disclosure obligations available to us as a result of those respective classifications. We will remain an emerging growth company until the earlier of (i) December 31, 2019, the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement under the Securities Act; (ii) the last day of the fiscal year in which we have total annual gross revenues of $1 billion or more; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years; or (iv) the date on which we are deemed to be a large accelerated filer under applicable SEC rules. We expect that we will remain an emerging growth company for the foreseeable future, but cannot retain our emerging growth company status indefinitely and will no longer qualify as an emerging growth company on or before December 31, 2019.
In accordance with Section 12b-2 of the Exchange Act, we are no longer a smaller reporting company as of December 31, 2017. However, under Item 10(f)(2)(i) of Regulation S-K, we are permitted to avail ourselves of the scaled disclosure requirements available to smaller reporting companies in this Annual Report on Form 10-K. Having ceased to be a smaller reporting company, we will no longer be able to avail ourselves of scaled disclosure beginning with our Quarterly Report on Form 10-Q for the quarterly period ending March 31, 2018.
Employees
As of December 31, 2017, we had 68 full-time employees. None of our employees is represented by a labor union or covered by a collective bargaining agreement. We consider our relationship with our employees to be good. In order to successfully develop our drug candidates, we must be able to attract and retain highly skilled personnel. We anticipate hiring additional employees for research and development, clinical and regulatory affairs and general and administrative activities over the next few years. We also utilize the services of consultants, clinical research organizations, and other third parties on a regular basis.
Available Information
Our internet address is www.pieris.com. Copies of our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, and all amendments to those reports, are available to you free of charge through the Investors section of our website as soon as reasonably practicable after such materials have been electronically filed with, or furnished to, the SEC. The information contained on, or that can be accessed through, our website is not part of this Annual Report on Form 10-K. We have included our website address in this Annual Report solely as an inactive textual reference.
Investing in our common stock involves a high degree of risk. You should carefully consider the risks described below, together with the other information contained in this prospectus, including our consolidated financial statements and the related notes, before making any decision to invest in shares of our common stock. This Annual Report on Form 10-K contains forward-looking statements. If any of the events discussed in the risk factors below occurs, our business, prospects, results of operations, financial condition and cash flows could be materially harmed. If that were to happen, the trading price of our common stock could decline, and you could lose all or part of your investment. The risks and uncertainties described below are not the only ones we face. Additional risks not currently known to us or other factors not perceived by us to present significant risks to our business at this time also may impair our business operations.
Risks Related to Our Business, Financial Position and Capital Requirements
We have incurred significant losses since our inception and anticipate that we will continue to incur losses for the foreseeable future. We currently have no product revenues and no approved products, and will need to raise additional capital to operate our business.
We are a clinical-stage biopharmaceutical company. To date, we have not generated any commercial sales revenue and are not profitable, and have incurred losses since our inception in August 2000. For the years ended December 31, 2017 and 2016 we reported net loss of $17.6 million and $22.8 million, respectively. As of December 31, 2017, we had an accumulated deficit of $120.3 million. We expect to continue to incur losses for the foreseeable future, and we expect these losses to increase as we continue our development of, and seek regulatory approvals for, our drug candidates and the commercialization of approved products, if any.
We are currently focused primarily on the development of our lead drug candidates and programs, our immuno-oncology programs including PRS-343, currently in Phase I trials, and our preclinical immuno-oncology programs and drug candidates, our respiratory drug candidate PRS-060, currently in Phase I trials in partnership with AstraZeneca and our hepcidin antagonist, PRS-080, currently in Phase IIa trials, as well as our other programs, including additional partnered programs with AstraZeneca and Servier , which we believe will result in our continued incurrence of significant research, development and other expenses and resources related to those programs. If our research and development efforts, including preclinical studies or the clinical trials for any of our drug candidates fail or produce unsuccessful results and those drug candidates do not gain regulatory approval, or if any of our drug candidates, if approved, fail to achieve market acceptance, we may never become profitable. In addition, the failure of one drug candidate or program may have an adverse impact on other drug candidates and programs that include our class of Anticalin therapeutic proteins. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Our prior losses, combined with expected future losses, have had and will continue to have an adverse effect on our stockholders’ equity and working capital.
We will need substantial additional funding to continue our operations, which could result in significant dilution or restrictions on our business activities. We may not be able to raise capital when needed, if at all, or on terms acceptable to us, which would force us to delay, reduce or eliminate our product development programs or commercialization efforts and could cause our business to fail.
Our operations have consumed substantial amounts of cash since inception. We expect to need substantial additional funding to pursue the preclinical and clinical development of our drug candidates, launch, and commercialize any drug candidates for which we receive regulatory approval.
We will require additional capital for the further development and commercialization of our drug candidates and programs and may need to raise additional funds sooner than we currently anticipate if we choose to and are able to expand more rapidly than we currently anticipate. Further, we expect our expenses to increase in connection with our ongoing activities, particularly as we advance additional programs through preclinical development and into the clinic and monitor the performance and continue to advance and expand our clinical programs particularly PRS-343 as well as PRS-080 and PRS-060. In addition, if we obtain regulatory approval for any of our drug candidates, we expect to incur significant commercialization expenses related to regulatory requirements, product manufacturing, marketing, sales and distribution.
Furthermore, we expect to incur additional costs associated with operating as a public company. We may also encounter unforeseen expenses, difficulties, complications, delays and other unknown factors that may increase our capital needs and/or cause us to spend our cash resources faster than we expect.
To date, we have financed our operations through a mix of equity investments from private and public investors, the incurrence of debt, grant funding, and revenues from our various collaboration agreements, and we expect to continue to finance our operations through equity investments from public investors for the foreseeable future. Additional funding from those or other sources may not be available when or in the amounts needed, on acceptable terms, or at all.
If we raise capital through the sale of equity, or securities convertible into equity, it would result in dilution to our then existing stockholders, which could be significant depending on the price at which we may be able to sell our securities. If we raise additional capital through the incurrence of indebtedness, we would likely become subject to covenants restricting our business activities, and holders of debt instruments may have rights and privileges senior to those of our equity investors. In addition, servicing the interest and principal repayment obligations under debt facilities could divert funds that would otherwise be available to support research and development, clinical or commercialization activities.
If we obtain capital through collaborative arrangements, these arrangements could require us to relinquish rights to our Anticalin®-brand technology or drug candidates and could result in our receipt of only a portion of the revenues associated with the partnered drug.
If we are unable to raise capital when needed or on attractive terms, we could be forced to delay, reduce, or eliminate our research and development for our drug candidates or any future commercialization efforts. Any of these events could significantly harm our business, financial condition, and prospects.
Our limited operating history as a clinical stage company may hinder our ability to successfully meet our objectives.
We were formed in August 2000 and since that time our focus has been on discovery of Anticalin®-brand drug candidates. We are currently conducting clinical development of PRS-343, PRS-080 and, in partnership with AstraZeneca, PRS-060, and are also advancing other drug candidates through preclinical development with the intention of initiating additional clinical-stage programs. In addition to our focus in immuno-oncology and respiratory diseases, we are also exploring additional indications that may be suitable for Anticalin-brand drug therapeutics. Our drug candidates are in early stages of development, have not obtained marketing approval, have never generated any revenue from sales, and will require extensive testing before commercialization. We have limited experience with respect to clinical-stage operations, including manufacturing required to support clinical activities and have not yet demonstrated an ability to successfully overcome many of the risks and uncertainties frequently encountered by companies in new and rapidly evolving fields, particularly in the biopharmaceutical area. In addition, the early-stage nature of our drug discovery and development operations can only provide limited operating results upon which investors can evaluate our business and prospects.
Our limited operating history may adversely affect our ability to implement our business strategy and achieve our business goals, which include, among others, the following activities:
| |
• | developing our drug candidates using unproven technologies; |
| |
• | undertaking preclinical development and clinical trials as well as formulating and manufacturing products; |
| |
• | obtaining the human, financial and other resources necessary to develop, test, manufacture, commercialize and market our drug candidates; |
| |
• | engaging corporate partners to assist in developing, testing, manufacturing and marketing our drug candidates; |
| |
• | continuing to build and maintain an intellectual property portfolio covering our technology and our drug candidates; |
| |
• | satisfying the requirements of clinical trial protocols, including patient enrollment, establishing and demonstrating the clinical safety and efficacy of our drug candidates and obtaining necessary regulatory approvals; |
| |
• | achieving acceptance and use by the medical community of our Anticalin platform and drug candidates after they receive regulatory approvals; |
| |
• | maintaining, growing and managing our internal teams as and to the extent we increase our operations and develop new segments of our business; |
| |
• | developing and maintaining successful collaboration, strategic and other relationships for the development and commercialization of our drug candidates that receive regulatory approvals with existing and new partners; and |
| |
• | managing our cash flows and any growth we may experience in an environment where costs and expenses relating to clinical trials, regulatory approvals and commercialization continue to increase. |
If we are unsuccessful in accomplishing these objectives, we may not be able to develop drug candidates, raise capital, expand our business or continue our operations.
Our global operations subject us to various risks, and our failure to manage these risks could adversely affect our results of operations.
Our business is subject to certain risks associated with doing business globally. One of our growth strategies is to pursue opportunities for our business in several areas of the world, including the United States, Europe (including Germany) and Australia, any or all of which could be adversely affected by the risks set forth below. Accordingly, we face significant operational risks as a result of doing business internationally, such as:
| |
• | fluctuations in foreign currency exchange rates; |
| |
• | potentially adverse tax consequences and changes in tax laws; |
| |
• | challenges in providing solutions across a significant distance, in different languages and among different cultures; |
| |
• | different, complex and changing laws governing intellectual property rights, sometimes affording reduced protection of intellectual property rights in certain countries; |
| |
• | difficulties in staffing and managing foreign operations, particularly in new geographic locations; |
| |
• | restrictions imposed by local labor practices and laws on our business and operations; |
| |
• | rapid changes in government, economic and political policies and conditions, political or civil unrest or instability, terrorism or epidemics and other similar outbreaks or events; |
| |
• | compliance with a wide variety of complex foreign laws, treaties and regulations; |
| |
• | tariffs, trade barriers and other regulatory or contractual limitations on our ability to develop or sell our products in certain foreign markets; and |
| |
• | becoming subject to the laws, regulations and court systems of multiple jurisdictions. |
Our failure to manage the market and operational risks associated with our international operations effectively could limit the future growth of our business and adversely affect our results of operations.
Our international operations pose currency risks, which may adversely affect our operating results and net income.
Our operating results may be affected by volatility in currency exchange rates and our ability to effectively manage our currency transaction risks. Our reporting currency is the U.S. dollar, however, 65% of our operating expenses and all of our revenues come from operations outside of the United States. As such, the financial statements are translated for reporting purposes as follows: (1) asset and liability accounts at year-end rates, (2) income statement accounts at weighted average exchange rates for the year and (3) stockholders’ equity accounts at historical rates. Corresponding translation gains or losses are recorded in stockholders’ equity.
As we realize our strategy to expand in the United States, Germany, Australia and further internationally, our exposure to currency risks will increase. We do not manage our foreign currency exposure in a manner that would eliminate the effects of changes in foreign exchange rates. Therefore, changes in exchange rates between these foreign currencies and the U.S. dollar will affect our revenues and expenses and could result in exchange losses in any given reporting period.
We incur currency transaction risks whenever we enter into either a purchase or a sale transaction using a currency other than the U.S. dollar, our functional currency, in particular our arrangements for the purchase of supplies or licensing and collaboration agreements with partners outside of the United States. In such cases, we may suffer an exchange loss because we do not currently engage in currency swaps or other currency hedging strategies to address this risk.
Given the volatility of exchange rates, we can give no assurance that we will be able to effectively manage our currency transaction risks or that any volatility in currency exchange rates will not have an adverse effect on our results of operations.
If we fail to comply with environmental, health and safety laws and regulations that apply to us, we could become subject to fines or penalties or incur costs that could harm our business.
We are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures and the handling, use, storage, treatment and disposal of hazardous materials and wastes. Our operations involve the use of hazardous and flammable materials, including chemicals and biological materials. Our operations also produce hazardous waste products. We generally contract with third parties for the disposal of any hazardous materials we use and wastes we produce. The use of these materials in our business could result in contamination or injury, which could cause damage for which we may be responsible but may not have sufficient resources to pay. We also could incur significant costs associated with civil or criminal fines and penalties for failure to comply with these laws and regulations, which we may not be able to afford.
Although we maintain workers’ compensation insurance for our operations in Germany to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations applicable to us. These current or future laws and regulations may impair our research, development or production efforts or impact the research activities we pursue, particularly with respect to research involving human subjects or animal testing. Our failure to comply with these laws and regulations also may result in substantial fines, penalties or other sanctions, which could cause our financial condition to suffer.
Health and safety regulations in the United States, Germany, and Australia and in the countries where our technology and potential products are developed, licensed or sold may prevent the sale or use of our technology or products in the future.
We are subject to a variety of regulations regarding worker health and safety in the United States, European Union (Germany), Australia, and in the countries where our technology and potential products are licensed or sold. Because our technology and potential products may frequently involve the manufacture or use of certain chemical or biological compounds, we are required to certify their safety for industrial use and development in a variety of countries and contexts. As there has not been sufficient testing to determine the long-term health and environmental risks of our Anticalin drug candidates and the materials used in the production of such drug candidates and any future products, future regulations may ban the use of our products due to the potential risk they pose to workers or may limit the use of our drug candidates in research and commercial settings. Any such regulations may have a substantial negative impact on our business and revenues, and may cause our business to fail. Because we cannot guarantee the long-term safety of use or exposure to materials used during development or manufacture of our products, we may face liability for health risks or harms caused as a result of developing, manufacturing or other processes that use such materials. Any such claims may have a negative impact on our revenues and may prove substantially disruptive to our business in the future.
In addition, under the European Union regulation on classification, labeling and packaging of substances and mixtures, or CLP, and under other regulations in the United States or other countries related to the clinical development of our drug candidates (including, for example, submissions to regulatory authorities such as the FDA and EMA as well as submissions related to obtaining a non-proprietary (INN and USAN) name for our clinical drug candidates to the World Health Organization (WHO) and United States Adopted Name Council (USAN Council), we may be required to publicly disclose the composition of our proprietary products or substances, which may facilitate infringement or avoidance of our intellectual property by third parties and may potentially reduce the margin we are able to charge for our products by allowing competitors to more accurately determine our production costs. Future development of these regulations may have a further negative impact our revenues and a substantial negative impact on our business.
We may be limited in our use of our net operating loss carryforwards.
As of December 31, 2017, the Company had net operating loss carryforwards for United States federal income tax purposes of $24.5 million and net operating loss carryforwards for state income tax purposes of $21.3 million. These tax loss carryforwards expire through 2037. In the United States, utilization of the NOL carryforwards may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986 and similar state provisions due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of
NOL carryforwards that can be utilized annually to offset future taxable income and tax, respectively. If we were to lose the benefits of these loss carryforwards, our future earnings and cash resources would be materially and adversely affected. The Company has not currently completed a study to assess whether an ownership change has occurred, or whether there have been multiple ownership changes since the Acquisition.
As of December 31, 2017, the Company had German corporate income tax and trade tax net operating loss carryforwards of approximately $46.1 million and $45.6 million, respectively, which may be used to reduce our future taxable income in our German jurisdiction. Under current German laws, tax loss carryforwards may only be used to offset any relevant later assessment period (calendar year) $1.2 million plus 60% of the exceeding taxable income and trade profit of such period. In addition, certain transactions, including transfers of shares or interest in the loss holding entity, may result in the partial or total forfeiture of tax losses existing at that date. Partial or total forfeiture of tax losses may further occur in corporate reorganizations of the loss holding entity.
Our business and operations would suffer in the event of system failures, and our operations are vulnerable to interruption by natural disasters, terrorist activity, power loss and other events beyond our control, the occurrence of which could materially harm our business.
Despite the implementation of security measures, our internal computer systems and those of our contractors and consultants are vulnerable to damage from computer viruses, hacking, ransomware, cyber-attacks, unauthorized access as well as telecommunication and electrical failures. While we have not experienced any such system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our drug development programs. For example, the loss of clinical trial data from completed, ongoing, or planned clinical trials could result in delays in our regulatory approval efforts and we may incur substantial costs to attempt to recover or reproduce the data. If any disruption or security breach resulted in a loss of or damage to our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and/or the further development of our drug candidates could be delayed.
We are also vulnerable to accidents, electrical blackouts, labor strikes, terrorist activities, war and natural disasters and other events beyond our control, and we have not undertaken a systematic analysis of the potential consequences to our business as a result of any such events and do not have an applicable recovery plan in place. Except for our operations in Germany, where we have business interruption insurance against losses or damages resulting from fire, we do not carry other business interruption insurance that would compensate us for actual losses from interruptions of our business that may occur, and any losses or damages incurred by us could cause our business to materially suffer.
Our failure to comply with data protection laws and regulations could lead to government enforcement actions, private litigation and/or adverse publicity and could negatively affect our operating results and business.
We are subject to data protection laws and regulations that address privacy and data security. The legislative and regulatory landscape for data protection continues to evolve, and in recent years there has been an increasing focus on privacy and data security issues. In the United States, numerous federal and state laws and regulations, including state data breach notification laws, state health information privacy laws and federal and state consumer protection laws govern the collection, use, disclosure and protection of health-related and other personal information. Failure to comply with data protection laws and regulations could result in government enforcement actions, which could include civil or criminal penalties, private litigation and/or adverse publicity and could negatively affect our operating results and business. In addition, in May 2016, the European Union Parliament adopted the comprehensive General Data Privacy Regulation (the “GDPR”) to, among other things, impose more stringent data protection requirements and provide for greater penalties for noncompliance. The GDPR will become fully effective in May 2018. Complying with the enhanced obligations imposed by the GDPR and other regulations may result in significant costs to our business and require us to amend certain of our business practices. Further, enforcement actions and investigations by regulatory authorities related to data security incidents and privacy violations continue to increase. The future enactment of more restrictive laws, rules or regulations and/or future enforcement actions or investigations could have a materially adverse impact on us through increased costs or restrictions on our businesses, and noncompliance could result in regulatory penalties and significant legal liability.
Significant disruptions of information technology systems or security breaches could adversely affect our business.
We are increasingly dependent upon information technology systems, infrastructure and data to operate our business. In the ordinary course of business, we collect, store and transmit large amounts of confidential information (including, among other things, trade secrets or other intellectual property, proprietary business information and personal information). It is critical that we do so in a secure manner to maintain the confidentiality and integrity of such confidential information. We also have
outsourced elements of our operations to third parties, and as a result we manage a number of third-party vendors who may or could have access to our confidential information. The size and complexity of our information technology systems, and those of third-party vendors with whom we contract, and the large amounts of confidential information stored on those systems, make such systems vulnerable to service interruptions or to security breaches from inadvertent or intentional actions by our employees, third-party vendors, and/or business partners, or from cyber-attacks by malicious third parties. Cyber-attacks are increasing in their frequency, sophistication and intensity, and have become increasingly difficult to detect. Cyber-attacks could include the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering and other means to affect service reliability and threaten the confidentiality, integrity and availability of information.
Significant disruptions of our information technology systems, or those of our third-party vendors, or security breaches could adversely affect our business operations and/or result in the loss, misappropriation and/or unauthorized access, use or disclosure of, or the prevention of access to, confidential information, including, among other things, trade secrets or other intellectual property, proprietary business information and personal information, and could result in financial, legal, business and reputational harm to us. For example, any such event that leads to unauthorized access, use or disclosure of personal information, including personal information regarding our patients or employees, could harm our reputation, require us to comply with federal and/or state breach notification laws and foreign law equivalents, and otherwise subject us to liability under laws and regulations that protect the privacy and security of personal information. Security breaches and other inappropriate access can be difficult to detect, and any delay in identifying them may lead to increased harm of the type described above. While we have implemented security measures to protect our information technology systems and infrastructure, there can be no assurance that such measures will prevent service interruptions or security breaches that could adversely affect our business.
The recently passed tax reform bill in the United States could adversely affect our business and financial condition.
On December 22, 2017, President Trump signed into law the “Tax Cuts and Jobs Act,” or TCJA, which significantly reforms the Internal Revenue Code of 1986, as amended, or the Code. The TCJA, among other things, includes changes to U.S. federal tax rates, imposes significant additional limitations on the deductibility of interest and net operating loss carryforwards, allows for the expensing of capital expenditures, and puts into effect the migration from a “worldwide” system of taxation to a territorial system. Our net deferred tax assets and liabilities will be revalued at the newly enacted U.S. corporate rate, and the impact, if any, will be recognized in our tax expense in the year of enactment.
We continue to examine the impact this tax reform legislation may have on our business. Notwithstanding the reduction in the corporate income tax rate, the overall impact of the TCJA is uncertain and our business and financial condition could be adversely affected. The impact of this tax reform on holders of our securities is also uncertain and could be adverse. This annual report does not discuss any such tax legislation or the manner in which it might affect us or purchasers of our securities. We urge our investors to consult with their legal and tax advisors with respect to such legislation and the potential tax consequences of investing in our securities.
There could be an adverse change or increase in the laws and/or regulations governing our business.
We are subject to various laws and regulations in different jurisdictions, and the interpretation and enforcement of laws and regulations are subject to change. We are also subject to different tax regulations in each of the jurisdictions where we conduct our business or where our management is located. We expect the scope and extent of regulation in the jurisdictions in which we conduct our business, or where our management is located, as well as regulatory oversight and supervision, to generally continue to increase. There can be no assurance that future regulatory, judicial and legislative changes in any jurisdiction will not have a material adverse effect on us or hinder us in the operation of its business.
Risks Related to the Discovery and Development of Our Drug Candidates
We are heavily dependent on the successful development of our drug candidates and programs and we cannot be certain that we will receive regulatory approvals or be able to successfully commercialize our products even if we receive regulatory approvals.
We currently have no products that are approved for commercial sale. We expect that a substantial portion of our efforts and expenditures over the next few years will be devoted to our immuno-oncology programs, particularly PRS-343, our partnered programs with Servier, and other drug candidates, as well as our respiratory programs including PRS-060, our partnered program with AstraZeneca, PRS-080, our hepcidin antagonist, as well as our other programs. We completed dosing of healthy volunteers in a Phase Ia clinical trial with PRS-080 in June 2015, initiated a Phase Ib clinical trial in the first quarter of 2016 (which was completed) and a Phase IIa in the last quarter of 2017. For PRS-343 a Phase I trial was initiated in the second
quarter of 2017. In partnership with AstraZeneca, PRS-060 is in clinical development with Phase I trials initiated in the last quarter of 2017. We are engaged in research and development activities with respect to a number of additional drug candidates and programs. All of our other drug candidates are in the discovery or early preclinical to IND-enabling stage. Accordingly, our business is currently substantially dependent on the successful development, clinical testing, regulatory approval and commercialization of PRS-343, PRS-060, PRS-080 and our other immuno-oncology programs, which may never occur.
Before we can generate any revenues from sales of our lead drug candidates, we must complete the following activities for each of them, any one of which we may not be able to successfully complete:
| |
• | conduct additional preclinical and clinical development; |
| |
• | manage preclinical, manufacturing and clinical activities; |
| |
• | obtain regulatory approval; |
| |
• | establish manufacturing relationships for the clinical and post-approval supply of the applicable drug candidate; |
| |
• | build a commercial sales and marketing team, either internally or by contract with third parties; |
| |
• | develop and implement marketing strategies; and |
| |
• | invest significant additional cash in each of the above activities. |
Clinical testing of PRS-060 and PRS-343 were initiated in 2017 and clinical testing of PRS-080 is ongoing, while clinical testing for other programs including our immuno-oncology programs, have not yet commenced, and the results of any future clinical trials or preclinical studies of these programs, if unsuccessful, could lead to our abandonment of the development of those drug candidates. If studies of these drug candidates produce unsuccessful results and we are forced or elect to cease their development, our business and prospects would be substantially harmed.
Preclinical and clinical testing of our drug candidates that have been conducted to date or will be conducted in future may not have been or may not be performed in compliance with applicable regulatory requirements, which could lead to increased costs or material delays for their further development.
Given the complexity as well as the uncertainty inherent in biopharmaceutical preclinical studies and clinical trials, and because of our limited operating experience, we may discover that our own development activities have not been or are not in compliance with applicable regulatory requirements or have otherwise been or are deficient, and, therefore, advancement of the development of the drug candidates on the basis of those trials and studies is not warranted or will be delayed.
We have also entered into license, partnership, and option arrangements, such as with Allergan, Sanofi, Zydus, Roche, Servier, Aska, AstraZeneca, and Seattle Genetics, relating to certain of our drug candidates, and may continue to do so in the future. Under certain of such arrangements, the development of those drug candidates has been, or in the future may be, conducted wholly by such partners or any third parties with which the partners contract. As a result, we have not been or may not be closely involved with or have any control over those development activities. Although certain of such partners have provided information regarding those drug candidates and the related preclinical studies conducted to date, including certain data that is included in this Annual Report on Form 10-K, we have not received and do not yet have access to comprehensive information regarding those development activities, including the raw data from the studies that have been conducted, information regarding the design, procedural implementation and structure and information regarding the manufacture of the drug candidates used in the studies. Because we have had no input on the development to date of these drug candidates, we may discover that all or certain elements of the trials and studies our partners have performed have not been, or may not in the future be, in compliance with applicable regulatory standards or have otherwise been or may be deficient, and that advancement of the development of these drug candidates on the basis of those trials and studies is not warranted.
Further, the majority of our development activities for each of our drug candidates to date, including our Phase I and Phase II clinical trials with PRS-080, which is being conducted in Germany, our Phase I clinical trials with PRS-060, which is being conducted in Australia, and our Phase I clinical trial with PRS-343, which is currently being conducted in the United States, and our anticipated future clinical trials, have been, are being, or may in the future be conducted in or outside the United States, including in Europe or Australia, and we may conduct some of our future development activities in other countries or regions. As a result, although those studies may meet the standards of certain applicable foreign regulatory bodies, the structure and design of those clinical trials and preclinical studies may not meet applicable U.S. Food and Drug Administration, or FDA,
requirements and also may not meet the requirements of the applicable regulatory authorities in foreign countries in which we desire to pursue marketing approval for these drug candidates.
If the studies conducted by us or our partners or collaborators do not comply with applicable regulatory requirements or are otherwise not eligible for continued development in the United States, then we or our partners may be forced to conduct new studies in order to progress the development of our drug candidates. We, or our partners, may not have the funding or other resources to conduct or complete these additional studies, which would severely delay or prevent the development plans for these drug candidates and their commercialization. Any such deficiency and delay in the development of these drug candidates would significantly harm our business plans, product revenues and prospects.
Our research and development is based on a rapidly evolving area of science, and our approach to drug discovery and development is novel and may never lead to marketable products.
Biopharmaceutical product development is generally a highly speculative undertaking and by its nature involves a substantial degree of risk. Our specific line of business, the discovery of Anticalin-brand drug therapeutics for patients with a variety of diseases and conditions, such as anemia, asthma and cancer, is an emerging field, and the scientific discoveries that form the basis for our efforts to develop drug candidates are relatively new. Further, the scientific evidence to support the feasibility of developing drug candidates based on those discoveries is both preliminary and limited. In contrast to companies who focus on more traditional drug classes, such as antibodies and small molecules, we believe we are the first, if not the only company, to work with Anticalin-brand drug therapeutics and work to advance these to a clinical stage of development. We are not aware of any company that has successfully developed and obtained approval for a drug based on Anticalin proteins. As a result, identifying drug targets based in part on their suitability with Anticalin-brand drug therapeutics, which is a fundamental aspect of our business approach, may not lead to the discovery or development of any drugs that successfully treat patients with the diseases and conditions we intend to target. Moreover, the lack of successful precedents in the development of Anticalin proteins could result in added complexities or delays in our development efforts. The failure of the scientific underpinnings of our business model to produce viable drug candidates would substantially harm our operations and prospects.
We may not be successful in our efforts to build a pipeline of drug candidates.
A key element of our strategy is to use and expand our Anticalin drug platform to build a pipeline of drug candidates to address different targets, and progress those drug candidates through clinical development for the treatment of a variety of different types of diseases. Although our research efforts to date have resulted in identification of a series of targets, we may not be able to develop drug candidates that have good drug-like properties (target affinity, stability, half-life, etc.) and are safe and effective inhibitors or promoters of all or any of these targets. Even if we are successful in building a product pipeline, the potential drug candidates that we identify may not be suitable for clinical development for a number of reasons, including causing harmful side effects or demonstrating other characteristics that indicate a low likelihood of receiving marketing approval or achieving market acceptance. If our methods of identifying potential drug candidates fail to produce a pipeline of potentially viable drug candidates, then our success as a business will be dependent on the success of fewer potential drug candidates, which introduces risks to our business model and potential limitations to any success we may achieve.
Clinical drug development involves a lengthy and expensive process with uncertain outcomes, is very difficult to design and implement, and any of our clinical trials could produce unsuccessful results or fail at any stage in the process.
Clinical trials conducted on humans are expensive and can take many years to complete, and outcomes are inherently uncertain. Failure can occur at any time during the clinical trial process. Additionally, any positive results of preclinical studies and early clinical trials of a drug candidate may not be predictive of the results of later-stage clinical trials, such that drug candidates may reach later stages of clinical trials and fail to show the desired safety and efficacy traits despite having shown indications of those traits in preclinical studies and initial clinical trials. A number of companies in the biopharmaceutical industry have suffered significant setbacks in advanced clinical trials due to lack of efficacy or adverse safety profiles, notwithstanding promising results in earlier phases of the trials. Therefore, the results of any ongoing or future clinical trials we conduct may not be successful.
We initiated Phase I clinical trials for PRS-060 and PRS-343 in 2017. In addition, we completed dosing of healthy volunteers in the clinical Phase Ia trial for PRS-080 in 2015 and initiated a Phase Ib trial in the first quarter of 2016 (which has been completed) and initiated a Phase IIa trial in 2017. We may however experience delays in pursuing those or any other clinical trials, and any planned clinical trials may not begin on time, may require redesign, may not enroll sufficient healthy volunteers or patients in a timely manner, and may not be completed on schedule, if at all.
Clinical trials may be delayed for a variety of reasons, including delays related to:
| |
• | obtaining regulatory approval to commence a trial; |
| |
• | reaching agreement on acceptable terms with prospective contract research organizations, or CROs, and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| |
• | obtaining institutional review board, or IRB, approval at each trial site; |
| |
• | enrolling suitable volunteers or patients to participate in a trial; |
| |
• | developing and validating companion diagnostics, if they are deemed necessary, on a timely basis; |
| |
• | changes in dosing or administration regimens; |
| |
• | failure of patients to complete a trial or return for post-treatment follow-up; |
| |
• | inability to monitor patients adequately during or after treatment; |
| |
• | clinical investigators deviating from trial protocols or dropping out of a trial; |
| |
• | regulators instituting a clinical hold due to observed safety findings or other reasons; |
| |
• | adding new clinical trial sites; and |
| |
• | manufacturing sufficient quantities of drug candidate of acceptable quality for use in clinical trials. |
We rely and plan to continue to rely on CROs, CMOs and clinical trial sites to ensure the proper and timely conduct of our clinical trials. Although we have and expect that we will have agreements in place with CROs and CMOs governing their contracted activities and conduct, we will have limited influence over their actual performance. As a result, we ultimately do not and will not have control over a CRO or CMO’s compliance with the terms of any agreement it may have with us, its compliance with applicable regulatory requirements, or its adherence to agreed upon time schedules and deadlines, and a future CRO or CMO’s failure to perform those obligations could subject any of our clinical trials to delays or failure.
Further, we may also encounter delays if a clinical trial is suspended or terminated by us, by any IRB or Ethics Committee at an institution in which such trials are being conducted, by the Data Safety Monitoring Board, or DSMB, for the trial, if applicable, or by the FDA, the European Medicines Agency, or EMA, or other regulatory authorities. Such authorities may impose such a suspension or termination due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements, inspection of the clinical trial operations or trial site by the FDA, EMA or other regulatory authorities resulting in the imposition of a clinical hold, exposing participants to health risks caused by unforeseen safety issues or adverse side effects, development of previously unseen safety issues, failure to demonstrate a benefit from using a drug candidate, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial. Therefore, we cannot predict with any certainty the schedule for commencement or completion of any currently ongoing, planned or future clinical trials.
If we experience delays in the commencement or completion of, or suspension or termination of, any clinical trial for our drug candidates, the commercial prospects of the drug candidate could be harmed, and our ability to generate product revenues from the drug candidate may be delayed or eliminated. In addition, any delays in completing our clinical trials will increase our costs, slow down our drug candidate development and approval process and jeopardize regulatory approval of our drug candidates and our ability to commence sales and generate revenues. The occurrence of any of these events could harm our business, financial condition, results of operations and prospects significantly.
If we experience delays or difficulties in the enrollment of research subjects in clinical trials, those clinical trials could take longer than expected to complete and our receipt of necessary regulatory approvals could be delayed or prevented.
We may not be able to initiate or continue clinical trials for our drug candidates if we are unable to locate and enroll a sufficient number of research subjects to participate in these trials. In particular, for some diseases and conditions we are or will be focused on, our pool of suitable patients may be smaller and more selective and our ability to enroll a sufficient number of suitable patients may be limited or take longer than anticipated. In addition, some of our competitors have ongoing clinical
trials for drug candidates that treat the same indications as our drug candidates, and volunteers or patients who would otherwise be eligible for our clinical trials may instead enroll in clinical trials of our competitors’ drug candidates.
Patient enrollment for any of our clinical trials may also be affected by other factors, including without limitation:
| |
• | the severity of the disease under investigation; |
| |
• | the frequency of the molecular alteration we are seeking to target in the applicable trial; |
| |
• | the eligibility criteria for the clinical trial in question; |
| |
• | the perceived risks and benefits of the drug candidate under the clinical trial; |
| |
• | the extent of the efforts to facilitate timely enrollment in clinical trials; |
| |
• | the patient referral practices of physicians; |
| |
• | the ability to monitor volunteers or patients adequately during and after treatment; |
| |
• | the presence of other drug candidates in clinical development for the same indication or against the same target; and |
| |
• | the proximity and availability of clinical trial sites. |
Our inability to enroll a sufficient number of patients for our clinical trials would result in significant delays and could require us to abandon one or more clinical trials altogether. Enrollment delays in our clinical trials may result in increased development costs for our drug candidates, and we may not have or be able to obtain sufficient cash to fund such increased costs when needed, which could result in the further delay or termination of the trial.
The review processes of regulatory authorities are lengthy, time consuming, expensive and inherently unpredictable. If we are unable to obtain approval for our drug candidates from applicable regulatory authorities, we will not be able to market and sell those drug candidates in those countries or regions and our business could be substantially harmed.
The research, testing, manufacturing, labeling, approval, sale, marketing and distribution of drug products are, and will remain, subject to extensive regulation by the FDA in the United States and by the respective regulatory authorities in other countries, which regulations differ from country to country. We are not permitted to market our drug candidates in the United States until we receive the respective approval of a biologics license application, or BLA, from the FDA, or in any foreign countries until we receive the requisite approval from the respective regulatory authorities in such countries. The time required to obtain approval, if any, by the FDA, EMA and comparable foreign authorities is unpredictable, but typically takes many years following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion of the regulatory authorities. We have not submitted a marketing application such as a BLA to the FDA, a marketing authorization application, or MAA, to the EMA or any similar application to any other jurisdiction. Moreover, we have not obtained regulatory approval for any drug candidate in any jurisdiction and it is possible that none of our existing drug candidates or any drug candidates we may seek to develop in the future will ever obtain regulatory approval.
Our drug candidates could fail to receive regulatory approval for many reasons, including any one or more of the following:
| |
• | the FDA, EMA or comparable foreign regulatory authorities may disagree with the design or implementation of our clinical trials; |
| |
• | we may be unable to demonstrate to the satisfaction of the FDA, EMA or comparable foreign regulatory authorities that a drug candidate is safe and effective for its proposed indication; |
| |
• | the results of clinical trials may not meet the level of statistical significance required by the FDA, EMA or comparable foreign regulatory authorities for approval; |
| |
• | we may be unable to demonstrate that a drug candidate’s clinical and other benefits outweigh its safety risks; |
| |
• | the FDA, EMA or comparable foreign regulatory authorities may disagree with our interpretation of data from preclinical studies or clinical trials; |
| |
• | the data collected from clinical trials of our drug candidates may not be sufficient to support the submission of a BLA or other submission or to obtain regulatory approval in the United States or elsewhere; |
| |
• | the manufacturing processes or facilities of third-party manufacturers with which we contract for clinical and commercial supplies may fail to meet the requirements of the FDA, EMA or comparable foreign regulatory authorities; |
| |
• | the FDA, EMA or comparable foreign regulatory authorities may fail to approve the companion diagnostics we contemplate developing internally or with partners; and |
| |
• | the change of the standard of care or approval policies or regulations of the FDA, EMA or comparable foreign regulatory authorities may significantly change in a manner rendering our clinical data insufficient for approval. |
The time and expense of the approval process, as well as the unpredictability of future clinical trial results and other contributing factors, may result in our failure to obtain regulatory approval to market, in one or more jurisdictions, PRS-343, PRS-060, PRS-080, our other IO programs, our discovery stage programs, or any other drug candidates we are developing or may seek to develop in the future, which would significantly harm our business, results of operations and prospects. In such case, we may also not have the resources to conduct new clinical trials and/or we may determine that further clinical development of any such drug candidate is not justified and may discontinue any such programs.
In order to market and sell our products in any jurisdiction, we or our third-party collaborators must obtain separate marketing approvals in that jurisdiction and comply with its regulatory requirements. The review and approval procedures can vary drastically among jurisdictions, and each jurisdiction may impose different testing and other requirements to obtain and maintain marketing approval. Further, the time required to obtain those approvals, if any, may differ substantially among jurisdictions. In addition, in many countries or regions outside the United States, it is required that the product be approved for reimbursement before the product can be approved for sale in that country or region. Moreover, approval by the FDA or an equivalent foreign authority does not ensure approval by regulatory authorities in any other countries or regions. As a result, the ability to market and sell a drug candidate in more than one jurisdiction can involve significant additional time, expense and effort, and would subject us and our collaborators to the numerous and varying post-approval requirements of each jurisdiction governing commercial sales, manufacturing, pricing and distribution of our drug candidates. We or any third parties with whom we may collaborate may not have the resources to pursue those approvals, and we or they may not be able to obtain any approvals that are pursued. The failure to obtain marketing approval for our drug candidates in foreign jurisdictions could severely limit their potential market and ability to generate revenue.
In addition, even if we were to obtain regulatory approval in one or more jurisdictions, regulatory authorities may approve any of our drug candidates for fewer or more limited indications than we request, may not approve prices we may propose to charge for our products, may grant approval contingent on the performance of costly post-marketing clinical trials, or may approve a drug candidate with a label that does not include the labeling claims necessary or desirable for the successful commercialization of that drug candidate. Any of the foregoing circumstances could materially harm the commercial prospects for our drug candidates.
We may expend our limited resources to pursue a particular drug candidate or indication that does not produce any commercially viable products and may fail to capitalize on drug candidates or indications that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and managerial resources, we must focus our efforts on particular research programs and drug candidates for specific indications. As a result, we may forgo or delay pursuit of opportunities with other drug candidates or for other indications that later prove to have greater commercial potential. Further, our resource allocation decisions may result in our use of funds for research and development programs and drug candidates for specific indications that may not yield any commercially viable products.
If we do not accurately evaluate the commercial potential or target market for a particular drug candidate, we may relinquish valuable rights to that drug candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such drug candidate. Any such failure to improperly assess potential drug candidates could result in missed opportunities and/or our focus on drug candidates with low market potential, which would harm our business and financial condition.
Risks Related to Our Dependence on Third Parties
We rely on third parties to conduct our clinical trials and preclinical studies. If these third parties do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for our drug candidates and our business could be substantially harmed.
We depend upon independent investigators and contractors, such as CROs, universities and medical institutions, to conduct our clinical trials and preclinical studies. We rely upon, and plan to continue to rely upon, such third-party entities to execute our clinical trials and preclinical studies and to monitor and manage data produced by and relating to those studies and trials. However, we may not be able to in the future establish arrangements with CROs when needed or on terms that are acceptable to us, or at all, which could negatively affect our development efforts with respect to our drug candidates and materially harm our business, operations and prospects. As a result of the use of third-party contractors, we will have only limited control over certain aspects of their activities. Nevertheless, we are responsible for ensuring that each of our studies, including each of our clinical trials, is conducted in accordance with the applicable protocol, legal and regulatory requirements as well as scientific standards, and our reliance on any third-party entity will not relieve us of our regulatory responsibilities.
Based on our present expectations, we and our third-party contractors will be required to comply with current Good Clinical Practice, or cGCP, for all of our drug candidates in clinical development. Regulatory authorities enforce cGCP through periodic inspections of trial sponsors, clinical investigators and trial sites. If we or any of our contractors fail to comply with applicable cGCP, the clinical data generated in the applicable trial may be deemed unreliable and the FDA, EMA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving a drug candidate for marketing, which we may not have sufficient cash or other resources to support and which would delay our ability to generate revenue from any sales of such drug candidate. Any agreements governing our relationships with outside contractors such as CROs, or CROs or other contractors we may engage in the future, may provide those outside contractors with certain rights to terminate a clinical trial under specified circumstances. If such an outside contractor terminates its relationship with us during the performance of a clinical trial, we would be forced to seek an engagement with a substitute contractor, which we may not be able to do on a timely basis or on commercially reasonable terms, if at all, and the applicable clinical trial would experience delays or may not be completed.
If our contractors do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they need to be replaced or if the quality or accuracy of the data they obtain is compromised due to a failure to adhere to our clinical protocols, legal and regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for, or successfully commercialize, the affected drug candidates. In addition, we will be unable to control whether or not they devote sufficient time and resources to our preclinical and clinical programs. These outside contractors may not assign as great a priority to our programs or pursue them as diligently as we would if we were undertaking such programs ourselves. As a result, our operations and the commercial prospects for the effected drug candidates would be harmed, our costs could increase and our ability to generate revenues could be delayed. These contractors may also have relationships with other commercial entities, some of whom may compete with us. If our contractors assist our competitors to our detriment, our competitive position would be harmed.
We rely and expect to continue to rely completely on third parties to formulate and manufacture our preclinical, clinical trial and post-approval drug supplies. The development and commercialization of any of our drug candidates could be stopped, delayed or made less profitable if those third parties fail to provide us with sufficient quantities of such drug supplies or fail to do so at acceptable quality levels, including in accordance with applicable regulatory requirements or contractual obligations, and our operations could be harmed as a result.
We have no experience in drug formulation or manufacturing. We do not currently have, nor do we plan to acquire, the infrastructure or capability internally, such as our own manufacturing facilities, to manufacture our preclinical and clinical drug supplies for use in the conduct of our clinical trials and preclinical studies or commercial quantities of any drug candidates that may obtain regulatory approval. Therefore, we lack the resources and expertise to formulate or manufacture our own drug candidates. We have entered into agreements with third-party contract manufacturers, or CMOs, for the clinical-stage manufacture of certain of our drug candidates, including PRS-343, PRS-080 and others. We plan to enter into agreements with one or more manufacturers to manufacture, supply, store, and distribute drug supplies for our current and future clinical trials and/or commercial sales. We intend to establish or continue those relationships for the supply of our drug candidates, however, there can be no assurance that we will be able to retain those relationships on commercially reasonable terms, if at all. If we are unable to maintain those relationships, we could experience delays in our development efforts as we locate and qualify new CMOs. If any of our current drug candidates or any drug candidates we may develop or acquire in the future receive regulatory approval, we will rely on one or more CMOs to manufacture the commercial supply of such drugs.
Our reliance on a limited number of CMOs exposes us to the following risks:
| |
• | We may be unable to identify manufacturers on acceptable terms, or at all, because the number of potential manufacturers is limited Following BLA approval, a change in the manufacturing site could require additional approval from the FDA. This approval would require new testing and compliance inspections. In addition, a new manufacturer would have to be educated in, or develop substantially equivalent processes for the production of our products after their receipt of FDA approval, if any. |
| |
• | Our third-party manufacturers might be unable to formulate and manufacture our drugs in the volume and of the quality required to meet our clinical needs and commercial needs, if any. |
| |
• | Our future contract manufacturers may not perform as contractually agreed or may not remain in the contract manufacturing business for the time required to supply our clinical trials or to successfully produce, store and distribute our products. |
| |
• | Drug manufacturers are subject to ongoing periodic unannounced inspection by the FDA, and corresponding state agencies to ensure strict compliance with current good manufacturing practices, or cGMP, regulations and other government regulations and corresponding foreign standards. We do not have control over third-party manufacturers’ compliance with these regulations and standards. |
| |
• | If any third-party manufacturer makes improvements in the manufacturing process for our products, we may not own, or may have to share, the intellectual property rights to the innovation. |
Each of these risks could delay our clinical trials, the approval, if any, of our drug candidates by the FDA or the commercialization of our drug candidates or result in higher costs or deprive us of potential product revenues.
Although our agreements with our CMOs require them to perform according to certain cGMP standards such as those relating to quality control, quality assurance and qualified personnel, we cannot control the ability of our contract manufacturers to implement and maintain these standards. If any of our contract manufacturers cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA, EMA or other comparable foreign authorities, we would be prevented from obtaining regulatory approval for our drug candidates unless and until we engage a substitute contract manufacturer that can comply with such requirements, which we may not be able to do. Any such failure by any of our contract manufacturers would significantly impact our ability to develop, obtain regulatory approval for or market our drug candidates, if approved.
Further, we plan to rely on our manufacturers to purchase from third-party suppliers the materials necessary to produce our drug candidates for our clinical trials. We do not have, nor do we expect to enter into, any agreements for the commercial production of these raw materials, and we do not expect to have any control over the process or timing of our contract manufacturers’ acquisition of raw materials needed to produce our drug candidates. Any significant delay in the supply of a drug candidate or the raw material components thereof for an ongoing clinical trial due to a manufacturer’s need to replace a third-party supplier of raw materials could considerably delay completion of our clinical trials, product testing and potential regulatory approval of our drug candidates. Additionally, if our future manufacturers or we are unable to purchase these raw materials to commercially produce any of our drug candidates that gains regulatory approvals, the commercial launch of our drug candidates would be delayed or there would be a shortage in supply, which would impair our ability to generate revenues from the sale of our drug candidates.
Disagreements with respect to the commercial terms of our sales, licensing, purchase or manufacturing agreements may limit our commercial success.
The rights and obligations of the partners to which we may license our Anticalin technology are governed by the licensing and collaboration agreements we enter into with those partners. In addition, our relationships with CROs and CMOs are governed by the service agreements between us and each manufacturer. Although we attempt to address the full range of possible events that may occur during the development or the manufacturing of Anticalin drug candidates and products, unanticipated or extraordinary events may occur beyond those contemplated by said agreements. Furthermore, our business relationships with our product manufacturers and our collaborators may include assumptions, understandings or agreements that are not included in our agreements with them, or that are inaccurately or incompletely represented by their terms. In addition, key terms in such agreements may be misunderstood or contested, even when both we and the other party previously believed that we had a mutual understanding of our obligations.
Any differences in interpretation or misunderstandings between us and other parties may result in substantial costs and delays with respect to the development, manufacturing or sale of Anticalin drugs, and may negatively impact our revenues and
operating results. Product manufacturers may fail to produce the products and partners may fail to develop the drug candidates with the diligence or under the timeline or in the manner we anticipated, and results may differ from the terms upon which we had agreed. As a result, we may be unable to supply drugs of the quality or in the quantity demanded or required. We may suffer harm to our reputation in the market from missed development goals or deadlines, and may be unable to capitalize upon market opportunities as a result. Resolution of these problems may entail costly and lengthy litigation or dispute resolution procedures. In addition, there is no guarantee that we will prevail in any such dispute or, if we do prevail, that any remedy we receive, whether legal or otherwise, will adequately redress the harm we have suffered. The delays and costs associated with such disputes may themselves harm our business and reputation and limit our ability to successfully compete in the market going forward.
We depend on third parties and intend to continue to license or collaborate with third parties, and events involving these strategic partners or any future collaboration could delay or prevent us from developing or commercializing products.
Our business strategy and our short- and long-term operating results depend in part on our ability to execute on existing strategic collaborations and to license or partner with new strategic partners. We have entered into and expect in the future to enter into collaborative arrangements with established pharmaceutical companies, which will lead, finance or otherwise collaborate or assist in the development, manufacture and marketing of drug products. We believe collaborations allow us to leverage our resources and technologies and we anticipate deriving some revenues from research and development fees, license fees, milestone payments, and royalties from collaborative partners.
Our prospects, therefore, may depend to some extent upon our ability to attract and retain collaborative partners and to develop technologies and products that meet the requirements of current or prospective collaborative partners. We have limited control over the amount and timing of resources that our current collaborators or any future collaborators devote to our collaborations or potential products. These collaborators may breach or terminate their agreements with us or otherwise fail to conduct their collaborative activities successfully and in a timely manner. Further, our collaborators may not develop or commercialize products that arise out of our collaborative arrangements or devote sufficient resources to the development, manufacture, marketing or sale of these products. In addition, our collaborative partners may have the right to abandon research projects, guide strategy regarding prosecution of relevant patent applications and terminate applicable agreements, including funding obligations, prior to or upon the expiration of the agreed-upon research terms. By entering into such collaborations, we may preclude opportunities to collaborate with other third parties who do not wish to associate with our existing third-party strategic partners. In the event of termination of a collaboration agreement, termination negotiations may result in less favorable terms.
There can be no assurance that we will be successful in establishing collaborative arrangements on acceptable terms or at all, that collaborative partners will not terminate funding before completion of projects, that our collaborative arrangements will result in successful product commercialization, or that we will derive any revenues from such arrangements. Potential collaborators may reject collaborations based upon their assessment of our financial, regulatory or intellectual property position and our internal capabilities. Additionally, the negotiation, documentation and implementation of collaborative arrangements are complex and time-consuming. Our discussions with potential collaborators may not lead to the establishment of new collaborations on favorable terms and may have the potential to provide collaborators with access to our key intellectual property filings.
Our success depends in part on the efforts of our current and possible future collaborators, who will likely have substantial control and discretion over the continued development and commercialization of drug candidates that are the subject of our collaborations.
Our current collaborators and future collaborators will have significant discretion in determining the efforts and amount of resources that they dedicate to our collaborations. Our collaborators may determine not to proceed with clinical development or commercialization of a particular drug candidate for a number of reasons that are beyond our control, even under circumstances where we might have continued such a program. In addition, our rights to receive milestone payments and royalties from our collaborators will depend in part on our collaborators’ abilities to establish the safety and efficacy of our drug candidates, obtain regulatory approvals and achieve market acceptance of products developed from our drug candidates. We may also depend on our collaborators to manufacture clinical scale quantities of some of our drug candidates and, possibly, for commercial scale manufacture, distribution and sales. Our collaborators may not be successful in manufacturing our drug candidates or successfully commercializing them.
We face additional risks in connection with our existing and future collaborations, including the following:
| |
• | our collaborators may develop and commercialize, either alone or with others, products that are similar to or competitive with the products that are the subject of the collaboration with us; |
| |
• | our collaborators may underfund, not commit sufficient resources to, or conduct in an unsatisfactory manner the development, testing, marketing, distribution or sale of our drug candidates; |
| |
• | our collaborators may not properly maintain or defend our intellectual property rights or they may utilize our proprietary information in such a way as to invite litigation that could jeopardize or potentially invalidate our intellectual property or proprietary information or expose us to potential liability; |
| |
• | our collaborators may encounter conflicts of interest, changes in business strategy or other business issues which could adversely affect their willingness or ability to fulfill their obligations to us (for example, pharmaceutical and biotechnology companies historically have re-evaluated their priorities following mergers and consolidations, which have been common in recent years in these industries); |
| |
• | disputes may arise between us and our collaborators delaying or terminating the research, development, manufacture or commercialization of our drug candidates, resulting in significant litigation or arbitration that could be time-consuming and expensive, or causing collaborators to act in their own self-interest and not in the interest of our stockholders; |
| |
• | we might not have the financial or human resources to meet our obligations or take advantage of our rights under the terms of our existing and future collaborations; and |
| |
• | our existing collaborators may exercise their respective rights to terminate without cause their collaborations with us, in which event, we might not be able to complete development and commercialization of our drug candidates on our own. |
Our collaborative relationships may not produce the financial benefits that we are anticipating, which could cause our business to suffer.
Part of our strategy is to partner with, or out-license selective products to, other pharmaceutical companies in order to mitigate the cost of developing a drug through clinical trials to commercialization. Our option agreement with ASKA Pharmaceutical Co., Ltd., or Aska, is an example of this strategy. Following an analysis period after the completion of the planned Phase IIa study conducted by Pieris, Aska may exercise its option to obtain an exclusive license to develop and commercialize PRS-080 in Japan, South Korea and certain other Asian markets. Should Aska exercise the option, Pieris would be eligible for more than $80 million in combined option exercise fee and milestones associated with development and commercialization of PRS-080 in the first indication in Japan. We may receive further development milestones in additional indications, as well as in other countries within the Aska territory. Even if we successfully complete our Phase IIa study and such study yields favorable results, we cannot guarantee that Aska will exercise its option with respect to PRS-080. If Aska chooses not to exercise its option, we may or may not continue to develop PRS-080 on our own, but the post-option exercise developmental and sales milestone payments described in the agreement, plus additional royalty revenues, will never be realized. Alternatively, if Aska chooses not to exercise its option, we may choose not to continue with the PRS-080 program. If our collaboration with Aska or other similar partners is not successful, and if we cannot earn revenue from collaborative arrangements such as this agreement, our future revenues and business will be harmed.
We may not receive any further milestone, royalty or license payments under our current collaborations.
Although we have received upfront, milestone and other payments to date under our current drug development collaborations, we may not receive any royalty payments or additional license and milestone fees under such agreements. In general, our receipt of milestone, royalty or license payments depends on many factors, including whether our collaborators want and are able to continue to pursue potential drug candidates, intellectual property issues, unforeseen complications in the development or commercialization process, and the ultimate commercial success of the drugs.
Risks Related to the Commercialization of Our Drug Candidates
Even if we receive regulatory approval for any of our drug candidates, we will be subject to ongoing regulatory obligations and review. Maintaining compliance with ongoing regulatory requirements may result in significant additional expense to us, and any failure to maintain such compliance could subject us to penalties and cause our business to suffer.
Any regulatory approvals that we receive for our drug candidates may be subject to limitations on the approved indicated uses for which the products may be marketed, or contain requirements for potentially costly post-marketing testing, including Phase
IV clinical trials. In addition, if the FDA, EMA or a comparable foreign regulatory authority approves any of our drug candidates, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion and record keeping for the products will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with cGMP. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
| |
• | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| |
• | fines or warning letters; |
| |
• | refusal of the FDA or other applicable regulatory authority to approve pending applications or supplements to approved applications; |
| |
• | product seizure or detention, or refusal to permit the import or export of products; and |
| |
• | consent decrees, injunctions or the imposition of civil or criminal penalties. |
In addition, regulatory authorities’ policies (such as those of the FDA or EMA) may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our drug candidates. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are otherwise not able to maintain regulatory compliance, we may lose any marketing approval that we may have obtained, which would adversely affect our business, prospects and ability to achieve or sustain profitability.
Our commercial success depends upon attaining significant market acceptance of our drug candidates, if approved, among physicians, patients, healthcare payors and other members of the medical community.
Even if we obtain regulatory approval for our drug candidates, the products may not gain market acceptance among physicians, health care payors, patients and other members of the medical community, which is critical to commercial success. Market acceptance of any drug candidate for which we receive approval depends on a number of factors, including:
| |
• | perceptions by the medical community, physicians, and patients, regarding the safety and effectiveness of our products; |
| |
• | the size of the markets for the drug candidate, based on the size of the patient subsets that we are targeting, in the territories for which we gain regulatory approval and have commercial rights; |
| |
• | the potential and perceived advantages of the drug candidate over alternative treatments; |
| |
• | the safety of the drug candidate as demonstrated through broad commercial distribution; |
| |
• | the availability of adequate reimbursement and pricing for our products from governmental health programs and other third-party payors; |
| |
• | relative convenience and ease of administration; |
| |
• | cost-effectiveness of our product relative to competing products; |
| |
• | the prevalence and severity of adverse effects; and |
| |
• | the effectiveness of sales, marketing and distribution efforts by us and our licensees and distributors, if any. |
If our drug candidates are approved but fail to achieve an adequate level of acceptance by key market participants, we will not be able to generate significant revenues, and we may not become or remain profitable, which may require us to seek additional financing.
Our product candidates have never been manufactured on a commercial scale, and there are risks associated with scaling up manufacturing to commercial scale.
Our product candidates have never been manufactured on a commercial scale, and there are risks associated with scaling up manufacturing to commercial scale including, among others, cost overruns, potential problems with process scale-up, process reproducibility, stability issues, lot consistency and timely availability of raw materials. There is no assurance that our manufacturers will be successful in establishing a larger-scale commercial manufacturing process for PRS-343, PRS-060, PRS-080 or other product candidates that achieves our objectives for manufacturing capacity and cost of goods. Even if we could otherwise obtain regulatory approval for any product candidate, there is no assurance that our manufacturers will be able to manufacture the approved product to specifications acceptable to the FDA or other regulatory authorities, to produce it in sufficient quantities to meet the requirements for the potential launch of the product or to meet potential future demand. If our manufacturers are unable to produce sufficient quantities of the approved product for commercialization, our commercialization efforts would be impaired, which would have an adverse effect on our business, financial condition, results of operations and growth prospects.
Reimbursement may be limited or unavailable in certain market segments for our drug candidates, which could make it difficult for us to sell on a profitable basis any products for which we obtain marketing approvals.
There is significant uncertainty related to the third-party coverage and reimbursement of newly approved drugs. Market acceptance and successful commercialization of any of our drug candidates that obtain regulatory approval in domestic or international markets will depend significantly on the availability of adequate coverage and reimbursement from governmental authorities, private health insurers, and other third-party payors for any of our drug candidates, and may be affected by existing and future healthcare reform measures.
Pricing and reimbursement for any of our drug candidates that obtain regulatory approval is uncertain. Government authorities, private health insurers and other third-party payors decide which drugs they will cover and establish reimbursement levels for them, and obtaining coverage and reimbursement approval for a product from any such third-party payors is a time consuming and costly process. Third-party payers are also increasingly challenging the effectiveness of and prices charged for medical products and services. As a result, any denial of private or government payor coverage or inadequate reimbursement for our drug candidates, if any are commercialized, could harm our business and reduce our prospects for generating revenue.
Further, there have been, and may continue to be, legislative and regulatory proposals at the U.S. federal and state levels and in foreign jurisdictions directed at broadening the availability and containing or lowering the cost of healthcare. Existing legislation aimed at patient affordability in the U.S. may be repealed or replaced. The continuing efforts of the government, insurance companies, managed care organizations and other third-party payors to contain or reduce costs of healthcare may adversely affect our ability to set prices for our products that would allow us to achieve or sustain profitability. In addition, governments may impose price controls on any of our products that obtain marketing approval, which may adversely affect our future profitability.
In some foreign countries, particularly in the European Union, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can be a long and expensive process after the receipt of marketing approval for a drug candidate. To obtain reimbursement or pricing approval in some countries, we may be required to conduct additional clinical trials that compare the cost-effectiveness of our drug candidates to other available therapies. If reimbursement of our drug candidates is unavailable or limited in scope or amount in a particular country, or if pricing is set at unsatisfactory levels, we may be unable to achieve or sustain profitability for sales of any of our drug candidates that are approved for marketing in that country.
We have no experience selling, marketing or distributing products and currently have no internal marketing and sales force. If we are unable to establish effective marketing and sales capabilities or enter into agreements with third parties to market and sell our drug candidates, we may not be able to effectively market and sell our drug candidates, if approved, or generate product revenues.
We currently have no sales, marketing or distribution capabilities and there can be no assurance that we will be able to market and sell our products in the United States or overseas. In order to commercialize any drug candidates, we must build on a territory-by-territory basis marketing, sales, distribution, managerial and other non-technical capabilities or make arrangements with third parties to perform these services, and we may not be successful in doing so. Therefore, with respect to the commercialization of all or certain of our drug candidates, we may choose to collaborate, either globally or on a territory-by-territory basis, with third parties that have direct sales forces and established distribution systems, either to augment our own sales force and distribution systems or in lieu of our own sales force and distribution systems. If so, our success will depend, in
part, on our ability to enter into and maintain collaborative relationships for such capabilities, such collaborator’s strategic interest in the products under development and such collaborator’s ability to successfully market and sell any such products.
If we are unable to enter into such arrangements when needed on acceptable terms or at all, we may not be able to successfully commercialize any of our drug candidates that receive regulatory approval or any such commercialization may experience delays or limitations. Further, to the extent that we depend on third parties for marketing and distribution, any revenues we receive will depend upon the efforts of such third parties, and there can be no assurance that such efforts will be successful.
To the extent that we decide not to, or are unable to, enter into collaborative arrangements with respect to the sales and marketing of our products, we may in the future need to establish an internal sales and marketing team with technical expertise and supporting distribution capabilities to commercialize our drug candidates, which could be expensive and time consuming and which would require significant attention of our executive officers to manage. Further, may not have sufficient resources to allocate to the sales and marketing of our products.
Any failure or delay in the development of sales, marketing and distribution capabilities, through collaboration with one or more third parties or through internal efforts, would adversely impact the commercialization of any of our products that we obtain approval to market. As a result, our future product revenue will suffer and we may incur significant additional losses.
We face significant competition from other biotechnology and pharmaceutical companies, and our operating results will suffer if we fail to compete effectively.
The biotechnology and pharmaceutical industries are intensely competitive and subject to rapid and significant technological advances. In addition, the competition in the anemia, asthma and cancer markets is intense. We have competitors both in the United States and internationally, including major multinational pharmaceutical companies, fully integrated pharmaceutical and biotechnology companies, smaller companies that are collaborating with larger pharmaceutical companies, academic institutions, and other public and private research organizations.
There are several third-party drug candidates that could be competitive with drug candidates in our pipeline.
Drug candidates interfering with hepcidin function and thus competing with PRS-080 include those that are being developed by Lilly (LY-2928057), Ferrumax (FMX-8) and Ionis/Xenon (XEN701). In addition, drug candidates being develop for anemia associated with chronic kidney disease targeting other pathways than hepcidin also competes with PRS-080, this include HIF-PH inhibitors such as FibroGen/AstraZeneca/Astella’s Roxadustat and Akebia/Mitsubishi/Otsuka’s Vadadustat. Drug candidates interfering with the function of type 2 helper T cells, or Th2, the biological pathway for PRS-060, and thus competing with PRS-060, include those that are being developed by Sanofi/Regnereron (dupilumab), GSK (mepolizumab/Nucala), Teva (reslizumab/Cinqair) and Amgen/AstraZeneca (tezepelumab). Drugs targeting immunomodulatory targets and thus competing with our 300-Series programs include those that are currently marketed by Bristol-Myers Squibb (Yervoy/ipilimumab, Opdivo/nivolumab), Merck & Co (Keytruda/pembrolizumab), Roche (Tecentriq/atezolizumab), Merck Serono/Pfizer (Bavencio/Avelumab) and AstraZeneca (Imfinzi/durvalumab) and drug candidates being developed by Bristol-Myers Squibb (e.g. urelumab/anti-CD137 and relatlimab/anti-LAG3), Pfizer (e.g. utomilumab /anti-CD137 and PF-04518600/anti-OX40) and others clinical stage drug candidates also competes with our 300-Series. Additionally, a number of other companies, such as Amgen, Affimed, Macrogenics, F-Star Molecular Partners, Xencor, Immunocore and Zymeworks, also pursue multispecific approaches in oncology, which therapies are in clinical or preclinical development. For additional information about our third-party drug candidates which could be competitive with the drug candidates in our pipeline, see “Business-Competition.”
These existing or future competing products may provide therapeutic convenience or clinical or other benefits for a specific indication greater than our products, or may offer comparable performance at a lower cost. If our products fail to capture and maintain market share, we may not achieve sufficient product revenue and our business will suffer.
Many of our competitors have substantially greater financial, technical and other resources than we do, such as larger research and development staff and experienced marketing and manufacturing organizations, as well as significantly greater experience in:
| |
• | undertaking preclinical testing and clinical trials; |
| |
• | obtaining FDA and other regulatory approvals of drugs; |
| |
• | prosecuting and enforcing intellectual property rights; |
| |
• | formulating and manufacturing drugs; and |
| |
• | launching, marketing and selling drugs. |
Established pharmaceutical companies may invest heavily to accelerate discovery and development of or in-license novel compounds that could make our drug candidates less competitive. In addition, any new product that competes with an approved product must demonstrate compelling advantages in efficacy, convenience, tolerability and safety in order to overcome price competition and to be commercially successful. Accordingly, our competitors may succeed in obtaining patent protection, receiving FDA, EMA or other regulatory approval, or discovering, developing and commercializing medicines before we do, which would have a material adverse effect on our business and ability to achieve profitability from future sales of our approved drug candidates, if any. For additional information about our competitors, please see “Business—Competition.”
We could be subject to product liability lawsuits based on the use of our drug candidates in clinical testing or, if obtained, following marketing approval and commercialization. If product liability lawsuits are brought against us, we may incur substantial liabilities and may be required to cease clinical testing or limit commercialization of our drug candidates.
We could be subject to product liability lawsuits if any drug candidate we develop allegedly causes injury or is found to be otherwise unsuitable for human use during product testing, manufacturing, marketing or sale. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, negligence, strict liability and a breach of warranties. Claims could also be asserted under state consumer protection acts.
Our inability to obtain and retain sufficient product liability insurance at an acceptable cost to protect against potential product liability claims could prevent or inhibit the clinical testing and commercialization of products we develop on our own or with collaborators. We do not currently carry general product liability insurance. We have put in place several applicable product liability insurance covering us as sponsor and the investigators involved in clinical trials. For our PRS-080 Phase IIa study in healthy volunteers we have one liability insurance in Germany, in an amount of up to the lesser of $0.6 million per enrolled subject or $12.0 million for the Phase IIa clinical trial in its entirety and one liability insurance in the Czech Republic, in an amount of $0.3 million per enrolled subject or $2.8 million for the Phase IIa clinical trial in its entirety. For our PRS-060 Phase Ia study in healthy volunteers we have one liability insurance in an amount of up to AUD 20.0 million ($15.6 million) per enrolled subject and in the annual aggregate. For our PRS-343 Phase Ia study in healthy volunteers we have one liability insurance in an amount of up to $3.0 million per enrolled subject and in the annual aggregate. In the future, we will seek to obtain appropriate insurance coverage with respect to any future clinical trials of our other drug candidates, but we may not be able to obtain the levels of coverage desired on acceptable terms, or at all. If we do secure product liability insurance, we may subsequently determine that additional amounts of coverage would be desirable at later stages of clinical development of our drug candidates or upon commencing commercialization of any drug candidate that obtains required approvals, but we may not be able to obtain such additional coverage amounts when needed on acceptable terms, or at all. Unless and until we obtain such insurance, we would be solely responsible for any product liability claims relating to our preclinical and clinical development activities. Further, even after any such insurance coverage is obtained, any claim that may be brought against us could result in a court judgment or settlement in an amount that is not covered, in whole or in part, by any insurance policies we may then have or that is in excess of the limits of our insurance coverage. We would be required to pay any amounts awarded by a court or negotiated in a settlement that exceed the coverage limitations or that are not covered by any product liability insurance we may obtain, and we may not have, or be able to obtain, sufficient capital to pay such amounts.
Risks Related to Our Intellectual Property
If we breach any of the agreements under which we license from third parties the intellectual property rights or commercialization rights to our drug candidates, particularly our license agreements with TUM and Enumeral, we could lose license rights that are important to our business and our operations could be materially harmed.
Under the TUM License Agreement, we in-license significant intellectual property related to our Anticalin® platforms from Technische Universität München, or TUM. Under the terms of the agreement, TUM assigns to us certain materials and records resulting from the research. We retain rights to inventions made by our employees, and TUM assigns to us all inventions made under the agreement jointly by our employees and TUM personnel, provided that our employees have made a certain inventive contribution. With respect to all other inventions made in the course of the research, TUM grants to us worldwide exclusive license rights under patents and patent applications claiming such inventions. TUM retains rights to practice these inventions
for research and teaching purposes. We bear the costs of filing, prosecution and maintenance of patents assigned or licensed to us under the agreement.
As consideration for the assignments and licenses, we are obliged to pay to TUM milestone payments on development of our proprietary products claimed by patents assigned or licensed to us by TUM. We also are obliged to pay low single-digit royalties, including annual minimum royalties, on sales of such products. Should we grant licenses or sublicenses to those patents to third parties, we are obliged to pay to TUM certain undisclosed fees as a function of out-licensing revenues in connection with those patents, or Out-License Fees, where such Out-License Fees are creditable against annual license payments to TUM. Our payment obligations are reduced by our proportionate contribution to a joint invention. Payment obligations terminate on expiration or annulment of the last patent covered by the TUM License Agreement that covers a proprietary product or is sublicensed, as applicable.
Under the Definitive License and Transfer Agreement with Enumeral Biomedical Holdings, Inc. or Enumeral, we in-licensed intellectual property related to an Enumeral-generated antibody against PD-1 and an option to in-license up to two additional antibodies against undisclosed targets. Under the terms of the agreement, we acquired a non-exclusive worldwide license under the applicable Enumeral patents and know-how to research, develop and commercialize fusion proteins incorporating Enumeral’s PD-1 antibody and one or more Anticalin proteins.
As consideration, we are obliged to pay to Enumeral development and sales milestones on development of products incorporating the Enumeral antibody. We are also obliged to pay low to lower-middle single-digit royalties as a percentage of net sales depending on the amount of net sales in the applicable years. In the event that we are required to pay a license fee or royalty to any third party related to the licensed products, our royalty payment obligations to Enumeral are reduced by the amount of such third-party fees or payments, up to 50% of the royalty payment for each calendar year due to Enumeral. Payment obligations terminate on a product-by-product and country-by-country basis on the later of ten years from the first commercial sale of a product incorporating the Enumeral antibody or the last to expire, lapse or be abandoned of a claim from the licensed Enumeral patents filed as of the effective date of the agreement that cover the manufacture, use, offer for sale, sale or import of a product incorporating the Enumeral antibody.
In addition to the TUM License Agreement and the Definitive License and Transfer Agreement with Enumeral, or the Enumeral License Agreement, we have other in-license agreements (such as with Kelun) and may seek to enter into additional agreements with other third parties in the future granting similar license rights with respect to other potential drug candidates. If we fail to comply with any of the conditions or obligations or otherwise breach the terms of the TUM License Agreement or the Enumeral License Agreement, the Kelun License Agreement, or any future license agreement we may enter on which our business or drug candidates are dependent, TUM or Enumeral or other licensors may have the right to terminate the applicable agreement in whole or in part and thereby extinguish our rights to the licensed technology and intellectual property and/or any rights we have acquired to develop and commercialize certain drug candidates, including, with respect to the TUM License Agreement and Enumeral License Agreement, our Anticalin drug therapies. Under the TUM License Agreement, we can terminate the licenses to any or all licensed patents upon specified advance notice to TUM. TUM may terminate the license provisions of the agreement only for cause. Termination of the agreement does not terminate our rights in patents assigned to us but would terminate our rights to patents licensed to us under the agreement. Under the Enumeral License Agreement, we can terminate the agreement upon 30 days’ notice to Enumeral. Enumeral may terminate the Enumeral License Agreement only upon a material breach by us that is not cured. The loss of the rights licensed to us under our license agreement with TUM or Enumeral, or any future license agreement that we may enter granting us rights on which our business or drug candidates are dependent, would eliminate our ability to further develop the applicable drug candidates and would materially harm our business, prospects, financial condition and results of operations.
Risks Related to Managing Any Growth We May Experience
We will need to grow the size of our organization, and we may not successfully manage any growth we may achieve.
Our success will depend upon the expansion of our operations and our ability to successfully manage our growth. Our future growth, if any, may place a significant strain on our management and on our administrative, operational, and financial resources requiring us to implement and improve our operational, financial, and management systems.
In addition, our ability to manage our growth effectively will hinge upon our ability to expand, train, manage, and motivate our employees. As of December 31, 2017, we have 68 full-time employees. As our development and commercialization plans and strategies develop, these demands may also require the hiring of additional research, development, managerial, operational, sales, marketing, financial, accounting, legal, and other personnel.
Moreover, future growth could require the development of additional expertise by management and impose significant added responsibilities on members of management, including:
| |
• | effectively managing our clinical trials and submissions to regulatory authorities for marketing approvals; |
| |
• | effectively managing our internal research and development efforts such as discovery research and preclinical development; |
| |
• | identifying, recruiting, maintaining, motivating and integrating additional employees; |
| |
• | effectively managing our internal and external business development efforts with current or future partners, such as entering into additional collaboration arrangements and increasing out-licensing revenues; |
| |
• | establishing relationships with third parties essential to our business and ensuring compliance with our contractual obligations to such third parties; |
| |
• | developing and managing new divisions of our internal business, including any sales and marketing segment we elect to establish; |
| |
• | maintaining our compliance with public company reporting and other obligations, including establishing and maintaining effective internal control over financial reporting and disclosure controls and procedures; and |
| |
• | improving our managerial, development, operational and finance systems. |
We may not be able to accomplish any of those tasks, and our failure to do so could prevent us from effectively managing future growth, if any, and successfully growing our company.
Any increase in resources devoted to research and product development without a corresponding increase in our operational, financial, and management systems could have a material adverse effect on our business, financial condition and results of operations.
We may make future acquisitions that could disrupt our business, cause dilution to our stockholders and harm our financial condition and operating results.
We may, in the future, make acquisitions of, or investments in, companies that we believe have products or capabilities that are a strategic or commercial fit with our current business or otherwise offer opportunities for our company. In connection with these acquisitions or investments, we may:
| |
• | issue common stock or other forms of equity that would dilute our existing stockholders’ percentage of ownership; |
| |
• | incur debt and assume liabilities; and |
| |
• | incur amortization expenses related to intangible assets or incur large and immediate write-offs. |
We may not be able to complete acquisitions on favorable terms, if at all. If we do complete an acquisition, we cannot assure you that it will ultimately strengthen our competitive position or that it will be viewed positively by customers, financial markets or investors. Furthermore, future acquisitions could pose numerous additional risks to our operations, including:
| |
• | problems integrating the purchased business, products or technologies; |
| |
• | challenges in achieving strategic objectives, cost savings and other anticipated benefits; |
| |
• | increases to our expenses; |
| |
• | the assumption of significant liabilities that exceed the limitations of any applicable indemnification provisions or the financial resources of any indemnifying party; |
| |
• | inability to maintain relationships with key customers, vendors and other business partners of the acquired businesses; |
| |
• | diversion of management’s attention from their day-to-day responsibilities; |
| |
• | difficulty in maintaining controls, procedures and policies during the transition and integration; |
| |
• | entrance into marketplaces where we have no or limited prior experience and where competitors have stronger marketplace positions; |
| |
• | potential loss of key employees, particularly those of the acquired entity; and |
| |
• | that historical financial information may not be representative or indicative of our results as a combined company. |
If our efforts to protect the proprietary nature of the intellectual property related to our technologies are not adequate, we may not be able to compete effectively and our business would be harmed.
We rely upon a combination of patents, trade secret protection and confidentiality agreements to protect the intellectual property related to our technologies. Any disclosure to, or misappropriation by, third parties of our proprietary information could enable competitors to quickly duplicate or surpass our technological achievements, thus eroding any competitive advantage we may derive from the proprietary information.
The strength of patents in the biotechnology and pharmaceutical fields can be uncertain and involve complex legal and scientific questions. No consistent policy regarding the breadth of claims allowed in patents has emerged to date in the United States. Accordingly, we cannot predict the breadth of claims that may be allowed or enforced, or that the scope of any patent rights could provide a sufficient degree of protection that could permit us to gain or keep our competitive advantage with respect to these products and technologies. For example, we cannot predict:
| |
• | the degree and range of protection any patents will afford us against competitors, including whether third parties will find ways to make, use, sell, offer to sell or import competitive products without infringing our patents; |
| |
• | if and when patents will be issued; |
| |
• | whether or not others will obtain patents claiming inventions similar to those covered by our patents and patent applications; or |
| |
• | whether we will need to initiate litigation or administrative proceedings (e.g. at the United States Patent and Trademark Office, or the USPTO, or the European Patent Office, or the EPO) in connection with patent rights, which may be costly whether we win or lose. |
As a result, the patent applications we own or license may fail to result in issued patents in the United States or in foreign countries. Third parties may challenge the validity, enforceability or scope of any issued patents we own or license or any applications that may issue as patents in the future, which may result in those patents being narrowed, invalidated or held unenforceable. Even if they are unchallenged, our patents and patent applications may not adequately protect our intellectual property or prevent others from developing similar products that do not fall within the scope of our patents. If the breadth or strength of protection provided by the patents we hold or pursue is threatened, our ability to commercialize any drug candidates with technology protected by those patents could be threatened. Further, if we encounter delays in our clinical trials, the period of time during which we would have patent protection for any covered drug candidates that obtain regulatory approval would be reduced. Since patent applications in the United States and most other countries are confidential for a period of time after filing, we cannot be certain at the time of filing that we are the first to file any patent application related to our drug candidates.
While patent term extensions under the Hatch-Waxman Act in the United States and under supplementary protection certificates in Europe may be available to extend our patent exclusivity for our drug candidates, the applicable patents may not meet the specified conditions for eligibility for any such term extension and, even if eligible, we may not be able to obtain any such term extension. Further, because filing, prosecuting, defending and enforcing patents in multiple jurisdictions can be expensive, we may elect to pursue patent protection relating to our drug candidates in only certain jurisdictions. As a result, competitors would be permitted to use our technologies in jurisdictions where we have not obtained patent protection to develop their own products, any of which could compete with our drug candidates.
In addition to the protection afforded by patents, we seek to rely on trade secret protection and confidentiality agreements to protect proprietary know-how that is not patentable, processes for which patents are difficult to enforce and any other elements of our discovery platform and drug development processes that involve proprietary know-how, information or technology that is not covered by patents or not amenable to patent protection. Although we require all of our employees and certain
consultants and advisors to assign inventions to us, and require all of our employees, consultants, advisors and any third parties who have access to our proprietary know-how, information or technology to enter into confidentiality agreements, our trade secrets and other proprietary information may be disclosed or competitors may otherwise gain access to such information or independently develop substantially equivalent information. Further, the laws of some foreign countries do not protect proprietary rights to the same extent or in the same manner as the laws of the United States. As a result, we may encounter significant difficulty in protecting and defending our intellectual property both in the United States and abroad. If we are unable to prevent material disclosure of the trade secrets and other intellectual property related to our technologies to third parties, we may not be able to establish or maintain the competitive advantage that we believe is provided by such intellectual property, which could adversely affect our market position and business and operational results.
Claims that we infringe the intellectual property rights of others may prevent or delay our drug discovery and development efforts.
Our research, development and commercialization activities, as well as any drug candidates or products resulting from those activities, may infringe or be accused of infringing a patent or other form of intellectual property under which we do not hold a license or other rights. Third parties may assert that we are employing their proprietary technology without authorization.
There may be third-party patents of which we are currently unaware with claims that cover the use or manufacture of our drug candidates or the practice of our related methods. Because patent applications can take many years to issue, there may be currently pending patent applications that may later result in issued patents that our drug candidates may infringe. In addition, third parties may obtain patents in the future and claim that use of our drug candidates infringes upon one or more claims of these patents. If our activities or drug candidates infringe the patents or other intellectual property rights of third parties, the holders of such intellectual property rights may be able to block our ability to commercialize such drug candidates or practice our methods unless we obtain a license under the intellectual property rights or until any applicable patents expire or are determined to be invalid or unenforceable.
Defense of any intellectual property infringement claims against us, regardless of their merit, would involve substantial litigation expense and would be a significant diversion of resources from our business. In the event of a successful claim of infringement against us, we may have to pay substantial damages, obtain one or more licenses from third parties, limit our business to avoid the infringing activities, pay royalties and/or redesign our infringing drug candidates or alter related formulations, processes, methods or other technologies, any or all of which may be impossible or require substantial time and monetary expenditure. Further, if we were to seek a license from the third-party holder of any applicable intellectual property rights, we may not be able to obtain the applicable license rights when needed or on reasonable terms, or at all. Some of our competitors may be able to sustain the costs of complex patent litigation or proceeding more effectively than us due to their substantially greater resources. The occurrence of any of the above events could prevent us from continuing to develop and commercialize one or more of our drug candidates and our business could materially suffer.
We may desire to, or be forced to, seek additional licenses to use intellectual property owned by third parties, and such licenses may not be available on commercially reasonable terms or at all.
Third parties may also hold intellectual property, including patent rights that are important or necessary to the development of our drug candidates, in which case we would need to obtain a license from that third party or develop a different formulation of the product that does not infringe upon the applicable intellectual property, which may not be possible. Additionally, we may identify drug candidates that we believe are promising and whose development and other intellectual property rights are held by third parties. In such a case, we may desire to seek a license to pursue the development of those drug candidates. Any license that we may desire to obtain or that we may be forced to pursue may not be available when needed on commercially reasonable terms or at all. Any inability to secure a license that we need or desire could have a material adverse effect on our business, financial condition and prospects.
The patent protection covering some of our drug candidates may be dependent on third parties, who may not effectively maintain that protection.
While we expect the right to fully prosecute any patents covering drug candidates we may in-license from third-party owners, there may be instances when the prosecution and maintenance of issued patents and pending patent applications that cover our drug candidates remain controlled by our licensors. Similarly, some of our future licensing partners may retain the right, or may seek the rights, to prosecute patents covering the drug candidates we license to them and we may grant such rights to those partners for business reasons. If such third parties fail to appropriately maintain that patent protection, we may not be able to prevent competitors from developing and selling competing products or practicing competing methods and our ability to generate revenue from any commercialization of the affected drug candidates may suffer.
Certain technologies and patents have been developed with partners and we may face restrictions on this jointly developed intellectual property.
We have entered into agreements with a number of commercial partners, including university partners, which cover intellectual property. We have, in some cases individually and in other cases along with our partners, filed for patent protection for a number of technologies developed under these agreements and may in the future file for further intellectual property protection and/or seek to commercialize such technologies. Under some of these agreements, certain intellectual property developed by us and the relevant partner may be subject to joint ownership by us and the partner and our commercial use of such intellectual property may be restricted, or may require written consent from, or a separate agreement with, the partner. In other cases, we may not have any rights to use intellectual property solely developed and owned by the partner. If we cannot obtain commercial use rights for such jointly owned intellectual property or partner-owned intellectual property, our future product development and commercialization plans may be adversely affected.
We may be involved in lawsuits to protect or enforce our patents or the patents of our licensors, which could be expensive, time-consuming and unsuccessful.
Competitors may infringe our patents or the patents of our current or potential licensors. To attempt to stop infringement or unauthorized use, we may need to enforce one or more of our patents, which can be expensive and time-consuming and distract management. If we pursue any litigation, a court may decide that a patent of ours or any of our licensors’ is not valid or is unenforceable, or may refuse to stop the other party from using the relevant technology on the grounds that our patents do not cover the technology in question. Further, the legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents, which could reduce the likelihood of success of, or the amount of damages that could be awarded resulting from, any infringement proceeding we pursue in any such jurisdiction. An adverse result in any infringement litigation or defense proceedings could put one or more of our patents at risk of being invalidated, held unenforceable, or interpreted narrowly and could put our patent applications at risk of not issuing, which could limit our ability to exclude competitors from directly competing with us in those jurisdictions.
Interference proceedings provoked by third parties or brought by the USPTO or at its foreign counterparts (such as the EPO) to determine the priority of inventions with respect to our patents or patent applications or those of our licensors. An unfavorable outcome could require us to cease using the related technology or to attempt to license rights to use it from the prevailing party. Our business could be harmed if the prevailing party does not offer us a license on commercially reasonable terms, or at all.
Litigation or interference proceedings may fail and, even if successful, may result in substantial costs and distract our management and other employees.
If we are unsuccessful in obtaining or maintaining patent protection for intellectual property in development, our business and competitive position would be harmed.
We are seeking patent protection of our technology and for our drug candidates. Patent prosecution is a challenging process and is not assured of success. If we are unable to secure patent protection for our technology and drug candidates, our business may be adversely impacted.
In addition, issued patents and pending applications require regular maintenance. Failure to maintain our portfolio may result in loss of rights that may adversely impact our intellectual property rights, for example by rendering issued patents unenforceable or by prematurely terminating pending applications.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to seeking patents for our Anticalin-brand technology and some of our drug candidates, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We currently, and expect in the future to continue to, seek to protect these trade secrets, in part by entering into confidentiality agreements with parties who have access to them, such as our employees, collaborators, contract manufacturers, consultants, advisors, investigators and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for any such disclosure. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them, or those to whom they disclose the trade secrets, from using that
technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor, our competitive position would be harmed.
If we fail to protect our trademark rights, competitors may be able to take advantage of our goodwill, which would weaken our competitive position, reduce our revenues and increase our costs.
We believe that the protection of our trademark rights is an important factor in product recognition, maintaining goodwill, and maintaining or increasing market share. We may expend substantial cost and effort in an attempt to register, maintain and enforce our trademark rights. If we do not adequately protect our rights in our trademarks from infringement, any goodwill that we have developed in those trademarks could be lost or impaired.
Third parties may claim that the sale or promotion of our products, when and if we have any, may infringe on the trademark rights of others. Trademark infringement problems occur frequently in connection with the sale and marketing of pharmaceutical products. If we become involved in any dispute regarding our trademark rights, regardless of whether we prevail, we could be required to engage in costly, distracting and time-consuming litigation that could harm our business. If the trademarks we use are found to infringe upon the trademark of another company, we could be liable for damages and be forced to stop using those trademarks, and as result, we could lose all the goodwill that has been developed in those trademarks.
Certain of our employees and their inventions are subject to German law.
Many of our employees work in Germany and are subject to German employment law. Ideas, developments, discoveries and inventions made by such employees and consultants are subject to the provisions of the German Act on Employees’ Inventions (Gesetz über Arbeitnehmererfindungen), which regulates the ownership of, and compensation for, inventions made by employees. We face the risk that disputes can occur between us and such employees or ex-employees pertaining to alleged non-adherence to the provisions of this act that may be costly to defend and take up our management’s time and efforts whether we prevail or fail in such dispute. In addition, under the German Act on Employees’ Inventions, certain employees retained rights to patents they invented or co-invented prior to 2009. Although most of these employees have subsequently assigned their interest in these patents to us, there is a risk that the compensation we provide to them may be deemed insufficient and we may be required under German law to increase the compensation due to such employees for the use of the patents. In those cases where employees have not assigned their interests to us, we may need to pay compensation for the use of those patents. If we are required to pay additional compensation or face other disputes under the German Act on Employees’ Inventions, our results of operations could be adversely affected.
The future growth of our business may expose our intellectual property to a high risk of counterfeiting or unauthorized use.
As part of our business strategy, we intend to license our Anticalin technology and sell our potential products, if any, in many different countries. As a result, we may do business with third parties in countries where intellectual property rights have been or are routinely disregarded, and the future growth of our business may expose our intellectual property to a high risk of counterfeiting or unauthorized use. Although we attempt to obtain broad international intellectual property rights for our Anticalin technology and proteins, we cannot guarantee that such rights, to the extent we can obtain them, will be enforceable in a timely fashion or at all in any particular country or jurisdiction, or that if enforced, will offer us adequate commercial protection or adequate redress for any harm suffered. Counterfeiting or unauthorized use of our technologies or products may also expose our business to harm for which no adequate monetary redress exists, and to the extent we are unable to stop such use, may cause us to lose rights with respect to intellectual property that is crucial to our business. Any such misuse of our intellectual property may have a substantial negative impact on our business and revenues, and may cause our business to fail.
Risks Related to our Employees
If we are not able to attract and retain highly qualified personnel, we may not be able to successfully implement our business strategy.
Our ability to compete in the highly competitive biotechnology and pharmaceuticals industries depends upon our ability to attract and retain highly qualified personnel. We are highly dependent on our management, scientific and medical personnel, especially Stephen S. Yoder, our Chief Executive Officer and President, whose services are critical to the successful implementation of our drug candidate development, our business development and partnerships, and our regulatory and commercialization strategies. Further, as our approach is built upon the drug discovery and development experience of our drug development team, which we believe is a significant contributor to our competitive advantage, we are dependent on the maintenance and growth of that team with qualified members containing high levels of expertise in specific scientific fields. We may in the future hire additional employees for research and development or general and administrative activities.
We are not aware of any present intention of any of our executive officers or other members of our senior management team to leave our company, but our industry tends to experience a high rate of turnover of management personnel and our employees are generally able to terminate their relationships with us on short notice. Pursuant to German employment law, our employment arrangements with employees of Pieris GmbH are governed by employment contracts, which provide certain defined terms for either party to terminate the employment relationship.
The loss of the services of any of our executive officers, in particular Mr. Yoder, or other key employees and our inability to find suitable replacements could potentially harm our business, financial condition and prospects. Our success also depends on our ability to continue to attract, retain and motivate highly skilled junior and mid-level managers as well as junior and mid-level scientific and medical personnel.
Moreover, there is intense competition for a limited number of qualified personnel among biopharmaceutical, biotechnology, pharmaceutical and other related businesses. Many of the other companies against which we compete for qualified personnel have greater financial and other resources, different risk profiles, longer histories in the industry and greater ability to provide valuable cash or stock incentives to potential recruits than we do. They also may provide more diverse opportunities and better chances for career advancement. Some of these characteristics may be more appealing to high quality candidates than what we are able to offer as an early stage company. If we are unable to continue to attract and retain high quality personnel, the rate and success at which we can develop and commercialize drug candidates will be limited.
We may be subject to labor claims brought by our employees against us.
In the United States, an employment relationship with no specified duration is presumed to be employment “at-will” and the employer or employee may terminate the employment relationship at any time, with or without cause, except for public policy reasons including discrimination, participating in union activity, or refusing to carry out an activity that violates the law.
In contrast, in Germany, there is no analogous doctrine of “employment at will”. By law, German employees must have written employment contracts that reflect the key aspects of the employment relationship. Our relations between German employers and employees are extensively regulated under German labor and employment laws and regulations. German employees have special protection against dismissals provided the employee has been employed by a company for more than six months and such company employs more than ten employees.
German employment termination law is regulated by various codes, in particular the Kündigungsschutzgesetz (German Termination Protection Act) and is intended to give the employee maximum protection against unfair dismissal, including among other things:
| |
• | the employer must observe the applicable notice period, which is ordinarily determined by law (between four weeks and seven months, depending upon the length of employment), if a longer period is not otherwise agreed by the parties, and has to deliver a written notice of termination to the employee; |
| |
• | for companies with more than ten employees, the German Termination Protection Act generally restricts termination of employment if the employee has been employed for more than six months, wherein the employee may be terminated only for a particular reason, including certain behavioral or personal reasons relating to the employee or certain developments relating to the business of the employer, such as a business restructuring which reduces the number of employee positions; |
| |
• | special termination protection against unlawful dismissal applies to several other groups of employees, such as an employee that is an officially acknowledged handicapped person, an employee who was appointed as a company’s data protection officer or as a member of the works council of a company, if any, an employee on three years’ maternity leave or a pregnant employee; in these cases, approval of various German authorities is required prior to termination but usually very difficult to obtain; and |
| |
• | if a company engages in a mass layoff, which is deemed to occur when the employer intends to dismiss a large percentage of its employees during a one-month period, prior written notification to the German employment office is required. |
In this regard, if we downsize for any reason and fail to adhere to the complex requirements articulated by the employee protection law, we could face legal actions brought by affected employees or former employees, and, as a result, we may incur operational or financial losses and the attention of our executive officers may be distracted from managing our business.
We may be subject to claims by third parties asserting that our employees or we have misappropriated their intellectual property, or claiming ownership of what we regard as our own intellectual property.
Many of our employees were previously employed at universities or other biotechnology or pharmaceutical companies, including our competitors or potential competitors. We may be subject to claims that these employees or we have used or disclosed intellectual property, including trade secrets or other proprietary information, of any such employee’s former employers. Litigation may be necessary to defend against any such claims.
In addition, while it is our policy to require our employees and contractors, who may be involved in the development of intellectual property, to execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact contributes to the development of intellectual property that we regard as our own. Further, the terms of such assignment agreements may be breached and we may not be able to successfully enforce their terms, which may force us to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of intellectual property rights we may regard and treat as our own.
Our employees may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could cause our business to suffer.
We are exposed to the risk of employee fraud or other misconduct. Misconduct by employees could include intentional failures to comply with FDA or EMA regulations, provide accurate information to the FDA or EMA, comply with manufacturing standards we have established, comply with federal, state and international healthcare fraud and abuse laws and regulations as they may become applicable to our operations, report financial information or data accurately or disclose unauthorized activities to us. Employee misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter employee misconduct, and the precautions and procedures we currently take or may establish in the future as our operations and employee base expand to detect and prevent this type of activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure by our employees to comply with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant fines or other sanctions.
Risks Related to the Ownership of our Common Stock
Our share price is expected to be volatile and may be influenced by numerous factors, some of which are beyond our control.
Market prices for shares of biotechnology companies such as ours are often volatile, and the quoted price of our common stock is therefore likely to be highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control. In addition to the factors discussed in this “Risk Factors” section and elsewhere in this Annual Report on Form 10-K, these factors include:
| |
• | the drug candidates we seek to pursue, and our ability to obtain rights to develop, commercialize and market those drug candidates; |
| |
• | our decision to initiate a clinical trial, not to initiate a clinical trial or to terminate an existing clinical trial; |
| |
• | actual or anticipated adverse results or delays in our clinical trials; |
| |
• | our failure to commercialize our drug candidates, if approved; |
| |
• | unanticipated serious safety concerns related to the use of any of our drug candidates; |
| |
• | adverse regulatory decisions; |
| |
• | additions or departures of key scientific or management personnel; |
| |
• | changes in laws or regulations applicable to our drug candidates, including without limitation clinical trial requirements for approvals; |
| |
• | disputes or other developments relating to patents and other proprietary rights and our ability to obtain patent protection for our drug candidates; |
| |
• | our dependence on third parties, including CROs and CMOs as well as our current and potential partners that produce companion diagnostic products; |
| |
• | failure to meet or exceed any financial guidance or expectations regarding development milestones that we may provide to the public; |
| |
• | actual or anticipated variations in quarterly operating results; |
| |
• | failure to meet or exceed the estimates and projections of the investment community; |
| |
• | overall performance of the equity markets and other factors that may be unrelated to our operating performance or the operating performance of our competitors, including changes in market valuations of similar companies; |
| |
• | conditions or trends in the biotechnology and biopharmaceutical industries; |
| |
• | introduction of new products offered by us or our competitors; |
| |
• | announcements of significant acquisitions, strategic partnerships, joint ventures or capital commitments by us or our competitors; |
| |
• | our ability to maintain an adequate rate of growth and manage such growth; |
| |
• | issuances of debt or equity securities; |
| |
• | sales of our common stock by us or our stockholders in the future, or the perception that such sales could occur; |
| |
• | trading volume of our common stock; |
| |
• | ineffectiveness of our internal control over financial reporting or disclosure controls and procedures; |
| |
• | general political and economic conditions; |
| |
• | effects of natural or man-made catastrophic events; and |
| |
• | other events or factors, many of which are beyond our control. |
In addition, the stock market in general, and the stocks of biotechnology companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance. In addition, other biotechnology companies or our competitors’ programs could have positive or negative results that impact their stock prices and their results or stock fluctuations could have a positive or negative impact on our stock price regardless of whether such impact is direct or not. The realization of any of the above risks or any of a broad range of other risks, including those described in these “Risk Factors,” could have a dramatic and material adverse impact on the market price of our common stock.
We have broad discretion in how we use our cash, cash equivalents and marketable securities, including the net proceeds from our collaborations, public and private securities offerings, and may not use these financial resources effectively, which could affect our results of operations and cause our stock price to decline.
Our management has considerable discretion in the application of our cash, cash equivalents and marketable securities, including the fees and milestone payments from our collaborations and the net proceeds of our securities offerings. We intend to use the cash, cash equivalents and marketable securities to advance our product candidates and for working capital and other general corporate purposes, which will include the hiring of additional personnel and capital expenditures. As a result, investors will be relying upon management's judgment with only limited information about our specific intentions for the use of the cash, cash equivalents and marketable securities. We may use the cash, cash equivalents and marketable securities for purposes that
do not yield a significant return or any return at all for our stockholders. In addition, pending their use, we may invest the financial resources from our collaborations and securities offerings in a manner that does not produce income or that loses value.
If securities or industry analysts do not publish, or cease publishing, research or publish inaccurate or unfavorable research about our business or our market, or if they change their recommendations regarding our stock adversely, our stock price and any trading volume could decline.
Any trading market for our common stock that may develop will depend in part on the research and reports that securities or industry analysts publish about us or our business, markets or competitors. Securities and industry analysts do not currently, and may never, publish research on us or our business. If no securities or industry analysts commence coverage of our company, the trading price for our stock would be negatively affected. If securities or industry analysts initiate coverage, and one or more of those analysts downgrade our stock or publish inaccurate or unfavorable research about our business or our market, our stock price would likely decline. If one or more of these analysts cease coverage of our company or fail to publish reports on us regularly, demand for our stock could decrease, which might cause our stock price and any trading volume to decline.
We currently have and have had in the past material weaknesses in our internal controls over financial reporting in the past. If we fail to maintain proper and effective internal controls, our ability to produce accurate and timely financial statements could be impaired, which could harm our operating results, our ability to operate our business and investors’ views of us.
We are required to comply with Section 404 of the Sarbanes-Oxley Act of 2002, as amended, or the Sarbanes-Oxley Act, subject to certain exceptions. Section 404 of the Sarbanes-Oxley Act requires public companies to conduct an annual review and evaluation of their internal controls and to obtain attestations of the effectiveness of internal controls by independent auditors. However, as discussed in detail below, as an emerging growth company, we are not required to obtain an auditor attestation.
Under the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, issuers that qualify as “emerging growth companies” under the JOBS Act will not be required to provide an auditor’s attestation report on internal controls for so long as the issuer qualifies as an emerging growth company. We currently qualify as an emerging growth company under the JOBS Act, and we may choose not to provide an auditor’s attestation report on internal controls. However, if we cannot favorably assess the effectiveness of our internal control over financial reporting, or if we require an attestation report from our independent registered public accounting firm in the future and that firm is unable to provide an unqualified attestation report on the effectiveness of our internal controls over financial reporting, investor confidence and, in turn, our stock price could be materially adversely affected.
Ensuring that we have adequate internal financial and accounting controls and procedures in place so that we can produce accurate financial statements on a timely basis is a costly and time-consuming effort that will need to be evaluated frequently. Our failure to remediate our material weakness in internal controls and thereafter to maintain the effectiveness of our internal controls in accordance with the requirements of the Sarbanes-Oxley Act could have a material adverse effect on the tradability of our common stock, which in turn would negatively impact our business. We could lose investor confidence in the accuracy and completeness of our financial reports, which could have an adverse effect on the price of our common stock. In addition, if our efforts to comply with new or changed laws, regulations, and standards differ from the activities intended by regulatory or governing bodies due to ambiguities related to practice, regulatory authorities may initiate legal proceedings against us and our business may be harmed.
Based on our assessment under the COSO Internal Control-Integrated Framework, management believes that, as of December 31, 2017, our internal control over financial reporting was not effective, as described below.
In connection with the preparation of our financial statements for the year ended December 31, 2017, we concluded that we had a material weakness relating to the financial statement close process due to the combination of deficiencies. The deficiencies resulted from two separate errors that were not identified by management; one related to the classification of certain operating expenses and one related to the reporting of foreign currency re-measurements on investments.
Shares of our common stock that have not been registered under federal securities laws are subject to resale restrictions imposed by Rule 144, including those set forth in Rule 144(i) which apply to a former “shell company.”
Prior to the closing of the Acquisition, we were deemed a “shell company” under applicable SEC rules and regulations because we had no or nominal operations and either no or nominal assets, assets consisting solely of cash and cash equivalents, or assets
consisting of any amount of cash and cash equivalents and nominal other assets. Pursuant to Rule 144 promulgated under the Securities Act, sales of the securities of a former shell company, such as us, under that rule are not permitted unless at the time of a proposed sale, we are subject to the reporting requirements of Section 13 or 15(d) of the Exchange Act and have filed all reports and other materials required to be filed by Section 13 or 15(d) of the Exchange Act, as applicable, during the preceding 12 months, other than current reports on Form 8-K. Additionally, our previous status as a shell company could also limit our use of our securities to pay for any acquisitions we may seek to pursue in the future. The lack of liquidity of our securities as a result of the inability to sell under Rule 144 for a longer period of time than a non-former shell company could cause the market price of our securities to decline.
If we issue additional shares of our capital stock in the future, our existing stockholders will be diluted.
Our Amended and Restated Articles of Incorporation authorize the issuance of up to 300,000,000 shares of our common stock and up to 10,000,000 shares of preferred stock with the terms, limitations, voting rights, relative rights and preferences and variations of each series that our Board of Directors may determine from time to time. Possible business and financial uses for our authorized capital stock include, without limitation, equity financing, future stock splits, acquiring other companies, businesses or products in exchange for shares of our capital stock, issuing shares of our capital stock to partners or other collaborators in connection with strategic alliances, attracting and retaining employees by the issuance of additional securities under our equity compensation plan, or other transactions and corporate purposes that our Board of Directors deems are in the interests of our company. Additionally, issuances of shares of our capital stock could have the effect of delaying or preventing changes in control or our management. Any future issuances of shares of our capital stock may not be made on favorable terms or at all, they may have rights, preferences and privileges that are superior to those of our common stock, and may have an adverse effect on our business or the trading price of our common stock. The issuance of any additional shares of our common stock will reduce the book value per share and may contribute to a reduction in the market price of the outstanding shares of our common stock. Additionally, any such issuance will reduce the proportionate ownership and voting power of all of our current stockholders.
Sales of a substantial number of shares of our common stock in the public market, or the perception that such sales could occur, could cause our stock price to fall.
If our existing stockholders sell, or indicate an intention to sell, substantial amounts of our common stock in the public market, the trading price of our common stock could decline. As of December 31, 2017, a total of 45,017,062 shares of our common stock were outstanding. Any sales of those shares or any perception in the market that such sales may occur could cause the trading price of our common stock to decline.
In addition, shares of common stock that are either subject to outstanding options or reserved for future issuance under our equity incentive plan will be eligible for sale in the public market to the extent permitted by the provisions of various vesting schedules. If these additional shares of common stock are sold, or if it is perceived that they will be sold, in the public market, the trading price of our common stock could decline.
The resale of shares covered by our effective resale registration statement could adversely affect the market price of our common stock in the public market, which result would in turn negatively affect our ability to raise additional equity capital.
The sale, or availability for sale, of our common stock in the public market may adversely affect the prevailing market price of our common stock and may impair our ability to raise additional equity capital. Pursuant to registration statements filed with the SEC, (i) we previously registered for resale 27,321,870 shares of our common stock, which represents all of the shares of our common stock issued and sold in our private placement consummated in December 2014, shares of our common stock issued to former stockholders of Pieris GmbH in connection with the closing of the Acquisition on December 17, 2014, and shares of common stock issuable upon exercise of common stock purchase warrants issued in connection with the closings of the private placement in December 2014 and (ii) registered for resale (w) 3,225,804 shares of our common stock, (x) 4,963,000 shares of common stock issuable upon the conversion of 4,963 shares of our Series A Convertible Preferred Stock, par value $0.001 per share, and (y) 4,913,280 shares of common stock issuable upon exercise of common stock purchase warrants, which represents all of the securities issued and sold in our private placement consummated in June 2016. In addition (z) 6,325,000 shares of our common stock were issued and sold in our underwritten public offering completed in February 2018. Such shares represented approximately 67% of our outstanding shares of common stock as of February 28, 2018. The resale registration statement permits the resale of these shares at any time without restriction. The resale of a substantial number of shares of our common stock in the public market could adversely affect the market price for our common stock and make it more difficult for investors to sell shares of our common stock at times and prices that investors feel are appropriate. Furthermore, because there are a large number of shares registered pursuant to the resale registration statement, we may continue to offer shares covered by the resale registration statement for a significant period of time, the precise duration of which cannot be predicted. Accordingly,
the adverse market and price pressures resulting from an offering pursuant to the resale registration statement may continue for an extended period of time and continued negative pressure on the market price of our common stock could have a material adverse effect on our ability to raise additional equity capital.
Future sales and issuances of our common stock or rights to purchase common stock, including pursuant to our equity incentive plans or otherwise, could result in dilution of the percentage ownership of our stockholders and could cause our stock price to fall.
Even after giving effect to the funds raised in the past, we expect that significant additional capital will be needed in the future to continue our planned operations. To raise capital, we may sell common stock, convertible securities or other equity securities in one or more transactions at prices and in a manner, we determine from time to time. If we sell common stock, convertible securities or other equity securities in more than one transaction, investors in a prior transaction may be materially diluted. Additionally, new investors could gain rights, preferences and privileges senior to those of existing holders of our common stock. Further, any future sales of our common stock by us or resales of our common stock by our existing stockholders could cause the market price of our common stock to decline.
Pursuant to our 2016 Employee, Director and Consultant Equity Incentive Plan, or the Pieris Plan, we are authorized to grant equity awards to our employees, directors and consultants for up to an aggregate of 3,750,000 shares of our common stock, plus shares granted under the 2014 Plan that expired or were cancelled on or after June 28, 2016, and as of December 31, 2017, we have granted options to purchase approximately 5.4 million shares of our common stock. Any future grants of options, warrants or other securities exercisable or convertible into our common stock, or the exercise or conversion of such shares, and any sales of such shares in the market, could have an adverse effect on the market price of our common stock.
Anti-takeover provisions in our organizational documents could delay or prevent a change of control.
Certain provisions of our Amended and Restated Articles of Incorporation and Amended and Restated Bylaws may have an anti-takeover effect and may delay, defer or prevent a merger, acquisition, tender offer, takeover attempt or other change of control transaction that a stockholder might consider to be in its interests, including attempts that might result in a premium over the market price for the shares held by our stockholders.
These provisions provide, among other things:
| |
• | a classified Board of Directors with staggered three-year terms; |
| |
• | the ability of our Board of Directors to issue one or more series of preferred stock with voting or other rights or preferences that could have the effect of impeding the success of an attempt to acquire us or otherwise effect a change of control; |
| |
• | advance notice for nominations of directors by stockholders and for stockholders to include matters to be considered at stockholder meetings; |
| |
• | certain limitations on convening special stockholder meetings and the prohibition of stockholder action by written consent; and |
| |
• | directors may only be removed for cause and only by the affirmative vote of the holders of at least eighty percent (80%) of the voting power of all of the then-outstanding shares of our capital stock entitled to vote at an election of directors, voting together as a single class. |
These anti-takeover provisions, including those noted above, could make it more difficult for a third party to acquire us, even if the third party’s offer may be considered beneficial by many of our stockholders. As a result, our stockholders may be limited in their ability to obtain a premium for their shares. See “Description of Capital Stock”.
Our Amended and Restated Articles of Incorporation designates the Eighth Judicial District Court of Clark County, Nevada, as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, and therefore limit our stockholders’ ability to choose a forum for disputes with us or our directors, officers, employees or agents.
Our Amended and Restated Articles of Incorporation provide that, to the fullest extent permitted by law, and unless we consent to the selection of an alternative forum, the Eighth Judicial District Court of Clark County, Nevada shall be the sole and exclusive forum for any (i) derivative action or proceeding brought in the name or right of the corporation or on its behalf,
(ii) action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers, employees or agents to the corporation or any of our stockholders, (iii) any action arising or asserting a claim arising pursuant to any provision of Chapters 78 or 92A of the Nevada Revised Statutes or any provision of our articles of incorporation or bylaws, (iv) any action to interpret, apply, enforce or determine the validity of our articles of incorporation or bylaws or (v) any action asserting a claim governed by the internal affairs doctrine. Our Amended and Restated Articles of Incorporation further provide that any person purchasing or otherwise acquiring any interest in shares of our capital stock shall be deemed, to the fullest extent permitted by law, to have notice of and consented to the foregoing provision.
We believe the choice-of-forum provision in our Amended and Restated Articles of Incorporation will help provide for the orderly, efficient and cost-effective resolution of Nevada-law issues affecting us by designating courts located in the State of Nevada (our state of incorporation) as the exclusive forum for cases involving such issues. However, this provision may limit a stockholder’s ability to bring a claim in a judicial forum that it believes to be favorable for disputes with us or our directors, officers, employees or agents, which may discourage such actions against us and our directors, officers, employees and agents. While there is no Nevada case law addressing the enforceability of this type of provision, Nevada courts have on prior occasion found persuasive authority in Delaware case law in the absence of Nevada statutory or case law specifically addressing an issue of corporate law. The Court of Chancery of the State of Delaware ruled in June 2013 that choice-of-forum provisions of a type similar to those included in our Amended and Restated Articles of Incorporation are not facially invalid under corporate law and constitute valid and enforceable contractual forum selection clauses. However, if a court were to find the choice-of-forum provision in our Amended and Restated Articles of Incorporation inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could adversely affect our business, financial condition or results of operations.
The elimination of personal liability of our directors and officers under Nevada law and the existence of indemnification rights held by our directors, officers and employees may result in substantial expenses.
Our Amended and Restated Articles of Incorporation eliminate to the furthest extent permitted under Nevada law the personal liability of our directors and officers to us, our stockholders and creditors for damages as a result of any act or failure to act in his or her capacity as a director or officer. Further, our Amended and Restated Articles of Incorporation, our Amended and Restated Bylaws and individual indemnification agreements that we have entered with each of our directors and officers provide that we are obligated to indemnify, subject to certain exceptions, each of our directors or officers to the fullest extent authorized by Nevada law and, subject to certain conditions, to advance the expenses incurred by any director or officer in defending any action, suit or proceeding prior to its final disposition. Those indemnification obligations could expose us to substantial expenditures to cover the cost of settlement or damage awards against our directors or officers, which we may be unable to afford. Further, those provisions and resulting costs may discourage us or our stockholders from bringing a lawsuit against any of our current or former directors or officers for such damages, even if such actions might otherwise benefit our stockholders.
We do not intend to pay cash dividends on our capital stock in the foreseeable future.
We have never declared or paid any cash dividends on our common stock and do not anticipate paying any dividends in the foreseeable future. We currently intend to retain all future earnings to fund the development and growth of our business. Any future payment of cash dividends in the future will be at the discretion of our Board of Directors and will depend on, among other things, our earnings, financial condition, capital requirements, level of indebtedness, statutory and contractual restrictions applying to the payment of dividends and other considerations that the Board of Directors deems relevant. Our stockholders should not expect that we will ever pay cash or other dividends on our outstanding capital stock.
We will incur increased costs associated with, and our management will need to devote substantial time and effort to, compliance with public company reporting and other requirements.
As a public company listed on the Nasdaq Capital Market, and particularly after we cease to be a "smaller reporting company" or an “emerging growth company”, we will incur significant legal, accounting and other expenses that we did not incur as a private company and that we did not incur prior to the listing of our common stock on the Nasdaq Capital Market, including costs associated with public company reporting requirements. In addition, the rules and regulations of the SEC and the Nasdaq Capital Market impose numerous requirements on public companies, including requirements relating to our corporate governance practices and requirements under Section 404 and other provisions of the Sarbanes-Oxley Act, with which we will now need to comply. Further, since we are subject to the Exchange Act, we are required to, among other things, file annual, quarterly and current reports with respect to our business and operating results. Our management and other personnel will need to devote substantial time to gaining expertise regarding operations as a public company and compliance with applicable laws and regulations, and our efforts and initiatives to comply with those requirements could be expensive. The expenses incurred by
public companies for reporting and corporate governance purposes have increased dramatically in recent years. We are unable currently to estimate these costs with any degree of certainty.
We are an emerging growth company and we cannot be certain if the reduced disclosure requirements applicable to emerging growth companies will make our common stock less attractive to investors.
We are an emerging growth company under the JOBS Act. For as long as we continue to be an emerging growth company, we intend to take advantage of certain exemptions from various reporting requirements that are applicable to other public companies including, but not limited to, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, exemptions from the requirements of holding a nonbinding advisory stockholder vote on executive compensation and any golden parachute payments not previously approved, exemption from the requirement of auditor attestation in the assessment of our internal control over financial reporting and exemption from any requirement that may be adopted by the Public Company Accounting Oversight Board. If we do, the information that we provide stockholders may be different than what is available with respect to other public companies. We cannot predict if investors will find our common stock less attractive because we will rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
Under the JOBS Act, emerging growth companies can delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected to take advantage of this extended transition period. Since we will not be required to comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies, our financial statements may not be comparable to the financial statements of companies that comply with the effective dates of those accounting standards.
We will remain an emerging growth company until the earliest of (1) the end of the fiscal year in which the market value of our common stock that is held by non-affiliates exceeds $700 million as of the end of the second fiscal quarter, (2) the end of the fiscal year in which we have total annual gross revenues of $1 billion or more during such fiscal year, (3) the date on which we issue more than $1 billion in non-convertible debt in a three-year period or (4) December 31, 2019, the end of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement filed under the Securities Act. Decreased disclosures in our SEC filings due to our status as an “emerging growth company” may make it harder for investors to analyze our results of operations and financial prospects.
Having availed ourselves of scaled disclosure available to smaller reporting companies, we cannot be certain if such reduced disclosure will make our common stock less attractive to investors.
Under Section 12b-2 of the Exchange Act, a "smaller reporting company" is a company that we are not an investment company, an asset backed issuer, or a majority-owned subsidiary of a parent company that is not a smaller reporting company, and has a public float of less than $75 million and annual revenues of less than $50 million during the most recently completed fiscal year. Similar to emerging growth companies, smaller reporting companies are permitted to provide simplified executive compensation disclosure in their filings; they are exempt from the provisions of Section 404(b) of the Sarbanes-Oxley Act requiring that independent registered public accounting firms provide an attestation report on the effectiveness of internal controls over financial reporting; and they have certain other decreased disclosure obligations in their SEC filings, including, among other things, only being required to provide two years of audited financial statements in annual reports. We are no longer a smaller reporting company as of December 31, 2017 as our public float exceeded $75 million at June 30, 2017; however, under Item 10(f)(2)(i) of Regulation S-K, we are permitted to avail ourselves of the scaled disclosure requirements available to smaller reporting companies in this Annual Report on Form 10-K. Having ceased to be a smaller reporting company, we will no longer be able to avail ourselves of scaled disclosure beginning with our Quarterly Report on Form 10-Q for the quarterly period ending March 31, 2018. Decreased disclosure in our SEC filings as a result of our having availed ourselves of scaled disclosure may make it harder for investors to analyze our results of operations and financial prospects.
|
| |
Item 1B. | UNRESOLVED STAFF COMMENTS |
Not applicable.
We have two leases for approximately 18,000 square feet of office and laboratory space in Freising, Germany. These leases must be renewed on a quarterly and annual basis, respectively, or would otherwise terminate within 12 months of any non-renewal. We lease 3,950 square feet of office space in Boston, Massachusetts under a sublease that houses our executive offices, clinical operations, and other operational functions. This sublease expires on February 27, 2022 or such earlier date pursuant to the termination provisions of the Sublease Agreement. We believe that our facilities are sufficient to meet our needs and will look for suitable additional space as and when needed.
As of the date of this Annual Report on Form 10-K, we are not currently involved in any material legal proceedings. However, from time to time, we could be subject to various legal proceedings and claims that arise in the ordinary course of our business activities. Regardless of the outcome, legal proceedings can have an adverse impact on us because of defense and settlement costs, diversion of management resources, and other factors.
|
| |
Item 4. | MINE SAFETY DISCLOSURES |
Not applicable.
PART II
|
| |
Item 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market Information
Our common stock is listed on the Nasdaq Capital Market under the symbol “PIRS” and on June 30, 2015 our common stock began trading on the Nasdaq Capital Market. The following table sets forth, for the periods indicated, the high and low closing bid quotations for our common stock, as reported by Nasdaq:
|
| | | | | | | | |
| | Common Stock |
| | High | | Low |
Year Ended December 31, 2017: | | | | |
First Quarter | | $ | 2.85 |
| | $ | 1.46 |
|
Second Quarter | | $ | 5.06 |
| | $ | 2.30 |
|
Third Quarter | | $ | 6.03 |
| | $ | 4.58 |
|
Fourth Quarter | | $ | 7.55 |
| | $ | 4.68 |
|
Year Ended December 31, 2016: | | | | |
First Quarter | | $ | 2.29 |
| | $ | 1.49 |
|
Second Quarter | | $ | 2.41 |
| | $ | 1.60 |
|
Third Quarter | | $ | 1.83 |
| | $ | 1.55 |
|
Fourth Quarter | | $ | 1.96 |
| | $ | 1.36 |
|
Stockholders
As of March 12, 2018, there were 84 and 5 stockholders of record of our common stock and preferred stock, respectively.
Dividends
We have never declared nor paid any cash dividends to stockholders. We do not intend to pay cash dividends on our common stock for the foreseeable future, and currently intend to retain any future earnings to fund our operations and the development and growth of our business. The declaration of any future cash dividend, if any, would be at the discretion of our Board of Directors (subject to limitations imposed under applicable Nevada law) and would depend upon our earnings, if any, our capital requirements and financial position, general economic conditions, and other pertinent conditions.
Unregistered Sales of Securities
None.
Issuer Purchases of Equity Securities
None.
|
| |
Item 6. | SELECTED FINANCIAL DATA |
Not applicable.
|
| |
Item 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes and other financial information included in this Annual Report on Form 10-K. Some of the information contained in this discussion and analysis or set forth elsewhere in this Annual Report, including information with respect to our plans and strategy for our business, includes forward-looking statements that involve risks and
uncertainties as described under the heading “Forward-Looking Statements” elsewhere in this Annual Report on Form 10-K. You should review the disclosure under the heading “Risk Factors” in this Annual Report on Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described in or implied by the forward-looking statements contained in the following discussion and analysis.
Overview
We are a clinical stage biotechnology company that discovers and develops Anticalin-based drugs to target validated disease pathways in a unique and transformative way. Our clinical pipeline includes an immuno-oncology bispecific targeting HER2 and 4-1BB, an inhaled IL-4 receptor alpha antagonist Anticalin protein to treat uncontrolled asthma and a half-life-optimized, hepcidin antagonizing Anticalin protein to treat anemia. Our proprietary Anticalin proteins are a novel class of therapeutics validated in the clinic and by partnerships with leading pharmaceutical companies. Each of our development programs focus on the following:
| |
• | Immuno-oncology drug candidates are multispecific Anticalin-based proteins designed to engage immunomodulatory targets and consist of a variety of multifunctional biotherapeutics that genetically link an antibody with one or more Anticalin proteins, thereby constituting a multispecific protein; |
| |
• | PRS-343 is our lead immuno-oncology program is a 4-1BB/HER2 bispecific, comprised of a HER2-targeting antibody genetically linked to a 4-1BB-targeting Anticalin protein, in which tumor-targeted drug clustering mediated by HER2 expressed on certain solid tumors is intended to drive tumor localized T cell activation for which standard treatment options are not available, are no longer effective, are not tolerated, or the patient has refused standard therapy. |
| |
• | PRS-332 is a bispecific Anticalin-antibody fusion protein comprising an anti-PD-1 antibody genetically fused to an Anticalin targeting an undisclosed checkpoint target. In order to improve on existing PD-1 therapies, we are developing PRS-332 with the intent to simultaneously block PD-1 and another immune checkpoint co-expressed on exhausted T cells. |
| |
• | PRS-060 is a drug candidate that binds to the IL-4Rα receptor, thereby inhibiting IL-4 and IL-13, two cytokines (small proteins mediating signaling between cells within the human body) known to be key mediators in the inflammatory cascade that causes asthma and other inflammatory diseases. |
| |
• | PRS-080 is an Anticalin protein that binds to hepcidin, a natural regulator of iron in the blood. It has been designed to target hepcidin for the treatment of functional iron deficiency in anemic patients with chronic kidney disease particularly in end-stage renal disease patients requiring dialysis. |
Our programs are in varying stages:
| |
• | Immuno-oncology drug candidates—We are conducting activities relating to lead candidate identification, lead candidate optimization, preclinical evaluation and IND filing preparation on several of our 300-Series lead candidates. |
| |
• | Our lead candidate, PRS-343, has been advanced through IND-enabling studies in 2016. Preclinical safety and efficacy studies were performed. A Master Cell bank was generated and GMP material to support initial clinical trials has been produced. We filed an investigational new drug application (“IND”) for our lead immuno-oncology drug candidate, PRS-343, and FDA accepted that IND. The Company commenced dosing the first patient in a Phase I study in HER2-positive solid tumors; and |
| |
• | PRS-332—PRS-332 is currently undergoing preclinical evaluation together with Servier. |
| |
• | PRS-080—We completed a Phase I single-ascending dose (“SAD”) clinical trial with PRS-080 in healthy volunteers in 2015. Based on the data we obtained in the Phase I clinical trial, we initiated a SAD Phase Ib clinical study in CKD5 patients requiring hemodialysis. We completed that Phase Ib study and presented the results in June 2017, which reflected that intravenous administration of PRS-080 was both well-tolerated at all doses, and resulted in a profound decrease in free hepcidin within one hour after infusion, followed by robust mobilization of serum iron, with dose-proportional increases in both the level and duration of serum iron concentration and transferrin saturation following treatment. The Company filed clinical trial applications (“CTA”s) with the German and Czech Republic regulatory authorities to conduct a multi-dose Phase IIa trial for PRS-080 in FID anemia patients in a randomized placebo-controlled trial with two dose cohorts of 4 mg per kg and 8 mg per kg body weight, with four patients |
receiving drug at each dosage level and two patients on placebo within each cohort. The Company initiated enrollment of patients for this study and Aska has the option, following completion of this trial, to obtain an exclusive license to develop and commercialize PRS-080 in Japan, South Korea, and certain other Asian markets (excluding China).
| |
• | PRS-060—We developed a bioprocess that has generated GMP material for use in preclinical safety studies and first in human clinical studies; demonstrated that PRS-060 is well tolerated in preclinical safety studies and that PRS-060 can be formulated for pulmonary delivery by inhalation. Our first-in-human clinical trial for PRS-060 started in December 2017 and we intend to start a study that demonstrate Proof of Mechanism for PRS-060 in the second half of 2018. |
Our core Anticalin technology and platform were developed in Germany, and we have collaboration arrangements with major multi-national pharmaceutical companies headquartered in the U.S., Europe, and Japan. We also established a collaboration with Les Laboratoires Servier and Institut de Recherches Internationales Servier, or Servier, in January 2017 and entered into an exclusive license agreement with Aska Pharmaceutical Co., Ltd., or Aska, in February 2017. In May 2017, we entered into a License and Collaboration Agreement, and a Non-Exclusive Anticalin Platform Technology License Agreement with AstraZeneca AB, or AstraZeneca. In February 2018 we entered into a collaboration and license agreement with Seattle Genetics Inc. ("Seattle Genetics"). We have also in-licensed different antibodies to drive the advancement of our multispecific immuno-oncology pipeline. These include existing agreements with Sanofi Group, or Sanofi, and F. Hoffmann-La Roche Ltd. and Hoffmann-La Roche Inc., or Roche.
Since inception, we have devoted nearly all of our efforts and resources to our research and development activities and have incurred significant net losses. For the years ended December 31, 2017 and 2016, we reported net loss of $17.6 million and $22.8 million, respectively. As of December 31, 2017, we had an accumulated deficit of $120.3 million. We expect to continue incurring substantial losses for the next several years as we continue to develop our clinical and preclinical drug candidates and programs. Our operating expenses are comprised of research and development expenses and general and administrative expenses.
We have not generated any revenues from product sales to date, and we do not expect to generate revenues from product sales for the foreseeable future. Our revenues for the fiscal years ended December 31, 2017 and 2016 were primarily from license and collaboration agreements with our partners.
A significant portion of our operations are conducted in countries other than the United States. Since we conduct our business in U.S. dollars, our main exposure, if any, results from changes in the exchange rates between the euro and the U.S. dollar. At each period end, we remeasure assets and liabilities to the functional currency of that entity (for example, U.S. dollar payables recorded by Pieris GmbH). Remeasurement gains and losses are recorded in the statement of operations line item Other income (expense), net. All assets and liabilities denominated in euros are translated into U.S. dollars at the exchange rate on the balance sheet date. Revenues and expenses are translated at the average rate during the period. Equity transactions are translated using historical exchange rates. All adjustments resulting from translating foreign currency financial statements into U.S. dollars are included in accumulated other comprehensive loss.
Key Financial Terms and Metrics
The following discussion summarizes the key factors our management believes are necessary for an understanding of our consolidated financial statements.
Revenues
We have not generated any revenues from product sales to date, and we do not expect to generate revenues from product sales for the foreseeable future. Our revenues for the last two years have been primarily from the license and collaboration agreements with Roche, Servier and AstraZeneca.
The revenues from Roche, Servier and AstraZeneca have been comprised primarily of upfront payments, research and development services, and milestone payments. We recognized revenues from upfront payments under these agreements based on multiple-element arrangement guidance as we have determined that the licenses to which the payments related did not have standalone value. Research service revenue is recognized when the costs are incurred and the services have been performed. For revenues from research, development, and commercial milestone payments, if the milestones are deemed substantive and the milestone payments are nonrefundable, such amounts are recognized entirely upon successful accomplishment of the milestones, assuming all other revenue recognition criteria are met. Milestones that are not considered substantive are accounted for as contingent revenue and will be recognized when achieved to the extent the Company has no remaining performance obligations under the arrangement. Revenues from sales milestone payments are accounted for as royalties and are
recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met. Royalty payments are recognized in revenues based on the timing of royalty payments earned in accordance with the agreements, which typically is the period when the relevant sales occur, assuming all other revenue recognition criteria are met. We expect our revenues for the next several years to consist of upfront payments, research funding and milestone payments from strategic collaborations we currently have or may establish in the future.
Research and Development Expenses
The process of researching and developing drugs for human use is lengthy, unpredictable, and subject to many risks. We expect to continue incurring substantial expenses for the next several years as we continue to develop our clinical and preclinical drug candidates and programs. We are unable, with any certainty, to estimate either the costs or the timelines in which those expenses will be incurred. Our current development plans focus on the following activities: Our PRS 300-series, which is a franchise currently comprised of the PRS-343 and PRS-332 programs as well as multiple additional programs with Servier, PRS-080, and PRS-060. These programs consume a large proportion of our current, as well as projected, resources.
Our research and development costs include costs that are directly attributable to the creation of certain of our Anticalin® drug candidates and are comprised of:
| |
• | internal recurring costs, such as labor and fringe benefits, materials and supplies, facilities and maintenance costs; and |
| |
• | fees paid to external parties who provide us with contract services, such as preclinical testing, manufacturing and related testing, and clinical trial activities. |
General and Administrative Expenses
General and administrative expenses consist primarily of payroll, employee benefits, equity compensation, and other personnel-related costs associated with executive, administrative and other support staff. Other significant general and administrative expenses include the costs associated with professional fees for accounting, auditing, insurance costs, consulting and legal services.
Results of Operations
Comparison of Years Ended December 31, 2017 and December 31, 2016
The following table sets forth our revenues and operating expenses for the fiscal years ended December 31, 2017 and 2016 (in thousands):
|
| | | | | | | | |
| | Year Ended December 31, 2017 | | Year Ended December 31, 2016 |
Revenues | | $ | 25,275 |
| | $ | 5,831 |
|
| | | | |
Research and development expenses | | 22,285 |
| | 19,699 |
|
General and administrative expenses | | 17,584 |
| | 8,891 |
|
Total operating expenses | | 39,869 |
| | 28,590 |
|
| | | | |
Interest Income | | 152 |
| | 2 |
|
Other income (expense), net | | (2,102 | ) | | 120 |
|
Loss before income taxes | | (16,544 | ) | | (22,637 | ) |
Income tax provision | | 1,103 |
| | 162 |
|
Net loss | | $ | (17,647 | ) | | $ | (22,799 | ) |
Revenues
The following table provides a comparison of revenues for the years ended December 31, 2017 and 2016 (in thousands):
|
| | | | | | | | | | | | |
| | Years ended December 31, | | |
| | 2017 | | 2016 | | Increase/(Decrease) |
License Fees | | $ | 11,285 |
| | $ | 2,736 |
| | $ | 8,549 |
|
Research and development services | | 1,403 |
| | 1,440 |
| | $ | (37 | ) |
Milestone payments | | 12,573 |
| | 1,655 |
| | $ | 10,918 |
|
Other | | 14 |
| | — |
| | $ | 14 |
|
Total Revenue | | $ | 25,275 |
| | $ | 5,831 |
| | $ | 19,444 |
|
| |
• | The $8.6 million increase in revenues from license fees in the twelve months ended December 31, 2017 compared to the twelve months ended December 31, 2016 relates to the recognition of revenue under our collaboration with AstraZeneca which commenced in May 2017 and recognition of revenue under our collaboration with Servier, which commenced in January 2017. These amounts are offset by slightly lower revenues under our collaboration with Roche due to fewer full-time equivalents used in 2017 compared to full-time equivalents used in 2016. |
| |
• | Revenues from research and development services for the twelve months ended December 31, 2017 were comparable to the twelve months ended December 31, 2016. Lower revenue from research and development services being provided to Roche in Fiscal Year 2017 compared to Fiscal Year 2016 was offset by revenue from research and development services being provided to Servier. The collaboration with Servier commenced in January 2017. |
| |
• | The $10.9 million increase in milestone revenue resulted from the achievement of one milestone during the twelve months ended December 31, 2017 under our collaboration with AstraZeneca compared to one milestone achieved under our prior collaboration with Daiichi during the twelve months ended December 31, 2016. |
Research and Development Expenses
The following table provides a comparison of the research and development expenses for our drug candidates and projects for the years ended December 31, 2017 and 2016 (in thousands):
|
| | | | | | | | | | | | |
| | Years ended December 31, | | |
| | 2017 | | 2016 | | Increase/(Decrease) |
PRS-300 series | | $ | 4,071 |
| | $ | 8,508 |
| | $ | (4,437 | ) |
PRS-060 | | 5,034 |
| | 1,729 |
| | $ | 3,305 |
|
PRS-080 | | 2,256 |
| | 1,355 |
| | $ | 901 |
|
Other research and development activities | | 10,924 |
| | 8,107 |
| | $ | 2,817 |
|
Total | | $ | 22,285 |
| | $ | 19,699 |
| | $ | 2,586 |
|
| |
• | the $4.4 million decrease in our PRS-300 series period-over-period is due primarily to a $4.4 million decrease in external manufacturing costs for drug supply in preparation of running clinical trials expected to be initiated in 2017. Additionally, higher pre-clinical and lab supply costs incurred in 2016 were offset by higher clinical costs, license fees, and compensation related expenses for clinical personnel in 2017 for the PRS-343 program which dosed its patient in the fourth quarter of 2017; |
| |
• | the $3.3 million increase for our PRS-060 program period-over-period is due primarily to increases of $0.8 million in our preclinical and CMC costs, $1.2 million for toxicology studies, $0.7 million in license fee payments to TUM and Selexis, and an additional $0.2 million for professional services. Payroll expenses increased by $0.1 million period-over-period. In addition, we recorded $0.8 million research and development tax credit, in connection with our PRS-060 program, in 2016 and no tax credit was available in the 2017 period. These amounts were offset by a $0.5 million decrease in clinical expenses year-over-year; |
| |
• | the $0.9 million increase for PRS-080 period-over-period is mainly due to higher CMC and clinical costs related to the Phase Ib study and preparation of the Phase IIa study which we initiated in the third quarter of 2017; |
| |
• | the $2.8 million increase in other research and development activities is mainly due to increases of $1.6 million of personnel expenses including bonus and stock compensation, $1.6 million in preclinical and CMC costs, $0.7 million in general lab supply expenses, and an additional $0.2 million for other costs including travel, maintenance and recruiting expenses. These amounts are offset by $1.0 million in license fees paid to Enumeral in 2016 and a $0.3 million license fee to TUM in connection with the Roche collaboration agreement in the first quarter of 2016. |
General and Administrative Expenses
General and administrative expenses were $17.6 million for the fiscal year ended December 31, 2017 as compared to $8.9 million for the fiscal year ended December 31, 2016. The period-over-period increase is due to an additional $3.4 million increase in professional services including $2.9 million of transaction fees for our license and collaboration agreements with AstraZeneca, Servier and Aska. Additionally, investments in our G&A functions including $2.8 million in personnel costs, $0.3 million in recruiting costs, $1.2 million professional services (audit, tax, legal and communications) and $1.0 million in other costs (travel, computer expenses & office supplies), have increased to support the growing business.
Non-operating expense (income), net
Our non-operating expense was $2.0 million for the year ended December 31, 2017 as compared to a net non-operating income of $0.1 million for the year ended December 31, 2016. This increase in expense is mainly a result of net foreign currency transaction losses due to the strengthening of the euro against the U.S. dollar, including foreign currency re-measurement of monetary assets, primarily U.S. dollar cash accounts and U.S. dollar receivables in Germany.
Income tax expense
Income tax expense was $1.1 million for the year ended December 31, 2017 as compared to $0.2 million income tax expense for the year ended December 31, 2016. The increase in income tax expense is related to our Australian jurisdiction, net of loss carryforwards, resulting from taxable income from the AstraZeneca agreement.
Liquidity and Capital Resources
Through December 31, 2017, we have funded our operations with $280.6 million of cash that has been obtained from the following main sources: $122.0 million from sales of equity; $137.8 million in total payments received under license and collaboration agreements, including $15.1 million for research and development services costs received from our collaboration partners; $14.2 million from government grants and $6.5 million from loans.
As of December 31, 2017, we had a total of $82.6 million in cash, cash equivalents and investments. We have incurred losses in every period since inception including the years ended December 31, 2017 and 2016, respectively, and have a total accumulated deficit of $120.3 million as of December 31, 2017.
In February 2018, we completed an underwritten public offering of its common stock in which it sold 6,325,000 shares of Common Stock, including the exercise in full by the underwriters of their option to purchase an additional 825,000 shares of Common Stock, to the public at a price of $8.00 per share. The offering was completed under the shelf registration statement that was filed on Form S-3 and declared effective by the SEC on August 3, 2016. Net proceeds of the underwritten public offering, after deducting the underwriting discounts and commissions, were $47.6 million, excluding our offering expenses of approximately $0.3 million.
We have several research and development programs underway in varying stages of development and we expect they will continue to require increasing amounts of cash for development, conducting clinical trials, and testing and manufacturing of product material. We expect cash necessary to fund operations will increase significantly over the next several years as we continue to conduct these activities necessary to pursue governmental regulatory approval of our 300-Series programs PRS-343 and PRS-332, and PRS-080 and PRS-060, and our other product candidates.
The following table provides a summary of operating, investing, and financing cash flows for the years ended December 31, 2017 and 2016 respectively (in thousands):
|
| | | | | | | | |
| | Year Ended December 31, 2017 | | Year Ended December 31, 2016 |
Net cash provided by (used in) operating activities | | $ | 49,810 |
| | $ | (14,388 | ) |
Net cash used in investing activities | | (45,874 | ) | | (560 | ) |
Net cash provided by financing activities | | 4,090 |
| | 15,221 |
|
Net cash provided by operating activities of $49.8 million for the year ended December 31, 2017 is comprised principally of operating expenses of $39.3 million, net of non-cash items, offset by aggregate receipts of $85.7 million from Astra Zeneca, Servier, Aska, and Roche and an increase in net working capital of $2.5 million. Net cash used in operating activities was $14.4 million for the year ended December 31, 2016, comprised principally of operating expenses amounting to $26.2 million offset by aggregate receipts of $9.5 million from Roche and Daiichi and an increase in net working capital of $2.2 million.
Net cash used in investing activities for the year ended December 31, 2017 is mainly attributable to purchase of investments and to a lesser extend to purchase of property and equipment. Net cash used in investing activities for the year ended December 31, 2016 is attributable to purchases of property and equipment.
Net cash provided by financing activities for the year ended December 31, 2017 was primarily due to the exercise of warrants and stock options. Net cash provided by financing activities for the year ended December 31, 2016 was due to the issuance of common and preferred stock under our 2016 Private Placement.
We expect that our existing cash, cash equivalents, and investments coupled with our February 2018 public offering and the upfront payment we received under our collaboration with Seattle Genetics will enable us to fund our operations and capital expenditure requirements for at least into the middle of 2020. Our requirements for additional capital will depend on many factors, including the following:
| |
• | the scope, rate of progress, results and cost of our clinical studies, preclinical testing and other related activities; |
| |
• | the cost of manufacturing clinical supplies, and establishing commercial supplies, of our drug candidates and any products that we may develop; |
| |
• | the number and characteristics of drug candidates that we pursue; |
| |
• | the cost, timing and outcomes of regulatory approvals; |
| |
• | the cost and timing of establishing sales, marketing and distribution capabilities; |
| |
• | the terms and timing of any collaborative, licensing and other arrangements that we may establish; |
| |
• | the timing, receipt and amount of sales, profit sharing or royalties, if any, from our potential products; |
| |
• | the cost of preparing, filing, prosecuting, defending and enforcing any patent claims and other intellectual property rights; and |
| |
• | the extent to which we acquire or invest in businesses, products or technologies, although we currently have no commitments or agreements relating to any of these types of transactions. |
Due to the often-volatile nature of the financial markets, equity and debt financing may be difficult to obtain. In addition, any unfavorable development or delay in the progress of our 300-Series programs PRS-343 and PRS-332, and PRS-080 and PRS-060 could have a material adverse impact on our ability to raise additional capital.
We may seek to raise any necessary additional capital through a combination of private or public equity offerings, debt financings, collaborations, strategic alliances, licensing arrangements and other marketing and distribution arrangements. To the extent that we raise additional capital through marketing and distribution arrangements or other collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our drug candidates, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us. If we raise additional capital through private or public equity offerings, the ownership interest of our existing stockholders will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect our stockholders’
rights. If we raise additional capital through debt financing, we may be subject to covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
Contractual Obligations
Leases
We lease office and laboratory space in Freising, Germany as well as office space in Boston. The German lease agreement has a defined termination date, which is eight months after the end of each quarter, with unlimited renewal periods at our discretion. In June 2016, we entered into a second lease agreement for additional office space in the same building, which has a fixed term of one year along with unlimited renewal periods. As we had not canceled this lease agreement as of June 2017, the term of the lease extends until June 2019. On August 2015, we entered into an Agreement of Sublease to lease approximately 3,950 square feet in Boston, MA. The term of the lease will expire on February 2022.
We record rent expense on a straight-line basis over the lease term period. For the years ended December 31, 2017 and 2016 respectively, we have recognized rent expense under its existing lease agreements in an amount of $0.5 million and $0.5 million for the years ended December 31, 2017 and 2016, respectively.
Our contractual commitments of the non-cancellable portion under theses operating leases as of December 31, 2017 are as follows (in thousands):
|
| | | |
| Total |
2018 | $ | 456 |
|
2019 | 218 |
|
2020 | 200 |
|
2021 | 204 |
|
2022 | 34 |
|
Total minimum lease payments | $ | 1,112 |
|
Off-Balance Sheet Arrangements
We do not have any off-balance sheet arrangements.
Critical Accounting Policies and Estimates
The discussion and analysis of our financial condition and results of operations are based on our financial statements, which have been prepared in accordance with accounting principles generally accepted in the United States, or GAAP. The preparation of these financial statements requires management to make estimates and judgments that affect reported amounts of assets and liabilities as of the date of the balance sheet and reported amounts of revenues and expenses for the periods presented. Management makes estimates and exercises judgment in revenue recognition, share-based payments and income taxes. Judgments must also be made about the disclosure of contingent liabilities, and these estimates and assumptions form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. We base our estimates on historical experience and on various other assumptions that we believe to be reasonable under the circumstances. Actual results may differ from those estimates and under different assumptions or conditions.
We have identified the following accounting policies that we believe require application of management’s most subjective judgments, often requiring the need to make estimates about the effect of matters that are inherently uncertain and may change in subsequent periods. Our actual results could differ from these estimates and such differences could be material.
Revenue Recognition
Multiple-element arrangements
When evaluating multiple-element arrangements, we identify the deliverables included within the agreement and evaluates which deliverables represent separate units of accounting based whether the delivered element has stand-alone value to the collaborator or if the arrangement includes a general right of return for delivered items.
The consideration received is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria are applied to each of the separate units of accounting. We used best estimate of selling
price (“BESP”) methodology to estimate the selling price for each deliverable and unit of accounting because we do not have vendor specific objective evidence (“VSOE”) or third-party evidence (“TPE”) of selling price for these deliverables. To determine the estimated selling price of a deliverable, we consider market conditions as well as entity-specific factors, including those factors contemplated in negotiating the agreements, terms of previous collaborative agreements, similar agreements entered into by third parties, market opportunity, estimated development costs, probability of success, and the time needed to commercialize a product candidate pursuant to the license. In validating our best estimate of selling price, we evaluate whether changes in the key assumptions used to determine the BESP will have a significant effect on the allocation of arrangement consideration among multiple deliverables.
Multiple element arrangements, such as license and development arrangements, are analyzed to determine whether the deliverables, which often include licenses and performance obligations such as research and development services and steering committee services, can be separated or whether they must be accounted for as a combined unit of accounting in accordance with U.S. GAAP. We recognize the arrangement consideration allocated to licenses as revenue upon delivery of the license only if the license has stand-alone value. If the license is considered not to have stand-alone value, the license would then be combined with other undelivered elements into a combined unit of accounting and the license payments and payments for performance obligations would be recognized as revenue when the revenue recognition criteria have been satisfied for the last deliverable within the unit of accounting. In the case of combined units of accounting that include delivered licenses and undelivered services to be provided over time, revenue would be recognized over the estimated period during which services will be provided. For units of accounting that include licenses to be delivered upon satisfactory completion of certain research services, revenue is deferred until the license is delivered and the performance obligation is satisfied.
If we are involved in a steering committee, as part of a multiple element arrangement, we assess whether its involvement constitutes a performance obligation or a right to participate. When steering committee services are determined to be performance obligations, we determine the fair value to be allocated to this deliverable and recognize the revenue over the expected term of the development period of the products. Otherwise, the fair value for participation is combined with other research services or performance obligations and is recognized over the term which we expect to complete its aggregate performance obligations.
We recognize arrangement consideration allocated to each unit of accounting when all of the revenue recognition criteria in ASC 605-25 are satisfied for that particular unit of accounting. For each unit of accounting, we must determine the period over which the performance obligations will be performed and revenue will be recognized. If there is no discernible pattern of performance or objectively measurable performance measures do not exist, then the Company recognizes revenue under the arrangement on a straight-line basis over the period the Company is expected to complete its performance obligations. Conversely, if the pattern of performance over which the service is provided to the customer can be determined and objectively measurable performance measures exist, then the Company recognizes revenue under the arrangement using the proportional performance method. Revenue recognized is limited to the lesser of the cumulative amount of payments received or the cumulative amount of revenue earned as of the period ending date.
Significant management judgment is required in determining the level of effort required under an arrangement and the period over which the Company is expected to complete its performance obligations under an arrangement.
The accounting treatment for options granted to collaborators is dependent upon the nature of the option granted to the collaborative partner. Options are considered substantive if, at the inception of an agreement, we are at risk as to whether the collaborative partner will choose to exercise the option(s) to secure additional goods or services. Factors that are considered in evaluating whether options are substantive include the overall objective of the arrangement, benefit the collaborator might obtain from the agreement without exercising the options, cost to exercise the options relative to the total upfront consideration, and additional financial commitments or economic penalties imposed on the collaborator as a result of exercising the options.
In arrangements where options to obtain additional deliverables are considered substantive, we determine whether the optional licenses are priced at a significant and incremental discount. If the prices include a significant and incremental discount, the option is considered a deliverable in the arrangement. However, if not priced at a discount, the option is not considered a deliverable in the arrangement. When a collaborator exercises an option considered to be at a significant and incremental discount to acquire an additional license, the exercise fee that is attributed to the additional license and any incremental discount allocated at inception are recognized in a manner consistent with the treatment of up-front payments for licenses (i.e., license and research services). In the event an option expires un-exercised, any incremental discounts deferred at the inception of the arrangement are recognized into revenue upon expiration. For options that are non-substantive, the additional licenses to which the options pertain are considered deliverables upon inception of the arrangement; we apply the multiple-element revenue recognition criteria to determine accounting treatment.
Payments or reimbursements resulting from our research and development efforts in multi-element arrangements, in which our research and development efforts are considered to be a deliverable, are included in allocable consideration and allocated to the units of accounting. These reimbursements are recognized as the services are performed and are presented on a gross basis, so long as there is persuasive evidence of an arrangement, the fee is fixed or determinable, and collection of the related receivable is reasonably assured. Revenue recognized cannot exceed the amount that has been earned and has been billed or is currently billable. Amounts received prior to satisfying the above revenue recognition criteria are recorded as deferred revenue in the accompanying balance sheets.
Milestone payments
At the inception of each agreement that includes milestone payments, we evaluate whether each milestone is substantive and at risk to both parties on the basis of the contingent nature of the milestone. This evaluation includes an assessment of whether: (a) the consideration is commensurate with either (1) the entity’s performance to achieve the milestone, or (2) the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting from the entity’s performance to achieve the milestone, (b) the consideration relates solely to past performance, and (c) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. We evaluate factors such as scientific, regulatory, commercial, and other risks that must be overcome to achieve the respective milestone, the level of effort and investment required to achieve the respective milestone, and whether the milestone consideration is reasonable relative to all deliverables and payment terms in the arrangement in making this assessment.
We aggregate milestones into four categories: (i) research milestones, (ii) development milestones, (iii) commercial milestones, and (iv) sales milestones. Research milestones are typically achieved upon reaching certain success criteria as defined in each agreement related to developing an Anticalin protein against the specified target. Development milestones are typically reached when a compound reaches a defined phase of clinical research or passes such phase, or upon gaining regulatory approvals. Commercial milestones are typically achieved when an approved pharmaceutical product reaches the status for commercial sale, including regulatory approval. Sales milestones are certain defined levels of net sales by the licensee, such as when a product first achieves global sales or annual sales of a specified amount.
For revenues from research, development, and commercial milestone payments, if the milestones are deemed substantive and the milestone payments are nonrefundable, such amounts are recognized entirely upon successful accomplishment of the milestones, assuming all other revenue recognition criteria are met. Milestones that are not considered substantive are accounted for as contingent revenue and will be recognized when achieved to the extent we have no remaining performance obligations under the arrangement. Revenues from sales milestone payments are accounted for as royalties and are recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met. Royalty payments are recognized in revenues based on the timing of royalty payments earned in accordance with the agreements, which typically is the period when the relevant sales occur, assuming all other revenue recognition criteria are met.
Contingencies
Accruals are recorded for loss contingencies when it is probable that a liability has been incurred and the amount of the related loss can be reasonably estimated. We evaluate, on a quarterly basis, developments in legal proceedings and other matters that could cause an increase or decrease in the amount of the liability that has been accrued previously. Considering facts known at the time of the assessment, we determine whether potential losses are considered reasonably possible or probable and whether they are estimable. Based upon this assessment, we carry out an evaluation of disclosure requirements and consider possible accruals in the financial statements.
Research and development expense
Research and development costs are charged to expense as incurred in performing research and development activities. Nonrefundable advance payments for research and development goods or services to be received in the future are deferred and capitalized. The capitalized amounts are expensed as the related goods are delivered or the services are performed. Research and development expenses are comprised of costs incurred in performing research and development activities, including salaries and benefits, facilities costs, pre-clinical and clinical costs, contract services, consulting, depreciation and amortization expense, and other related costs. Costs associated with acquired technology, in the form of upfront fees or milestone payments, are charged to research and development expense as incurred.
Income taxes
Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and operating losses and tax credit carry forwards. Deferred tax assets and liabilities are measured using enacted statutory tax rates expected to apply to taxable income in the jurisdictions and years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date.
Based on the level of historical operating results and projections for the taxable income for the future, we have determined that it is more likely than not that its net deferred tax assets will not be realized. Accordingly, we have recorded a full valuation allowance to reduce its net deferred tax assets.
We recognize, measure, present and disclose in our financial statements any uncertain tax positions that we have taken, or expect to take on a tax return. We operate in multiple jurisdictions, both within and outside the United States, and may be subject to audits from various tax authorities. Management’s judgment is required in determining our provision for income taxes, our deferred tax assets and liabilities, liabilities for uncertain tax positions, and any valuation allowance recorded against our net deferred tax assets. We will monitor the realizability of our deferred tax assets and adjust the valuation allowance accordingly.
Our policy is to classify interest and penalties related to unrecognized tax benefits as income tax expense.
Recently Issued Accounting Pronouncements
We review new accounting standards to determine the expected financial impact, if any, that the adoption of each such standard will have. For the recently issued accounting standards that we believe may have an impact on our consolidated financial statements, see “Note 2—Summary of Significant Accounting Policies” in our consolidated financial statements.
Emerging Growth Company and Smaller Reporting Company Status
The Jumpstart Our Business Startups Act of 2012, or the JOBS Act, establishes a class of company called an “emerging growth company,” which generally is a company whose initial public offering was completed after December 8, 2011 and had total annual gross revenues of less than $1 billion during its most recently completed fiscal year. Additionally, Section 12b-2 of the Exchange Act establishes a class of company called a "smaller reporting company," which generally is a company with a public float of less than $75 million as of the last business day of its most recently completed second fiscal quarter or, if such public float is $0, had annual revenues of less than $50 million during the most recently completed fiscal year for which audited financial statements are available. For the year ended December 31, 2017, we qualify as both an emerging growth company and a smaller reporting company.
As an emerging growth company and a smaller reporting company, we are eligible to take advantage of certain exemptions from various reporting requirements that are not available to public reporting companies that do not qualify for those classifications, including without limitation the following:
| |
• | An emerging growth company is exempt from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and financial statements, commonly known as an “auditor discussion and analysis.” |
| |
• | An emerging growth company is not required to hold a nonbinding advisory stockholder vote on executive compensation or any golden parachute payments not previously approved by stockholders. |
| |
• | Neither an emerging growth company nor a smaller reporting company is required to comply with the requirement of auditor attestation of management’s assessment of internal control over financial reporting, which is required for other public reporting companies by Section 404 of the Sarbanes-Oxley Act. |
| |
• | A company that is either an emerging growth company or a smaller reporting company is eligible for reduced disclosure obligations regarding executive compensation in its periodic and annual reports, including without limitation exemption from the requirement to provide a compensation discussion and analysis describing compensation practices and procedures. |
| |
• | A company that is either an emerging growth company or a smaller reporting company is eligible for reduced financial statement disclosure in registration statements, which must include two years of audited financial statements rather than the three years of audited financial statements that are required for other public reporting companies. Smaller reporting companies are also eligible to provide such reduced financial statement disclosure in annual reports on Form 10-K. |
For as long as we continue to be an emerging growth company and/or a smaller reporting company, we expect that we will take advantage of the reduced disclosure obligations available to us as a result of those respective classifications. We will remain an emerging growth company until the earliest of (i) December 31, 2019, the last day of the fiscal year following the fifth anniversary of the date of the first sale of our common stock pursuant to an effective registration statement under the Securities Act; (ii) the last day of the fiscal year in which we have total annual gross revenues of $1 billion or more; (iii) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years; or (iv) on the end of the fiscal year on which we are deemed to be a large accelerated filer under applicable SEC rules. We expect that we will remain an emerging growth company for the foreseeable future, but cannot retain our emerging growth company status indefinitely and will no longer qualify as an emerging growth company on or before December 31, 2019.
Emerging growth companies may elect to take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected to take advantage of the benefits of this extended transition period. Our financial statements may therefore not be comparable to those of companies that comply with such new or revised accounting standards.
In accordance with Section 12b-2 of the Exchange Act, we are no longer a smaller reporting company as of December 31, 2017. However, under Item 10(f)(2)(i) of Regulation S-K, we are permitted to avail ourselves of the scaled disclosure requirements available to smaller reporting companies in this Annual Report on Form 10-K. Having ceased to be a smaller reporting company, we will no longer be able to avail ourselves of scaled disclosure beginning with our Quarterly Report on Form 10-Q for the quarterly period ending March 31, 2018.
|
| |
Item 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK |
Not applicable.
|
| |
Item 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
Our Consolidated Financial Statements required by this Item are as set forth in Item 15 beginning on page F-1 of this Annual Report on Form 10-K.
|
| |
Item 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE |
Not applicable.
|
| |
Item 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
Our management is responsible for establishing and maintaining “disclosure controls and procedures” as such term is defined in Rule 13a-15(e), under the Securities Exchange Act of 1934, as amended, or the Exchange Act, as well as for establishing and maintaining “adequate internal control over financial reporting” as such term is defined in Rule 13a-15(f) under the Exchange Act. The Company’s system of internal controls over financial reporting is designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of the consolidated financial statements in accordance with generally accepted accounting principles.
Because of the inherent limitations surrounding internal controls over financial reporting, our disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the disclosure controls and procedures are met. Additionally, in designing disclosure controls and procedures, our management necessarily was required to apply its judgment in evaluating the cost-benefit relationship of possible disclosure controls and procedures. The design of any disclosure controls and procedures also is based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.
Our management, under the supervision of and with the participation of the Chief Executive Officer and Chief Financial Officer, assessed the effectiveness of the Company’s internal control over financial reporting and disclosure controls and procedures as of December 31, 2017. In making this assessment, management used the updated criteria set forth in 2013 by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control-Integrated Framework.
Based on our assessment under the COSO Internal Control-Integrated Framework, management believes that, as of December 31, 2017, our internal control over financial reporting was not effective, as described below.
A material weakness is a deficiency, or a combination of deficiencies, in internal controls over financial reporting, such that there is a reasonable possibility that a material misstatement of a company’s annual or interim consolidated financial statements would not be prevented or detected on a timely basis.
In connection with the preparation of our financial statements for the year ended December 31, 2017, we concluded that we had a material weakness relating to the financial statement close process due to the combination of deficiencies. The deficiencies resulted from two separate errors that were not identified by management; one related to the classification of certain operating expenses and one related to the reporting of foreign currency re-measurements on investments. The Company implemented its treasury management and investing strategy in the fourth quarter of 2017, as such, this was the first period in which re-measurement of foreign currency impacts on investments was required. These errors were corrected in the financial statements prior to their issuance. We have developed and implemented a remediation plan for this material weakness. We will continue to execute our remediation plan, which includes, among other things, documenting accounting policies and procedures to improve the financial statement close process review function of our internal controls over financial reporting.
To address the material weaknesses described above, we performed additional analysis and other post-closing procedures to ensure that the consolidated financial statements were prepared in accordance with generally accepted accounting principles. These procedures included reviewing our classification of departmental and specific expenses and reviewing the re-measurement and translation requirements for foreign denominated assets and liabilities.
Notwithstanding this material weakness, we have concluded that the financial statements and other financial information included in this Annual Report on Form 10-K, fairly present in all material respects our financial condition, results of operations and cash flows as of, and for, the periods presented.
Changes in Internal Control over Financial Reporting
Except for material weakness described above, there have been no changes in internal control over financial reporting identified in connection with the evaluation of such internal control required by Rules 13a-15(d) and 15d-15(d) under the Exchange Act that occurred during the year ended December 31, 2017 that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
|
| |
Item 9B. | OTHER INFORMATION |
Not applicable.
PART III
|
| |
Item 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
MANAGEMENT
Directors and Executive Officers
The table below sets forth information about our directors and executive officers:
|
| | | | | |
Name | | Age | | Position |
Stephen S. Yoder | | 42 |
| | Chief Executive Officer, President and Director |
Allan Reine | | 43 |
| | Senior Vice President, Chief Financial Officer and Treasurer |
Louis A. Matis | | 67 |
| | Senior Vice President and Chief Development Officer |
James Geraghty (3) | | 64 |
| | Chairman of the Board of Directors |
Michael Richman (1)(3) | | 57 |
| | Director |
Jean-Pierre Bizzari (3) | | 64 |
| | Director |
Steven Prelack (2) | | 61 |
| | Director |
Julian Adams (2) | | 64 |
| | Director |
Christopher Kiritsy (1)(2) | | 53 |
| | Director |
| |
(1) | Member of the compensation committee |
| |
(2) | Member of the audit committee |
| |
(3) | Member of the nominating and corporate governance committee |
Business Experience
The following is a brief account of the education and business experience of our current directors and executive officers:
Stephen S. Yoder. Stephen S. Yoder joined Pieris GmbH as Chief Executive Officer in January 2010 and was appointed to the Board of Directors of Pieris and became Chief Executive Officer and President in December 2014. Prior to joining Pieris GmbH, from July 2003 to December 2010 he led the intellectual property and legal departments at MorphoSys AG, a biotechnology company involved in the development and research of antibodies, as General Counsel. Prior to MorphoSys AG, from September 1999 to June 2003 he worked in several Washington, D.C. law firms, specializing in a life sciences intellectual property practice. Mr. Yoder holds degrees in molecular biology and Spanish from Grove City College and a Juris Doctorate, with honors, from The George Washington University Law School. As an attorney, he is licensed to practice before the United States Patent and Trademark Office, and in the jurisdictions of Maryland and Washington, D.C. We believe that Mr. Yoder adds value to our Board of Directors based on his intimate knowledge of our business plans and strategies of our business and his years of experience in the biotechnology and life sciences industry.
Allan Reine, M.D. Dr. Reine joined Pieris Pharmaceuticals, Inc. as Senior Vice President, Chief Financial Officer and Treasurer in August 2017. Prior to joining Pieris, from 2012 through August 2017, Dr. Reine was a portfolio manager at Lombard Odier Asset Management, where he ran a healthcare portfolio focused on biotechnology and pharmaceutical companies. Before joining Lombard Odier, Dr. Reine served as a healthcare portfolio manager at various funds, from 2003 through 2012 including Citi Principal Strategies, SAC Capital, Trivium Capital and Alexandra Investment Management. Dr. Reine began his career in 2001 at CIBC World Markets where he worked in both biotechnology investment banking and biotechnology equity research. Dr. Reine received his M.D. from the University of Toronto, and his Bachelor of Science in Statistical Sciences from the University of Western Ontario.
Louis A. Matis, Ph.D. Louis A. Matis was appointed Senior Vice President and Chief Development Officer in August 2015. Prior to joining Pieris, Dr. Matis served since June 2011 as Executive Director, Strategic Evaluation at Alexion Pharmaceuticals, where he also served from 1993 to 2000, during which time he advanced to the position of Chief Scientific Officer and had a leading role in discovering the first-in-class complement inhibitor monoclonal antibody Soliris (eculizumab). Before re-joining Alexion in 2011, Dr. Matis served as Chief Executive Officer of CGI Pharmaceuticals, Inc. from 2000 to 2006, and of the Immune Tolerance Institute from 2007 to 2010. From 1977 until joining Alexion in 1993, Dr. Matis held senior research and clinical positions at the National Cancer Institute (the NCI), National Institutes of Health and the FDA Center for Biologics Evaluation and Research. Dr. Matis received a B.A. from Amherst College, an M.D. from the University of Pennsylvania, Perelman School of Medicine, and his clinical training in Internal Medicine at the University of Chicago
Hospitals and Clinics, and in Medical Oncology at the NCI. Dr. Matis is the author of over 120 publications in major scientific and medical journals and is a co-inventor on multiple patents.
James Geraghty. James Geraghty joined the Board of Directors of Pieris in May 2017 and was appointed as Pieris' Chairman of the Board of Directors in December 2017. Mr. Geraghty is an industry leader with thirty years of strategic and leadership experience, including more than twenty years as a senior member of executive teams at biotechnology companies developing and commercializing innovative therapies. He was most recently an Entrepreneur in Residence at Third Rock Ventures, a leading biotech venture and company-formation fund, and previously served as Senior Vice President, North America Strategy and Business Development at Sanofi, which he joined upon its acquisition of Genzyme. Mr. Geraghty spent twenty years at Genzyme, where his roles included Senior Vice President International Development, President of Genzyme Europe, and General Manager of Genzyme's cardiovascular business. He is Chairman of the Board of Idera Pharmaceuticals and Juniper Pharmaceuticals and serves on the Board of Voyager Therapeutics and Fulcrum Therapeutics. He is also a member of the BIO Ventures for Global Health Board of Directors. He started his career in healthcare strategy consulting at Bain and Company. A graduate of the Yale Law School, Mr. Geraghty holds an M.S. from the University of Pennsylvania and a B.A. from Georgetown University. We believe that Mr. Geraghty adds value to our Board of Directors due to his strong life sciences pedigree and extensive background in company building.
Michael Richman. Mr. Richman joined the Board of Directors of Pieris in December 2014 and has served on the supervisory board of Pieris GmbH since October 2014. He is currently the President and Chief Executive Officer of NextCure, Inc. From 2008 through 2015 Mr. Richman was President and Chief Executive Officer of Amplimmune, Inc., a privately held biologics company focused on cancer and autoimmune diseases which was acquired by AstraZeneca in 2013. From May 2007 through June 2008, he served as President and Chief Operating Officer of Amplimmune, Inc. Prior to such time, Mr. Richman has gained years of experience working in research, intellectual property and business development capacities in companies such as Chiron Corporation (now Novartis), MedImmune, Inc. (now AstraZeneca) and MacroGenics. He is a member of the board of directors of Opexa Therapeutics, Inc., a public company, GenVec., a public company, Madison Vaccines, Inc., a private company, and was previously director of Cougar Biotechnology until its acquisition by Johnson & Johnson. Mr. Richman obtained his B.S. in genetics/molecular biology at the University of California at Davis and his M.S.B.A. in international business at San Francisco State University. We believe that Mr. Richman adds value to our Board of Directors due to his extensive experience in mergers and acquisitions, business development and strategic planning for life science companies, as well as executive leadership and management experience.
Jean-Pierre Bizzari, M.D. Dr. Bizzari joined the Board of Directors of Pieris in May 2015. Dr. Bizzari served as Executive Vice-President, Group Head, Clinical Oncology Development at Celgene Corporation, a role he held from October 2008 until his retirement in December 2015. In this position, Dr. Bizzari was responsible for Celgene’s clinical development and operations-statistics teams across the U.S., Europe and Asia/Japan, and has overseen the development and approval of a number of leading oncology products including REVLIMID® (lenalidomide), VIDAZA® (azacitidine), ISTODAX® (romidepsin) and ABRAXANE (nab-paclitaxel). In addition, he was Chairman of Celgene’s hematology oncology development committee and a member of the company’s management committee. Prior to his role at Celgene and from 2004 to 2008, Dr. Bizzari was the Vice President, Clinical Oncology Development for Sanofi-Aventis where he oversaw the approval of Eloxatin (oxaliplatin), Taxotere® (docetaxel) and Elitek (rasburicase). From 2002 to 2004, he was Vice President, Clinical Development Oncology for Sanofi-Synthelabo and from 1993 to 2002 served in the same role for Rhône-Poulenc Rorer (Aventis). Dr. Bizzari is a member of the Scientific Advisory Board of France’s National Cancer Institute and a board member of the EORTC. He is also currently a member of the board of directors of Halozyme Therapeutics, Inc., Transgene SA, iTeos Therapeutics SA, Onxeo SA and Nordic Nanovector ASA. Mr. Bizzari also served as a member of the board of directors of Celator Pharmaceuticals, Inc. from March 2015 until its merger with Jazz Pharmaceuticals plc in July 2106. Dr. Bizzari received his medical degree from the University of Nice (France) and is an oncologist, having trained at La Pitié-Salpêtrière Hospital in Paris, followed by training at the Ontario Cancer Institute and McGill Cancer Center. We believe that Dr. Bizzari adds value to our Board of Directors based on his considerable experience in the pharmaceutical industry and his insight on clinical, regulatory and commercial aspects of drug development, particularly in oncology and global drug approval strategy.
Steven Prelack. Mr. Prelack joined the Board of Directors of Pieris in December 2014. Mr. Prelack is the Senior Vice President and Chief Operating Officer of VetCor, which owns and operates veterinary hospitals across the United States, and has served in this position since June 2012. Prior to that time and since May 2010, Mr. Prelack served at VetCor as Senior Vice President of Operations and as Chief Financial Officer. From 2001 until May 2010, he was the Senior Vice President, Chief Financial Officer and Treasurer of VelQuest Corporation, a provider of automated compliance software solutions for the pharmaceutical industry. He is currently a director and audit committee chair of Galectin Therapeutics, Inc., a publicly traded clinical-stage biotechnology company engaged in drug research and development to create new therapies for fibrotic disease and cancer. Mr. Prelack also previously served as director and audit committee chair for BioVex Group, Inc., a clinical-stage biotechnology company focused on the development and future commercialization of targeted treatments for cancer and the prevention of
infectious disease, which was sold to Amgen in 2011, and as a director of VelQuest Corporation, OPCAT, Inc. and Foodsafe Solutions, Inc. Mr. Prelack is a Certified Public Accountant, received a B.B.A. degree from the University of Massachusetts at Amherst in 1979 and is a member of the National Association of Corporate Directors. We believe that Mr. Prelack adds value to our Board of Directors due to his extensive executive leadership experience, director experience within the biotechnology sector and his many years serving in senior financial and operational management roles.
Julian Adams, Ph.D. Julian Adams, Ph.D., joined the Board of Directors of Pieris in July 2016 and is Chairman & Chief Executive Officer for Gamida Cell. Previously, Dr. Adams was Chief Science Officer and President of Clal Biotechnology Industries and, prior to that, was President of Research & Development of Infinity. Prior to joining Infinity, Dr. Adams served as Senior Vice President, Drug Discovery and Development at Millennium Pharmaceuticals, Inc., where he led the development of bortezomib, also known as Velcade®. Dr. Adams served as Senior Vice President, Research and Development at LeukoSite Inc., a private biopharmaceutical company, from July 1999 until its acquisition by Millennium Pharmaceuticals, Inc. in December 1999. Dr. Adams also served as a director and Executive Vice President of Research and Development at ProScript, Inc., a private biopharmaceutical company, from 1994 until its acquisition by LeukoSite Inc. in 1999. Prior to joining ProScript, Inc., Dr. Adams held a variety of positions with Boehringer Ingelheim, a private pharmaceutical company, and Merck & Co., Inc., a publicly traded pharmaceutical company. Dr. Adams served as a director of Aileron Therapeutics, Inc., a privately held biopharmaceutical company, from 2011 and to 2013, a director of Warp Drive Bio, LLC, a privately held life sciences company, since 2013, and a director of the Princess Margaret Cancer Foundation since November 2014. Dr. Adams received a B.S. from McGill University and a Ph.D. from the Massachusetts Institute of Technology in the field of synthetic organic chemistry. We believe that Dr. Adams adds value to our Board of Directors based on his considerable experience in the pharmaceutical industry and his experience as an executive of successful companies, both public and private, in the life sciences industry.
Christopher Kiritsy. Christopher Kiritsy joined the Board of Directors of Pieris in September 2016. Mr. Kiritsy is a co-founder of Arisaph Pharmaceuticals, Inc. (“Arisaph”) and has served as Arisaph’s President and Chief Executive Officer since 2005. Prior to Arisaph, Mr. Kiritsy served as Executive Vice President, Corporate Development and Chief Financial Officer of Kos Pharmaceuticals, Inc., where he played a key operating role in building the company from start-up to highly profitable, publicly traded, commercial company. During his 10-year tenure at Kos, Mr. Kiritsy spearheaded more than 10 major corporate development transactions and raised approximately $500 million in public equity, including Kos’s initial public offering. Kos was acquired by Abbott Laboratories for $3.7 billion in 2016. Mr. Kiritsy is a seasoned entrepreneur, who possesses more than 20 years of business and technical experience, previously holding senior management positions in R&D, business development and finance. We believe that Mr. Kiritsy adds value to our Board of Directors based on his considerable experience in the pharmaceutical industry and his expertise in corporate development.
Term of Office of Directors
We currently have authorized seven directors. In accordance with our Amended and Restated Articles of Incorporation and Amended and Restated Bylaws, our board of directors is divided into three classes with staggered three-year terms. At each annual meeting of stockholders, the successors to the directors whose terms then expire will be elected to serve until the third annual meeting following the election. Our directors are divided among the three classes as follows:
| |
• | the Class I director are Jean-Pierre Bizzari, Julian Adams, and Christopher Kiritsy and their terms will expire at the annual meeting of stockholders to be held in 2018; |
| |
• | the Class II directors are and Steven Prelack and James Geraghty, and their terms will expire at the annual meeting of stockholders to be held in 2019; and |
| |
• | the Class III directors are Stephen S. Yoder and Michael Richman, and their terms will expire at the annual meeting of stockholders to be held in 2020. |
Any additional directorships resulting from an increase in the number of directors will be distributed among the three classes so that each class will consist of approximately one-third of the directors.
Family Relationships
There are no family relationships among any of our current or former directors or executive officers.
Involvement in Certain Legal Proceedings
None of our directors, executive officers, significant employees, promoters or control persons has been involved in any legal proceeding in the past 10 years that would require disclosure under Item 401(f) of Regulation S-K promulgated under the Securities Act.
Nominations to the Board of Directors
Director candidates are considered based upon various criteria, including without limitation their broad-based business and professional skills and experiences, expertise in or knowledge of the life sciences industry and ability to add perspectives relating to that industry, concern for the long-term interests of our stockholders, diversity, and personal integrity, judgment, and the need of the Board of Directors. Our Board of Directors has a critical role in guiding our strategic direction and overseeing the strategy of our business, and accordingly, we seek to attract and retain highly qualified directors who have sufficient time to engage in the activities of our Board of Directors and to understand and enhance their knowledge of our industry and business plans.
Stockholder recommendations for director candidates must be submitted to our corporate secretary at Pieris Pharmaceuticals, Inc., 255 State Street, 9th Floor, Boston, MA 02109, who will forward all recommendations to the nominating and corporate governance committee. Recommendations for director candidates must be submitted in a timely manner as set forth in our by-laws, and must include the information regarding the stockholder and the proposed director candidate and follow all other procedures regarding stockholder nominations of proposed director candidates set forth in the by-laws. The committee will review and evaluate the qualifications of any such proposed director candidate and conduct inquiries it deems appropriate. All proposed director candidates will be evaluated in the same manner, without regard to the source of the initial recommendation.
Committees of the Board of Directors
Our board has established three standing committees—audit, compensation, and nominating and corporate governance—each of which operates under a charter that has been approved by our board. Our board has determined that all of the members of each of the board’s three standing committees are independent as defined under the rules of the Nasdaq Capital Market. In addition, all members of the audit committee meet the independence requirements contemplated by Rule 10A-3 under the Exchange Act.
Audit Committee
The audit committee’s main function is to oversee our accounting and financial reporting processes and the audits of our financial statements. This committee’s responsibilities include, among other things:
| |
• | appointing our independent registered public accounting firm; |
| |
• | evaluating the qualifications, independence and performance of our independent registered public accounting firm; |
| |
• | approving the audit and non-audit services to be performed by our independent registered public accounting firm; |
| |
• | reviewing the design, implementation, adequacy and effectiveness of our internal accounting controls and our critical accounting policies; |
| |
• | discussing with management and the independent registered public accounting firm the results of our annual audit and the review of our quarterly unaudited financial statements; |
| |
• | reviewing, overseeing and monitoring the integrity of our financial statements and our compliance with legal and regulatory requirements as they relate to financial statements or accounting matters; |
| |
• | reviewing on a periodic basis, or as appropriate, any investment policy and recommending to our board any changes to such investment policy; |
| |
• | preparing the report that the SEC requires in our annual proxy statement; |
| |
• | reviewing and approving any related party transactions and reviewing and monitoring compliance with our code of conduct and ethics; and |
| |
• | reviewing and evaluating, at least annually, the performance of the audit committee and its members including compliance of the audit committee with its charter. |
The members of our audit committee are Steven Prelack, Julian Adams, and Christopher Kiritsy. Steven Prelack serves as the chairperson of the committee. All members of our audit committee meet the requirements for financial literacy under the applicable rules and regulations of the SEC and the NASDAQ Capital Market. Our board of directors has determined that Steven Prelack is an “audit committee financial expert” as defined by applicable SEC rules and has the requisite financial sophistication as defined under the applicable NASDAQ rules and regulations.
Compensation Committee
Our compensation committee reviews and approves policies relating to compensation of our officers and directors and oversees our overall compensation structure, policies and programs. The compensation committee reviews and approves corporate goals and objectives relevant to the compensation of our Chief Executive Officer and other executive officers, evaluates the performance of these officers in light of those goals and objectives and approves the compensation of these officers based on such evaluations. The compensation committee also reviews and approves the issuance of stock options and other awards under our equity plan. The compensation committee will review and evaluate, at least annually, the performance of the compensation committee and its members, including compliance by the compensation committee with its charter.
The members of our compensation committee are Michael Richman and Christopher Kiritsy. Christopher Kiritsy serves as the chairperson of the committee.
Nominating and Corporate Governance Committee
The nominating and corporate governance committee is responsible for assisting our board of directors in discharging the board’s responsibilities regarding the identification of qualified candidates to become board members, the selection of nominees for election as directors at our annual meetings of stockholders (or special meetings of stockholders at which directors are to be elected), and the selection of candidates to fill any vacancies on our board of directors and any committees thereof. In addition, the nominating and corporate governance committee is responsible for overseeing our corporate governance policies, reporting and making recommendations to our board of directors concerning governance matters and oversight of the evaluation of our board of directors.
The members of our nominating and corporate governance committee are James Geraghty, Michael Richman, and Jean-Pierre Bizzari. Mr. Geraghty serves as the chairperson of the committee.
SECTION 16(A) BENEFICIAL OWNERSHIP REPORTING COMPLIANCE
Section 16(a) of the Exchange Act requires directors, executive officers, and persons owning more than 10 percent of a Company’s class of equity securities registered under Section 12 of the Exchange Act to file reports on a timely basis on the initiation of their status as a reporting person and any changes with respect to their beneficial ownership of such equity securities with the SEC. Executive officers, directors and greater than 10 percent stockholders are required by SEC regulations to furnish those companies copies of all Section 16(a) forms they file.
Our records reflect all reports which were required to be filed pursuant to Section 16(a) of the Securities Exchange Act of 1934, as amended, were filed on a timely basis in 2017.
CODE OF CONDUCT AND ETHICS
We have adopted a Code of Ethics and Whistler Blower Policy that applies to all of our employees, including our chief executive officer and acting chief financial and accounting officer. The text of the code of conduct and ethics is posted on our website at www.pieris.com, is filed as an exhibit hereto, and will be made available to stockholders without charge, upon request, in writing to the Corporate Secretary at Pieris Pharmaceuticals, Inc., 255 State St. 9th Floor, Boston, MA 02109. Disclosure regarding any amendments to, or waivers from, provisions of the code of conduct and ethics that apply to our directors, principal executive and financial officers will be included in a Current Report on Form 8-K within four business days following the date of the amendment or waiver, unless website posting or the issuance of a press release of such amendments or waivers is then permitted by the rules of The Nasdaq Stock Market.
|
| |
Item 11. | EXECUTIVE COMPENSATION |
The following table summarizes the compensation earned in each of our fiscal years ended December 31, 2017 and 2016 by our named executive officers, which consisted of our principal executive officer and our two next most highly compensated executive officers who earned more than $100,000 during the fiscal year ended December 31, 2017 and were serving as executive officers as of such date. The table also includes an additional executive who would have been among the two most highly compensated executive officers except for the fact that he was not serving as an executive officer of the Company as of the end of 2017. We refer to the executive officers listed below as the Named Executive Officers.
Summary Compensation Table
|
| | | | | | | | | | | | | | | | | | | | | | |
Name and Principal Position | Year | | Salary | | Bonus ($) | | Option Awards ($) (1) | | All other compensation ($) (2) | | | Total |
Stephen S. Yoder | 2017 | | $ | 450,000 |
| | $ | 225,000 |
| | $ | 564,201 |
| | $ | 10,800 |
| | | $ | 1,250,001 |
|
Chief Executive Officer | 2016 | | $ | 415,000 |
| | $ | 180,000 |
| | $ | 484,740 |
| | $ | 10,600 |
| | | $ | 1,090,340 |
|
Allan Reine Chief Financial Officer | 2017 | | $ | 148,352 |
| (3) | $ | 150,000 |
| | $ | 1,662,130 |
| | | | | $ | 1,960,482 |
|
Louis Matis | 2017 | | $ | 375,000 |
| | $ | 150,000 |
| | $ | 188,348 |
| | $ | — |
| | | $ | 713,348 |
|
Chief Development Officer | 2016 | | $ | 350,000 |
| | $ | 140,000 |
| | | | $ | — |
| | | $ | 490,000 |
|
Claude Knopf | 2017 | | $ | 370,000 |
| | $ | 148,000 |
| | $ | — |
| | $ | 394,275 |
| (2)(4) | | $ | 912,275 |
|
Former Chief Business Officer | 2016 | | $ | — |
| | $ | — |
| | $ | — |
| | $ | — |
| | | $ | — |
|
| |
(1) | These amounts represent the aggregate grant date fair value for the option awards granted during the fiscal years presented, determined in accordance with FASB ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in Note 10 to our Financial Statements, included in this Annual Report on Form 10-K. |
| |
(2) | Represents 401(k) Plan employer contribution. |
| |
(3) | Dr. Reine began employment with us on August 9, 2017. Salary amounts represent the salary actually paid for the fiscal year. |
| |
(4) | Amount includes $383,475 of severance related payments made in accordance with Mr. Knopf´s employment agreement. |
Narrative Disclosure to Summary Compensation Table
Stephen S. Yoder, Chief Executive Officer
Stephen S. Yoder serves as our President and Chief Executive Officer pursuant to an employment agreement dated December 17, 2014, which provides for a continuous term and may be terminated by either party at any time, provided that if Mr. Yoder resigns he shall provide us with at least 90 days’ prior written notice. Pursuant to his employment agreement, Mr. Yoder is eligible to receive an annual discretionary bonus of up to 40% of Mr. Yoder’s then-effective annual base salary, based upon achievement of individual and corporate performance objectives as determined by the Board of Directors or a committee thereof. The Board increased Mr. Yoder’s annual base salary to $415,000 for 2016 and $450,000 for 2017. For 2018 the Board increased Mr. Yoder’s annual base salary to $475,000 and his annual discretionary bonus target of up to 50%.
Pursuant to his employment agreement, Mr. Yoder is prohibited during the term of the agreement, subject to certain exceptions, from (i) accepting any other employment or consultancy, (ii) serving on the board of directors or similar body of any other entity, unless approved by the Chairman of the Board of Directors, and (iii) acquiring, assuming or participating in, directly or indirectly, any financial position, investment or interest known by Mr. Yoder to be adverse or antagonistic to Pieris, its business or prospects, financial or otherwise, or in any competing business.
Mr. Yoder's employment agreement also contains (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter, and
(iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter. Mr. Yoder also agreed to assign certain intellectual property rights to Pieris.
Allan Reine, M.D., Chief Financial Officer
Dr. Allan Reine serves as our Senior Vice President, Chief Financial Officer, and Treasurer pursuant to an employment agreement dated August 9, 2017, which provides for a continuous term and may be terminated by either party at any time, provided that if Dr. Reine resigns, he shall provide us with at least 90 days’ written notice. Pursuant to this agreement, Dr. Reine receives a base salary of $375,000 and is eligible to receive an annual discretionary bonus award of up to 40% of his then-current base salary, based upon the achievement of specific individual and/or Company-wide performance goals as determined by the Board of Directors, or a committee thereof, in its sole discretion. Dr. Reine is entitled to participate in any employee benefit programs, plans and practices on the same terms as other salaried employees on a basis consistent with the participation of other Named Executive Officers.
Dr. Reine, in connection with his employment, was granted an inducement nonqualified stock option to purchase 450,000 shares of common stock of the Company. Twenty-five percent (25%) of the option shall vest on the first anniversary of his employment, with the remaining (75%) to vest over the next three years in equal installments on a quarterly basis beginning on the last day of the next calendar quarter after the vesting start date.
Dr. Reine was also granted an incentive stock option to purchase up to 50,000 shares of common stock of the Company. The incentive option is based on Dr. Reine substantially meeting the following 2017 personal objectives: (1) oversight of the Company's corporate finance functions, (2) contributions to the Company's strategy and leadership, (3) oversight of investor and public relations, and (4) management of the Company's treasury functions. As Dr. Reine achieved all of the objectives the full 50,000 shares vest as to twenty-five percent (25%) of the shares on February 20, 2019 and the remaining seventy-five percent (75%) of the shares shall vest in equal installments on a quarterly basis on the last day of each calendar quarter thereafter, subject in each case to Dr. Reine's continued employment in good standing.
Pursuant to his employment agreement, Dr. Reine is prohibited during the term of the agreement, subject to certain exceptions, from (A) accepting any other employment or consultancy or (B) serving on the board of directors or similar body of any entity, unless such position is approved by the Chief Executive Officer. The agreement also contains (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter, (iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter, and (iv) assignment of certain intellectual property rights to Pieris.
Louis Matis, Chief Development Officer
Dr. Louis Matis serves as our Senior Vice President and Chief Development Officer pursuant to an employment agreement dated July 20, 2015, which provides for a continuous term and may be terminated by either party at any time, provided that if Dr. Matis resigns, he shall provide us with at least 90 days’ written notice. Pursuant to this agreement, Dr. Matis initially received a base salary of $350,000, which was increased to $375,000 for 2017, and increased to $391,875 for 2018 and is eligible to receive an annual discretionary bonus award of up to 40% of his then-current base salary, based upon the achievement of specific individual and/or Company-wide performance goals as determined by the Board or a committee of the Board in its sole discretion.
Dr. Matis is entitled to participate in any employee benefit programs, plans and practices on the same terms as other salaried employees on a basis consistent with the participation of other senior executive officers.
Pursuant to his employment agreement, Dr. Matis is prohibited during the term of the agreement, subject to certain exceptions, from (i) accepting any other employment or consultancy, (ii) serving on the board of directors or similar body of any other entity, unless approved by the Chief Executive Officer, and (iii) acquiring, assuming or participating in, directly or indirectly, any financial position, investment or interest known by Dr. Matis to be adverse or antagonistic to Pieris, its business or prospects, financial or otherwise, or in any competing business.
The agreement also contains (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter and (iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter. Dr. Matis also agreed to assign certain intellectual property rights to Pieris.
Claude Knopf, Former Chief Business Officer
Mr. Knopf served as our Senior Vice President, Chief Business Officer through October 27, 2017 pursuant to an employment agreement dated November 11, 2016, which provides for a continuous term and may be terminated by either party at any time, provided that if Mr. Knopf resigns, he shall provide us with at least 30 days’ written notice. Pursuant to this agreement, Mr. Knopf received a base salary of $370,000 and was eligible to receive an annual discretionary bonus award of up to 40% of his then-current base salary, based upon the achievement of specific individual and/or Company-wide performance goals as determined by the Board of Directors, or a committee thereof, in its sole discretion. Mr. Knopf was also entitled to participate in any employee benefit programs, plans and practices on the same terms as other salaried employees on a basis consistent with the participation of other Named Executive Officers.
The agreement also contains (i) customary confidentiality obligations which are not limited by the term of the agreement, (ii) certain non-compete provisions extending during the term of the agreement and one year thereafter, (iii) certain non-solicitation provisions during the term of the agreement and for one year thereafter, and (iv) assignment of certain intellectual property rights to Pieris.
2017 Bonus Payments
On February 20, 2018, our Compensation Committee approved discretionary cash bonus payments to (i) Mr. Yoder in the amount of $225,000, which was 10% above his target bonus amount of 40% of his annual base salary due to accomplishments above expectations during 2017, (ii) Dr. Reine in the amount of $150,000, which was equal to his target bonus amount, and (iii) Dr. Matis in the amount of $150,000, which was equal to his target bonus amount.
Potential Payments upon Termination or Change in Control
Stephen S. Yoder, Chief Executive Officer
Mr. Yoder’ employment agreement provides that if Mr. Yoder’s employment is terminated (i) by us without cause or (ii) by him for good reason, then we must pay Mr. Yoder (i) a lump-sum payment equal to 12 months of his base salary, (ii) a pro rata portion of the bonus for the year in which the termination occurs, based on year-to-date performance as determined by the Board of Directors, or a committee thereof, in its sole discretion, (iii) an amount equal to his health insurance premium, paid directly or as a reimbursement to Mr. Yoder, for up to a maximum of 12 months and (iv) all unvested equity awards then held by Mr. Yoder will immediately vest in full and become exercisable. The severance and acceleration of any unvested options is expressly conditioned on Mr. Yoder executing and delivering to Pieris a release of claims.
Mr. Yoder's employment agreement also provides that if within 12 months following a change of control Mr. Yoder’s employment is terminated or Mr. Yoder terminates his employment for good reason, and Mr. Yoder executes and delivers to Pieris a release of claims, then he will receive (i) a lump-sum payment equal to 12 months of his base salary at the time of his termination, (ii) his target bonus amount for the year in which the termination occurs, and (iii) an amount equal to 12 months of his health insurance premium, paid directly or as a reimbursement to Mr. Yoder. In addition, all outstanding unvested equity awards will immediately vest in full and become exercisable following termination and any forfeiture restrictions will immediately lapse.
Cause is defined as the occurrence of any of the following events, as determined by the Board of Directors or a committee designated by the Board of Directors, in its sole discretion: (i) Mr. Yoder’s commission of any felony or any crime involving fraud, dishonesty, or moral turpitude under the laws of Germany, the United States or any state thereof; (ii) Mr. Yoder’s attempted commission of, or participation in, a fraud against Pieris; (iii) Mr. Yoder’s intentional, material violation of any contract or agreement between Mr. Yoder and Pieris or of any statutory duty owed to Pieris; (iv) Mr. Yoder’s unauthorized use or disclosure of Pieris’ confidential information or trade secrets; or (v) Mr. Yoder’s gross misconduct.
Good reason means Mr. Yoder’s resignation from all positions he then holds with Pieris if (i) (a) there is a material diminution in Mr. Yoder’s duties and responsibilities with Pieris; (b) there is a material reduction of Mr. Yoder’s base salary; provided, however, that a material reduction in Mr. Yoder’s base salary pursuant to a salary reduction program affecting all or substantially all of the employees of Pieris and that does not adversely affect Mr. Yoder to a greater extent than other similarly situated employees shall not constitute good reason; or (c) Mr. Yoder is required to relocate Mr. Yoder’s primary work location to a facility or location that would increase Mr. Yoder’s one-way commute distance by more than 50 miles from Mr. Yoder’s primary work location as of immediately prior to such change, (ii) Mr. Yoder provides written notice outlining such conditions, acts or omissions to Pieris within 30 days immediately following such material change or reduction, (iii) such material change or reduction is not remedied by Pieris within 30 days following Pieris’ receipt of such written notice and (iv) Mr. Yoder’s resignation is effective not later than 30 days after the expiration of such 30 day cure period.
Allan Reine, Chief Financial Officer
Dr. Reine’s employment agreement provides that if Dr. Reine’s employment is terminated (i) by us without cause or (ii) by him for good reason, then Dr. Reine will be entitled to receive (a) six months of his base salary, (b) a bonus equal to his target bonus amount, pro-rated based on the total number of days elapsed in the calendar year as of the termination date if, as of the date of termination, the Company and Dr. Reine were “on target” to achieve all applicable performance goals, and (c) continuation of COBRA health insurance premiums at the Company’s then-normal rate of contribution for twelve months. In addition, outstanding equity awards held by Dr. Reine shall automatically become vested and, if applicable, exercisable and any forfeiture restrictions shall immediately lapse with respect to seventy-five percent (75%) of the then unvested equity awards. The severance and acceleration of any unvested options is expressly conditioned on Dr. Reine executing and delivering to Pieris a release of claims.
Dr. Reine's employment agreement also provides that if within 12 months following a change of control Dr. Reine's employment is terminated or Dr. Reine terminates his employment for good reason, and Dr. Reine executes and delivers to Pieris a release of claims, then he will receive (a) twelve months of his base salary, (b) his target bonus amount for the year in which the termination occurs, and (c) continuation of COBRA health insurance premiums at the Company’s then-normal rate of contribution for twelve months. In addition, all then outstanding unvested equity awards held by Dr. Reine shall immediately vest in full and, if applicable, become exercisable and all forfeiture restrictions shall immediately lapse.
Louis Matis, Chief Development Officer
Dr. Matis' employment agreement provides that if Dr. Matis’ employment is terminated (i) by us without cause or (ii) by him for good reason, then Dr. Matis will be entitled to receive (a) six months base salary (b) an amount equal to his target bonus amount, pro-rated based on the total number of days elapsed in the calendar year as of the termination date if, as of the date of termination, the Company and Dr. Matis were “on target” to achieve all applicable performance goals and (c) continuation of COBRA health insurance premiums at the Company’s then-normal rate of contribution for twelve months. In addition, outstanding equity awards held by Dr. Matis shall automatically become vested and, if applicable, exercisable and any forfeiture restrictions shall immediately lapse with respect to seventy-five percent (75%) of the then unvested equity awards.
If, in connection with a change of control of Pieris, Dr. Matis’ employment is terminated without cause or Dr. Matis terminates his employment for good reason, he will be entitled to receive (a) twelve months of his base salary, (b) the target bonus amount for the year of termination and (b) continuation of COBRA health insurance premiums at the Company’s then-normal rate of contribution for twelve months. In the case of such a termination in connection with a change in control, outstanding equity awards held by Dr. Matis shall automatically become vested and if, applicable, exercisable and all forfeiture restrictions shall immediately lapse.
“Good Reason” for Dr. Reine and Dr. Matis shall mean the executive’s resignation from all positions he or she then holds with the Company if (i) (A) there is a material diminution in the executive’s duties and responsibilities with the Company or in job title; (B) there is a material reduction of the executive’s base salary; provided, however, that a material reduction in the executive’s base salary pursuant to a salary reduction program affecting all or substantially all of the employees of the Company and that does not adversely affect the executive to a greater extent than other similarly situated employees shall not constitute good reason; or (C) the executive is required to relocate the executive’s primary work location to a facility or location that would increase the executive’s one-way commute distance by more than fifty (50) miles from the executive’s primary work location as of immediately prior to such change, (ii) the executive provides written notice outlining such conditions, acts or omissions to the Company within thirty (30) days immediately following such material change or reduction, (iii) such material change or reduction is not remedied by the Company within thirty (30) days following the Company’s receipt of such written notice and (iv) the executive’s resignation is effective not later than thirty (30) days after the expiration of such thirty (30) day cure period.
“Change of control” for each of the executives shall be deemed to occur (i)when any “person” (as such term is used in Sections 13(d) and 14(d) of the Exchange Act) becomes the “Beneficial Owner” (as defined in Rule 13d-3 under the Exchange Act), directly or indirectly, of securities of Pieris representing 50% or more of the total voting power represented by Pieris’ then outstanding voting securities (excluding for this purpose any such voting securities held by the Pieris or its affiliates or by any employee benefit plan of Pieris) pursuant to a transaction or a series of related transactions which the Board of Directors does not approve; or (ii) a merger or consolidation of Pieris whether or not approved by the Board of Directors, other than a merger or consolidation which would result in the voting securities of Pieris outstanding immediately prior thereto continuing to represent (either by remaining outstanding or by being converted into voting securities of the surviving entity or the parent of such corporation) more than 50% of the total voting power represented by the voting securities of Pieris or such surviving entity or parent of such corporation, as the case may be, outstanding immediately after such merger or consolidation; or (iii) the sale or disposition by Pieris of all or substantially all of its assets in a transaction requiring stockholder approval.
Claude Knopf, former Chief Business Officer
In connection with Mr. Knopf's separation we agreed to pay him the following severance consistent with the terms of his employment : (i) 12 months base salary l, (ii) his target bonus amount of 40% of base salary, (iii) acceleration of the vesting of 75% of the then unvested portion of his option, such that 281,250 shares became vested (in addition to the 125,000 shares vested as of the date of his last day of employment), and (iv) continued participation in the Company's health and dental insurance plans at the Company's expense (except for Mr. Knopf's co-pay or Mr. Knopf's portion of premium payments, if any, which shall be paid directly by Mr. Knopf) for a period through the earlier of (i) December 31, 2018 and (ii) the date Mr. Knopf and his covered dependents, if any, become eligible healthcare coverage under another employer's plan(s).
Outstanding Equity Awards at Fiscal Year-End
The table below summarizes the aggregate stock and option awards held by our named executive officers as of December 31, 2017:
|
| | | | | | | | | | | | | | |
Name | Number of securities underlying unexercised options (#) exercisable | | Number of securities underlying unexercised options (#) unexercisable | | Equity Incentive Plan Awards: Number of Securities Underlying Unexercised Unearned Options (#) (#) unexercisable | | Option exercise price ($) | | Option expiration date |
Stephen S. Yoder Chief Executive Officer, President
| 1,280,000 |
| (1) | — |
| (1) | | | $ | 2.00 |
| | 12/17/2024 |
| 215,250 |
| (2) | 276,750 |
| (2) | | | $ | 1.52 |
| | 2/12/2026 |
| — |
| | 434,350 |
| (4) | | | $ | 1.99 |
| | 2/23/2027 |
Allan Reine Chief Financial Officer | — |
| | 450,000 |
| (5) | | | $ | 5.00 |
| | 8/9/2027 |
| — |
| | | | 50,000 |
| (6) | $ | 5.00 |
| | 8/9/2027 |
Louis Matis Chief Development Officer | 281,250 |
| | 218,750 |
| (3) | | | $ | 3.36 |
| | 8/17/2025 |
| — |
| | 145,000 |
| (4) | | | $ | 1.99 |
| | 2/23/2027 |
Claude Knopf, Former Chief Business Officer | 406,250 |
| | — |
| | | | $ | 1.45 |
| | 11/28/2026 |
| |
(1) | The option award has a grant date of December 17, 2014 and vests pursuant to the following schedule: 25% of the option vested immediately upon grant on December 17, 2014 and the remaining 75% of the option vested ratably over three years in equal installments on a quarterly basis thereafter. |
| |
(2) | The option award has a grant date of February 12, 2016 and vests pursuant to the following schedule: 25% of the option vested on the one-year anniversary of the grant date and the remaining 75% of the option shall vest ratably over three years in equal installments on a quarterly basis thereafter. |
| |
(3) | The option award has a grant date of August 17, 2015 and vests pursuant to the following schedule: 25% of the option vested on the one-year anniversary of the grant date and the remaining 75% of the option vests ratably over three years in equal installments on a quarterly basis thereafter. |
| |
(4) | The option award has a grant date of February 23, 2017 and vests pursuant to the following schedule: 25% of the option vests on the one-year anniversary of the grant date and 75% of the option shall vest ratably over three years in equal installments on a quarterly basis beginning on the last day of the next calendar quarter after the date of grant. |
| |
(5) | The option award has a grant date of August 9,2017 and vests pursuant to the following schedule: 25% of the option vests on the one-year anniversary of the grant date and 75% of the option shall vest ratably over three years in equal installments on a quarterly basis beginning on the last day of the next calendar quarter after the date of grant. |
| |
(6) | Up to 50,000 shares could be earned based on achievement of 2017 personal objectives. The Compensation Committee determined on February 20, 2018 that all of the objectives had been met and 100% of the award was earned. Therefore, twenty-five percent (25%) of the shares shall vest on February 20, 2019 and the remaining seventy-five percent (75%) of the shares shall vest in equal installments on a quarterly basis on the last day of the next calendar quarter thereafter. |
Director Compensation
The table below summarizes all compensation earned by each of our non-employee directors for services performed during our fiscal year ended December 31, 2017. Mr. Yoder is not in the table below because he receives no separate compensation for his services as a director of our company, and all of the compensation earned by Mr. Yoder during our 2017 fiscal year as an executive officer of our company is reflected in the Summary Compensation Table above.
|
| | | | | | | | | | | | |
Name | Fees earned or paid in cash ($) | | Option awards ($) | | | Total ($) |
James Geraghty (1) | $ | 26,045 |
| | $ | 234,610 |
| (8) | | $ | 260,655 |
|
Michael Richman (2) | $ | — |
| | $ | 72,590 |
| (8) | | $ | 72,590 |
|
Steven Prelack (3) | $ | 47,500 |
| | $ | 25,537 |
| (8) | | $ | 73,037 |
|
Jean-Pierre Bizzari (4) | $ | 37,656 |
| | $ | 25,537 |
| (8) | | $ | 63,193 |
|
Julian Adams (5) | $ | 36,719 |
| | $ | 25,537 |
| (8) | | $ | 62,256 |
|
Christopher Kiritsy (6) | $ | 45,938 |
| | $ | 25,537 |
| (8) | | $ | 71,475 |
|
Chau Khuong (7) | $ | — |
| | $ | 83,400 |
| (8) | | $ | 83,400 |
|
| |
(1) | As of December 31, 2017, James Geraghty held option awards for 70,000 shares at exercise prices ranging from $3.94 to $5.79. |
| |
(2) | As of December 31, 2017, Michael Richman held option awards for 180,470 shares at exercise prices ranging from $1.59 to $5.64. |
| |
(3) | As of December 31, 2017, Steven Prelack held option awards for 70,000 shares at an exercise price ranging from $1.59 to $2.01. |
| |
(4) | As of December 31, 2017, Jean-Pierre Bizzari held option awards for 70,000 shares at an exercise price ranging from $1.59 to $2.80. |
| |
(5) | As of December 31, 2017, Julian Adams held option awards for 50,000 shares at exercise prices ranging from$1.73 to $2.01. |
| |
(6) | As of December 31, 2017, Christopher Kiritsy held option awards for 45,000 shares at exercise prices ranging from $1.59 to $2.01. |
| |
(7) | As of December 31, 2017, Chau Khuong, former Chairmen of the Board of Directors, held option awards for 6,115 shares at an exercise price of $4.68. |
| |
(8) | These amounts represent the aggregate grant date fair value of option awards granted to each director in fiscal year 2017 computed in accordance with FASB ASC Topic 718. A discussion of the assumptions used in determining grant date fair value may be found in Note 10 to our Financial Statements, included in this Annual Report on Form 10-K. |
Our director compensation program is administered by our Board with the assistance of the Compensation Committee. The Compensation Committee conducts an annual review of director compensation and makes recommendations to the Board with respect thereto. The Compensation Committee has engaged the services of Pearl Meyer & Partners (“Pearl Meyer”), a national executive compensation consulting firm, to review and provide recommendations concerning the Company's non-employee director compensation policy. On January 11, 2015, our Board of Directors approved a director compensation policy applicable to our non-employee directors. The January 2015 policy provided for annual cash compensation of $25,000 for each non-employee member of the Board of Directors. In addition, the policy provides that the chair of the audit committee will receive additional annual cash compensation of $15,000; the chair of the compensation committee will receive additional annual cash compensation of $10,000; and the chair of the nominating and corporate governance committee will receive additional annual cash compensation of $7,500 and each member of the audit committee other than the chair will receive $7,500 additional cash
compensation, each member of the compensation committee other than the chair will receive $5,000 additional cash compensation and each member of the nominating and corporate governance committee other than the chair will receive $3,750 additional cash compensation. In March 2017, our Board of Directors approved an amendment to the policy to increase the amount of the annual cash retainer paid to each non-employee director from $25,000 to $35,000 and to provide that the Chairperson of the Board of Directors would receive additional annual cash compensation of $25,000. In November 2017 this policy was amended and restated to increase the additional annual cash compensation to be paid to the Chairperson of the Board to $30,000.
Since adoption of the policy in 2015 upon appointment, each new non-employee director receives an option to purchase 30,000 shares of our common stock and since 2016 each ongoing non-employee director receives an option to purchase 20,000 shares on January 25th each year, up from 15,000 shares in the year prior. Under the amended and restated November 2017 policy, the Chairperson of the Board will also receive an additional annual option to purchase 5,000 shares and any newly appointed Chairperson of the Board will receive an initial additional option to purchase 40,000 shares. All options granted under the policy shall have an exercise price equal to the fair market value of the Company’s common stock on the grant date, terminate ten years after the grant date and vest in equal quarterly installments at the end of each quarter following the grant date until the end of the fiscal year in which the grant was made, except that the initial additional option to the Chairperson shall vest as to twenty-five percent on the first anniversary of the date of the Chairman's appointment as Chairman, with the remaining seventy-five percent) vesting in twelve equal quarterly installments at the end of each full fiscal quarter following the initial vesting date.
|
| |
Item 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
Security Ownership of Certain Beneficial Owners and Management
The following table sets forth the number of shares of our common stock beneficially owned as of Feb 28, 2018, by (i) each of our current directors and named executive officers, (ii) all executive officers and directors as a group, and (iii) each person known by us to be the beneficial owner of more than 5% of the outstanding shares of our common stock. We have determined beneficial ownership in accordance with applicable rules of the SEC, which generally provide that beneficial ownership includes voting or investment power with respect to securities. Except as indicated by the footnotes to the table below, we believe, based on the information furnished to us, that the persons named in the table have sole voting and investment power with respect to all shares of common stock that they beneficially own, subject to applicable community property laws.
The information set forth in the table below is based on 45,017,062 shares of our common stock issued and outstanding on of December 31, 2017. In computing the number of shares of common stock beneficially owned by a person and the percentage ownership of that person, we deemed to be outstanding all shares of common stock subject to options, warrants or other convertible securities held by that person that are currently exercisable or will be exercisable within 60 days after February 28, 2018. We did not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person. Except as otherwise noted in the footnotes below, the address for each person listed in the table below, solely for purposes of filings with the SEC, is c/o Pieris Pharmaceuticals, Inc., 225 State Street, 9th Floor, Boston, Massachusetts 02109.
|
| | | | | | |
Name and Address of Beneficial Owner | | Number of Shares Beneficially Owned | | Percentage Beneficially Owned |
5%+ Stockholders: | | | | |
OrbiMed Advisors LLC (1) | | 5,550,000 |
| | 10.32 | % |
Biotechnology Value Fund, L.P. (2) | | 5,407,062 |
| | 9.99 | % |
Directors and Named Executive Officers: | | | | |
Stephen S. Yoder (3) | | 1,640,588 |
| | 2.96 | % |
Michael Richman (4) | | 182,713 |
| | * |
|
James Geraghty (5) | | 40,000 |
| | * |
|
Steven Prelack (6) | | 70,000 |
| | * |
|
Jean-Pierre Bizzari (7) | | 70,000 |
| | * |
|
Louis Matis (8) | | 358,750 |
| | * |
|
Julian Adams (9) | | 50,000 |
| | * |
|
Christopher Kiritsy (10) | | 50,000 |
| | * |
|
Allan Reine (11) | | 65,000 |
| | * |
|
Claude Knopf (12) | | 50,000 |
| | * |
|
All Current Directors and Executive Officers as a Group (9 persons) (13) | | 2,527,051 |
| | 4.60 | % |
| |
(1) | This information is based solely on a Schedule 13D/A filed with the Securities and Exchange Commission on February 21, 2018. Includes 5,500,730 shares held of record by OrbiMed Private Investments III, LP, or OPI III, and 49,270 shares held of record by OrbiMed Associates III, LP, or Associates III. OrbiMed Capital GP III LLC ("GP III") is the sole general partner of OPI III and as such may be deemed to indirectly be the beneficial owner of the shares held by OPI III. OrbiMed Advisors LLC (“OrbiMed”) is the sole managing member of GP III and the general partner of Associates III and as such GP III may be deemed to indirectly beneficially own the shares held by OPI III and Associates III. By virtue of this relationship, GP III, Associates III, and OrbiMed may be deemed to have beneficial ownership of such shares. Advisors exercises investment and voting power through a management committee comprised of Carl L. Gordon, Sven H. Borho and Jonathan T. Silverstein. Each of GP III, Associates III, OrbiMed, Carl L. Gordon, Sven H. Borho and Jonathan T. Silverstein disclaims beneficial ownership of the shares held by OPI III and Associates III, except to the extent of its or his pecuniary interest therein if any. The address for OPI III and Associates III is 601 Lexington Avenue, 54th Floor, New York, New York. |
| |
(2) | This information is based on a Schedule 13G/A filed with the Securities and Exchange Commission, on or about February 14, 2018, and information available to the Company which includes (i) 5,092,062 shares of common stock and (ii) 315,000 shares of common stock issuable upon the conversion of Series A Preferred Stock. The address of the principal business and office of BVF Inc. and certain of its affiliates is 1 Sansome Street, 30th Floor, San Francisco, California, 94104. BVF Inc. and its related entities beneficially hold (i) 2,623,562 shares of Common Stock, (ii) 4,963 shares a Series A Convertible Preferred Stock, which is convertible into 4,963,000 shares of common stock, and (iii) warrants exercisable for 2,977,800 shares of common stock. The Series A Preferred Stock may not be converted and the warrants may not be exercised if, after such conversion or exercise, BVF Inc. and its affiliates would beneficially own more than 9.99% of the number of shares of common stock then issued and outstanding. As a result of the limitation in the previous sentence, (i) 2,592,000 shares of common stock issuable upon the conversion of Series A Preferred Stock and (ii) 2,977,800 shares of common stock issuable upon the exercise of warrants are excluded from the table above. BVF Partners L.P., or Partners, is the general partner of Biotechnology Value Fund, L.P., or BVF, and Biotechnology Value Fund II, L.P., or BVF II; Partners is the investment manager of Biotechnology Value Trading Fund OS LP, or Trading Fund OS, and is the sole member of BVF Partners OS Ltd, or Partners OS. BVF Inc. is the general partner of Partners, and Mark N. Lampert is a director and officer of BVF Inc. Partners OS disclaims beneficial ownership of the shares of common stock beneficially owned by Trading Fund OS. Each of Partners, BVF Inc. and Mr. Lampert disclaims beneficial ownership of the shares of common stock beneficially owned by BVF, BVF II, Trading Fund OS, and certain Partners managed accounts. |
| |
(3) | Includes 6,000 shares of our common stock and 1,634,588 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(4) | Includes 182,713 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(5) | Includes 10,000 shares of our common stock and 30,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(6) | Includes 70,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(7) | Includes 70,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(8) | Includes 10,000 shares of our common stock and 348,750 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(9) | Includes 50,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(10) | Includes 5,000 shares of our common stock and 45,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(11) | Includes 65,000 our common stock and zero shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(12) | Includes 50,000 shares issuable upon the exercise of options to purchase common stock, which are exercisable within 60 days of February 28, 2018. |
| |
(13) | See notes 3 through 12 above. |
Securities Authorized for Issuance under Equity Compensation Plans
Equity Compensation Plan Information
The following table sets forth information as of December 31, 2017 with respect to compensation plans under which equity securities of the Company are authorized for issuance:
|
| | | | | | | | | | |
Plan Category | | Number of securities to be issued upon exercise of outstanding options, warrants and rights | | Weighted-average exercise price of outstanding options, warrants and rights | | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a) |
Equity compensation plans approved by security holders | | 5,443,485 |
| (1) | $ | 2.33 |
| | 1,612,514 |
|
Equity compensation plans not approved by security holders | | 1,531,250 |
| (2, 3, 4, 5) | $ | 3.62 |
| | — |
|
Total | | 6,974,735 |
| | | | 1,612,514 |
|
| |
(1) | All shares are to be issued pursuant to our 2014 Employee, Director and Consultant Equity Incentive Plan and our 2016 Employee, Director and Consultant Equity Incentive Plan. |
| |
(2) | Pursuant to a Stock Option Agreement with Dr. Matis, dated August 17, 2015, Dr. Matis was granted an option to purchase 500,000 shares of Common Stock at a price per share of $3.36, as an inducement material to his entering into employment with us. The grant has a term of ten years and is subject to a vesting schedule of 4 years, with 25% of the shares vesting on August 17, 2016 and 6.25% of the shares vesting each quarter thereafter, subject to his continued employment with the Company. |
| |
(3) | Pursuant to a termination agreement with Mr. Knopf, he has 406,250 options available for exercise as of December 31, 2017. |
| |
(4) | Pursuant to a Stock Option Agreement with Dr. Reine dated August 9, 2017, Dr. Reine was granted an option to purchase 450,000 shares of Common Stock at a price per share of $5.00, as an inducement material to his entering into employment with us. The grant has a term of ten years and is subject to a vesting schedule of 4 years, with 25% of the shares vesting on August 17, 2018 and 6.25% of the shares vesting each quarter thereafter, subject to his continued employment with the Company. |
| |
(5) | Pursuant to a Stock Option Agreement with Dr. Bruns, dated October 12, 2017, Dr. Bruns was granted an option to purchase 175,000 shares of Common Stock at a price per share of $5.87, as an inducement material to his entering into employment with us. The grant has a term of ten years and is subject to a vesting schedule of 4 years, with 25% of the shares vesting on October 12, 2018 and 6.25% of the shares vesting each calendar quarter beginning March 31, 2019, subject to his continued employment with the Company. |
|
| |
Item 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
Related Party Transactions
Except as described below, in the fiscal years ended December 31, 2017 and December 31, 2016, there has not been, nor is there currently proposed, any transaction to which we are or were a party in which the amount involved exceeds the lesser of $120,000 and 1% of the average of its total assets at year-end for the last two completed fiscal years, and in which any of our current directors, executive officers, holders of more than 5% of any class of our voting securities or any of their respective affiliates or immediate family members, had, or will have, a direct or indirect material interest.
We have entered into indemnification agreements with each of our directors and executive officers. Each of those indemnification agreements is in the form approved by our Board of Directors. Those indemnification agreements require that, under the circumstances and to the extent provided for therein, we indemnify such persons to the fullest extent permitted by applicable law against certain expenses and other amounts incurred by any such person as a result of such person being made a party to certain actions, suits and proceedings by reason of the fact that such person is or was a director, officer, employee or agent of our company, any entity that was a predecessor corporation of our company or any of our affiliates. The rights of each person who is a party to such an indemnification agreement are in addition to any other rights such person may have under applicable Nevada law, our Amended and Restated Articles of Incorporation, our Amended and Restated Bylaws, any other agreement, a vote of our stockholders, a resolution adopted by our Board of Directors or otherwise.
Review, Approval or Ratification of Transactions with Related Persons
Pursuant to the written charter of our audit committee, the audit committee is responsible for reviewing and approving all transactions in which we are a participant and in which any parties related to us, including our executive officers, our directors, beneficial owners of more than 5% of our securities, immediate family members of the foregoing persons and any other persons whom our Board of Directors determines may be considered related parties under Item 404 of Regulation S-K, has or will have a direct or indirect material interest. All of the transactions described in this section occurred prior to the adoption of the audit committee charter.
Director Independence
Our Board of Directors undertook a review of the composition of our Board of Directors and independence of each director. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, our Board of Directors has determined that each of James Geraghty, Jean-Pierre Bizzari, Michael Richman, Julian Adams, Christopher Kiritsy, and Steven Prelack would qualify as “independent” as that term is defined by Nasdaq Listing Rule 5605(a)(2). Stephen S. Yoder would not qualify as “independent” under applicable Nasdaq Listing Rules applicable to the Board of Directors generally or to separately designated board committees because he currently serves as our Chief Executive Officer. In making such determinations, our Board of Directors considered the relationships that each of our non-employee directors has with our company and all other facts and circumstances deemed relevant in determining independence, including the beneficial ownership of our capital stock by each non-employee director.
Subject to some exceptions, Nasdaq Listing Rule 5605(a)(2) provides that a director will only qualify as an “independent director” if, in the opinion of our Board of Directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director, and that a director cannot be an “independent director” if (a) the director is, or in the past three years has been, an employee of ours; (b) a member of the director’s immediate family is, or in the past three years has been, an executive officer of ours; (c) the director or a member of the director’s immediate family has received more than $120,000 per year in direct compensation from us within the preceding three years, other than for service as a director or benefits under a tax-qualified retirement plan or non-discretionary compensation (or, for a family member, as a non-executive employee); (d) the director or a member of the director’s immediate family is a current partner of our independent public accounting firm, or has worked for such firm in any capacity on our audit at any time during the past three years; (e) the director or a member of the director’s immediate family is, or in the past three years has been, employed as an executive officer of a company where one of our executive officers serves on the compensation committee; or
(f) the director or a member of the director’s immediate family is an executive officer, partner or controlling stockholder of a company that makes payments to, or receives payments from, us in an amount which, in any twelve-month period during our past three fiscal years, exceeds the greater of 5% of the recipient’s consolidated gross revenues for that year or $200,000 (except for payments arising solely from investments in our securities or payments under non-discretionary charitable contribution matching programs). Additionally, in order to be considered an independent member of an audit committee under Rule 10A-3 of the Exchange Act, a member of an audit committee may not, other than in his or her capacity as a member of the audit committee, the Board of Directors, or any other committee of the Board of Directors, accept, directly or indirectly, any consulting, advisory, or other compensatory fee from the applicable company or any of its subsidiaries or otherwise be an affiliated person of the applicable company or any of its subsidiaries.
|
| |
Item 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
Prior to April 4, 2016, the Audit Committee engaged Ernst & Young GmbH, or E&Y GmbH, as the Company’s independent registered public accounting firm to act as the principal accountant to audit the Company’s period ending 2015 financial statements. On April 4, 2016, the Audit Committee engaged Ernst & Young LLP, or E&Y LLP, as the Company’s independent registered public accounting firm to act as the principal accountant to audit the Company’s period ending 2016 and 2017 financial statements.
The following table presents audit fees for professional services rendered by E&Y GmbH in connection with the Company's annual financial statements for the year ended December 31, 2017:
|
| | | | | | | | |
| | 2017 | | 2016 |
Audit fees: (1) | | $ | 15,307 |
| | $ | 74,563 |
|
Audit related fees: | | — |
| | — |
|
Tax fees: | | — |
| | — |
|
All other fees: | | — |
| | — |
|
Total | | $ | 15,307 |
| | 74,563 |
|
The following table presents fees for professional audit services rendered by E&Y LLP for the audit of the Company’s annual financial statements for the year ended December 31, 2017 and fees billed for other services rendered by E&Y LLP during the period:
|
| | | | | | | | |
| | 2017 | | 2016 |
Audit fees: (1) | | $ | 995,650 |
| | $ | 646,061 |
|
Audit related fees: | | — |
| | — |
|
Tax fees: | | — |
| | — |
|
All other fees: | | — |
| | — |
|
Total | | $ | 995,650 |
| | 646,061 |
|
| |
(1) | Audit fees consisted of audit work performed on the annual financial statements, review of quarterly financial statements, as well as work generally only the independent registered public accounting firm can reasonably be expected to provide, such as the provision of consents in connection with the filing of registration statements, Current Reports on Form 8-K and related amendments and statutory audits. |
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Public Accountant
Consistent with SEC policies regarding auditor independence, the Audit Committee has responsibility for appointing, setting compensation and overseeing the work of our independent registered public accounting firm. In recognition of this responsibility, the Audit Committee has established a policy to pre-approve all audit and permissible non-audit services provided by our independent registered public accounting firm.
Prior to engagement of an independent registered public accounting firm for the next year’s audit, management will submit an aggregate of services expected to be rendered during that year for each of four categories of services to the Audit Committee for approval.
1. Audit services include audit work performed on the annual financial statements, as well as work that generally only an independent registered public accounting firm can reasonably be expected to provide, including comfort letters, statutory audits, and attest services and consultation regarding financial accounting and/or reporting standards.
2. Audit-Related services are for assurance and related services that are traditionally performed by an independent registered public accounting firm, including due diligence related to mergers and acquisitions, employee benefit plan audits, and special procedures required to meet certain regulatory requirements.
3. Tax services include all services performed by an independent registered public accounting firm’s tax personnel except those services specifically related to the audit of the financial statements, and includes fees in the areas of tax compliance, tax planning, and tax advice.
4. Other Fees are those associated with services not captured in the other categories. The Company generally does not request such services from our independent registered public accounting firm.
Prior to engagement, the Audit Committee pre-approves these services by category of service. The fees are budgeted and the Audit Committee requires our independent registered public accounting firm and management to report actual fees versus the budget at year end by category of service. During the year, circumstances may arise when it may become necessary to engage our independent registered public accounting firm for additional services not contemplated in the original pre-approval. In those instances, the Audit Committee requires pre-approval before engaging our independent registered public accounting firm.
The Audit Committee may delegate pre-approval authority to one or more of its members. The member to whom such authority is delegated must report, for informational purposes only, any pre-approval decisions to the Audit Committee at its next scheduled meeting.
PART IV
|
| | |
Item 15. | | EXHIBITS, FINANCIAL STATEMENT SCHEDULES |
| |
Item 15(a). | | The following documents are filed as part of this annual report on Form 10-K: |
| |
Item 15(a)(1) and (2) | | See “Index to Consolidated Financial Statements” on page F-1 to this Annual Report on Form 10-K. Other financial statement schedules have not been included because they are not applicable or the information is included in the financial statements or notes thereto. |
| |
Item 15(a)(3) | | Exhibits |
| |
| | The following is a list of exhibits filed as part of this Annual Report on Form 10-K. |
|
| | | | | | | | | |
Exhibit Number | | Exhibit Description | | | Incorporated by Reference herein from Form or Schedule | | Filing Date | | SEC File / Registration Number |
| | | | | | | | | |
| | Acquisition Agreement, dated as of December 17, 2014, by and among the Registrant, Pieris AG and the former stockholders of Pieris AG named therein | | | Form 8-K (Exhibit 2.1) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Amended and Restated Articles of Incorporation of the Registrant | | | Form 8-K (Exhibit 3.1) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Certificate of Designation of Series A Convertible Preferred Stock | | | Form 10-Q (Exhibit 3.1) | | August 11, 2016 | | 001-37471 |
| | | | | | | | | |
| | Amended and Restated Bylaws of the Registrant | | | Form 8-K (Exhibit 3.2) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Form of Common Stock certificate | | | Form 8-K (Exhibit 4.1) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Form of Common Stock certificate | | | Form 10-K (Exhibit 4.2) | | March 23, 2016 | | 001-37471 |
| | | | | | | | | |
| | 2014 Employee, Director and Consultant Equity Incentive Plan | # | | Form 8-K (Exhibit 10.1) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Form of Stock Option Award Agreement under the Registrant’s 2014 Employee, Director and Consultant Equity Incentive Plan | # | | Form 8-K (Exhibit 10.2) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | 2016 Employee, Director and Consultant Equity Incentive Plan | # | | Form 8-K (Exhibit 10.1) | | July 1, 2016 | | 001-37471 |
| | | | | | | | | |
| | Form of Stock Option Award Agreement under the Registrant’s 2016 Employee, Director and Consultant Equity Incentive Plan | # | | Form 10-K (Exhibit 10.4) | | March 30, 2017 | | 001-37471 |
| | | | | | | | | |
| | | | | | |
| | Research and Licensing Agreement by and between Pieris AG and Technische Universität München, dated as of July 26, 2007 | ± | | Form 10-K (Exhibit 10.10) | | March 30, 2015 | | 333-190728 |
| | | | | | |
| | | | | | | | | |
| | License and Transfer Agreement by and between the Company and Enumeral Biomedical Holdings, Inc dated as of April 18, 2016 | ± | | Form 10-Q/A (Exhibit 10.1) | | July 20, 2016 | | 001-37471 |
| | | | | | |
| | Definitive License and Transfer Agreement by and between the Company and Enumeral Biomedical Holdings, Inc. dated as of June 6, 2016 | ± | | Form 10-Q (Exhibit 10.1) | | August 11, 2016 | | 001-37471 |
| | | | | |
|
| | | | | | | | | |
Exhibit Number | | Exhibit Description | | | Incorporated by Reference herein from Form or Schedule | | Filing Date | | SEC File / Registration Number |
| | Amendment No.1 to Definitive License and Transfer Agreement by and between the Company and Enumeral Biomedical Holdings, Inc. effective as of January 3, 2017 | | | Form 10-K (Exhibit 10.14) | | March 30, 2017 | | 001-37471 |
| | | | | | |
| | Collaboration Agreement by and among the Registrant, Pieris Pharmaceuticals GmbH, Les Laboratoires Servier and Institut de Recherches Internationales Servier, dated as of January 4, 2017 | @ | | Form 10-K (Exhibit 10.15) | | March 30, 2017 | | 001-37471 |
| | | | | | |
| | Non-Exclusive Anticalin Platform Technology License Agreement Agreement by and among the Registrant, Pieris Pharmaceuticals GmbH, Les Laboratoires Servier and Institut de Recherches Internationales Servier, dated as of January 4, 2017 | @ | | Form 10-K (Exhibit 10.16) | | March 30, 2017 | | 001-37471 |
| | | | | | |
| | First Amendment to the License and Collaboration Agreement by and between Les Laboratoires Servier, Institut de Recherches Internationales Servier, Pieris Pharmaceuticals, Inc. and Pieris Pharmaceuticals GmbH, effective as of June 16, 2017 | @ | | Form 10-Q (Exhibit 10.4) | | August 11, 2017 | | 001-37471 |
| | | | | | | | | |
| | Exclusive Option Agreement by and among the Registrant, Pieris Pharmaceuticals GmbH and ASKA Pharmaceutical Co., Ltd., dated as of February 27, 2017 | @ | | Form 10-Q (Exhibit 10.3) | | May 15, 2017 | | 001-37471 |
| | | | | | | | | |
| | License & Collaboration Agreement by and between Pieris Pharmaceuticals Inc., Pieris Pharmaceuticals GmbH & Pieris Australia Pty. Limited and AstraZeneca AB, dated as of May 2, 2017 | @ | | Form 10-Q (Exhibit 10.1) | | August 11, 2017 | | 001-37471 |
| | | | | | | | | |
| | Non-Exclusive Anticalin® Platform Technology License Agreement, by and between Pieris Pharmaceuticals Inc., Pieris Pharmaceuticals GmbH and Pieris Australia Pty. Limited and AstraZeneca AB, dated as of May 2, 2017 | @ | | Form 10-Q (Exhibit 10.1) | | August 11, 2017 | | 001-37471 |
| | | | | | | | | |
| | Form of Indemnification Agreement by and between the Registrant and each of its current directors and executive officers | # | | Form 8-K (Exhibit 10.10) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Employment Agreement by and between the Registrant and Stephen S. Yoder, dated as of December 17, 2014 | # | | Form 8-K (Exhibit 10.15) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Separation Agreement by and between the Registrant and Darlene Deptula-Hicks, dated as of February 7, 2017 | # | | Form 10-K (Exhibit 10.26) | | March 30, 2017 | | 001-37471 |
| | | | | |
| | Employment Agreement by and between the Registrant and Louis A. Matis, M.D., dated as of July 20, 2015 | # | | Form 10-Q (Exhibit 10.1) | | November 13, 2015 | | 001-37471 |
| | | | | |
| | Employment Agreement by and between the Registrant and Claude Knopf, dated of November 14, 2016 | # | | Form 10-K (Exhibit 10.28) | | March 30, 2017 | | 001-37471 |
| | | | | |
|
| | | | | | | | | |
Exhibit Number | | Exhibit Description | | | Incorporated by Reference herein from Form or Schedule | | Filing Date | | SEC File / Registration Number |
| | Consulting Agreement by and between the Registrant and Danforth Advisors, LLC, dated as of February 1, 2017 | # | | Form 10-K (Exhibit 10.29) | | March 30, 2017 | | 001-37471 |
| | | | | |
| | Employment Agreement by and between Pieris Pharmaceuticals, Inc. and Allan Reine, dated as of August 9, 2017 | # | | Form 10-Q (Exhibit 10.5) | | August 11, 2017 | | 001-37471 |
| | | | | | | | | |
| | Non-Qualified Stock Option Agreement, dated August 9, 2017, between Pieris Pharmaceuticals Inc. and Allan Reine, M.D. | # | | Form 10-Q (Exhibit 10.2) | | November 13, 2017 | | 001-37471 |
. | | | | | | | | | |
| | Non-Employee Director Compensation Policy, as amended | *# | | | | | | |
| | | | | |
| | Lease Agreement by and between Pieris AG and Födergesellschft IZB mbH, dated as of May 4, 2011 | | | Form 8-K (Exhibit 10.23) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Agreement of Sublease by and between Berenberg Capital Markets LLC and the Registrant, dated as of August 27, 2015 | | | Form 10-Q (Exhibit 10.3) | | November 13, 2015 | | 001-37471 |
| | | | | |
| | Repayment Agreement by and between Pieris AG and tbg Technologie-Beteiligungs-Gesellschaft mbH, dated as of April 3, 2014 | | | Form 8-K (Exhibit 10.27) | | December 18, 2014 | | 333-190728 |
| | | | | | | | | |
| | Settlement Agreement (Accelerated Repayment Agreement) by and between Pieris AG and tbg Technologie-Beteiligungs-Gesellschaft mbH, dated as of December 11, 2014 | | | Form 8-K (Exhibit 10.28) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Consolidated Shareholders’ Agreement 2014, Pieris AG, Freising, Germany, by and among Pieris AG and the Stockholders party thereto, dated October 10, 2014 | | | Form 8-K (Exhibit 10.30) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Investment Agreement, Pieris AG, Freising, Germany, by and among Pieris AG, Stephen Yoder and the Existing Shareholders party thereto, dated October 10, 2014 | | | Form 8-K (Exhibit 10.31) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Agreement, by and among Pieris AG and the Stockholders party thereto, dated December 5, 2014 | | | Form 8-K (Exhibit 10.32) | | December 18, 2014 | | 333-190728 |
| | | | | |
| | Form of Securities Purchase Agreement, dated December 17, 2014, by and among the Registrant and the Purchasers | | | Form 8-K (Exhibit 10.1) | | December 23, 2014 | | 333-190728 |
| | | | | |
| | Form of Registration Rights Agreement, dated December 17, 2014, by and among the Registrant and the investors party thereto | | | Form 8-K (Exhibit 10.2) | | December 23, 2014 | | 333-190728 |
| | | | | |
| | Form of Warrant to Purchase Common Stock, dated December 17, 2014, issued by the Registrant | | | Form 8-K (Exhibit 10.3) | | December 23, 2014 | | 333-190728 |
| | | | | |
| | Securities Purchase Agreement, dated June 2, 2016, by and among the Registrant and the Investors named therein | | | Form 8-K (Exhibit 10.1) | | June 6, 2016 | | 001-37471 |
| | | | | |
|
| | | | | | | | | |
Exhibit Number | | Exhibit Description | | | Incorporated by Reference herein from Form or Schedule | | Filing Date | | SEC File / Registration Number |
| | Form of Warrant to purchase Common Stock, dated June 2, 2016, issued by the Registrant | | | Form 8-K (Exhibit 10.2) | | June 6, 2016 | | 001-37471 |
| | | | | |
| | Registration Rights Agreement, dated June 2, 2016, by and among the Registrant and the Investors named therein | | | Form 8-K (Exhibit 10.3) | | June 6, 2016 | | 001-37471 |
| | | | | |
| | Corporate Code of Ethics and Conduct and Whistleblower Policy | * | | | | | | |
| | | | | |
| | List of Subsidiaries | * | | | | | | |
| | | | | |
| | Consent of Ernst & Young LLP | * | | | | | | |
| | | | | |
| | Certification of Stephen S. Yoder, Chief Executive Officer and President, pursuant to Section 302 of the Sarbanes—Oxley Act of 2002 | * | | | | | | |
| | | | | |
| | Certification of Allan Reine, Chief Financial Officer, pursuant to Section 302 of the Sarbanes—Oxley Act of 2002 | * | | | | | | |
| | | | | | | | | |
| | Certification of Stephen S. Yoder, Chief Executive Officer and President, pursuant to Section 906 of the Sarbanes—Oxley Act of 2002, 18 U.S.C. Section 1350 | ** | | | | | | |
| | | | | |
| | Certification of Allan Reine, Chief Financial Officer, pursuant to Section 906 of the Sarbanes—Oxley Act of 2002, 18 U.S.C. Section 1350 | ** | | | | | | |
| | | | | |
101.INS | | XBRL Instance Document | * | | | | | | |
| | | | | |
101.SCH | | XBRL Taxonomy Extension Schema Document | * | | | | | | |
| | | | | |
101.CAL | | XBRL Taxonomy Extension Calculation Linkbase Document | * | | | | | | |
| | | | | |
101.DEF | | XBRL Taxonomy Extension Definition Linkbase Document | * | | | | | | |
| | | | | |
101.LAB | | XBRL Taxonomy Extension Label Linkbase Document | * | | | | | | |
| | | | | |
101.PRE | | XBRL Taxonomy Extension Presentation Linkbase Document | * | | | | | | |
| | | | | | | | | |
* | Filed herewith | | | | | | | |
| | | | | | | | | |
** | Furnished herewith | | | | | | | |
| | | | | | | | | |
± | Confidential treatment received as to portions of the exhibit. Confidential materials omitted and filed separately with the SEC. | | | | | | | |
| | | | | | | | | |
@ | Confidential treatment requested as to portions of the exhibit. Confidential materials omitted and filed separately with the SEC | | | | | | | |
| | | | | | | | | |
# | Indicates a management contract or compensatory plan | | | | | | | |
We may voluntarily include a summary of information required by Form 10-K under this Item 16. We have elected not to include such summary information.
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
| | | |
| PIERIS PHARMACEUTICALS, INC. |
| | | |
March 15, 2018 | By: | | /s/ Stephen S. Yoder |
| | | Stephen S. Yoder |
| | | Chief Executive Officer and President |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities indicated below and on the dates indicated.
|
| | | | |
Signature | | Title | | Date |
| | |
/s/ Stephen S. Yoder | | President, Chief Executive Officer and | | March 15, 2018 |
Stephen S. Yoder | | Director (Principal Executive Officer) | | |
| | |
/s/ Allan Reine | | Chief Financial Officer and | | March 15, 2018 |
Allan Reine | | Treasurer (Principal Financial and Accounting Officer) | | |
| | |
/s/ James Geraghty | | Chairman of the Board of Directors | | March 15, 2018 |
James Geraghty | | | | |
| | |
/s/ Jean-Pierre Bizzari | | Director | | March 15, 2018 |
Jean-Pierre Bizzari | | | | |
| | |
/s/ Michael Richman | | Director | | March 15, 2018 |
Michael Richman | | | | |
| | |
/s/ Steven Prelack | | Director | | March 15, 2018 |
Steven Prelack | | | | |
| | |
/s/ Julian Adams | | Director | | March 15, 2018 |
Julian Adams | | | | |
| | |
/s/ Christopher Kiritsy | | Director | | March 15, 2018 |
Christopher Kiritsy | | | | |
PIERIS PHARMACEUTICALS, INC.
Table of Contents
Report of Independent Registered Public Accounting Firm
To The Shareholders and the Board of Directors of Pieris Pharmaceuticals, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Pieris Pharmaceuticals, Inc. (the “Company”) as of December 31, 2017 and 2016, and the related consolidated statements of operations, comprehensive loss, stockholders' equity, and cash flows for each of the two years in the period ended December 31, 2017, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2017 and 2016, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2017, in conformity with U.S. generally accepted accounting principles.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company´s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain and understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
|
| | | | |
/s/ Ernst & Young LLP | | | | |
We have served as the Company's auditor since 2016. | | | | |
Boston, Massachusetts
| | | | |
March 15, 2018 | | | | |
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED BALANCE SHEETS
(In thousands, except share and per share data)
|
| | | | | | | | |
| | December 31, |
| | 2017 | | 2016 |
Assets | | | | |
Current assets: | | | | |
Cash and cash equivalents | | $ | 37,878 |
| | $ | 29,356 |
|
Short term investments | | 34,751 |
| | — |
|
Accounts receivable | | 15,546 |
| | 57 |
|
Prepaid expenses and other current assets | | 1,615 |
| | 3,260 |
|
Total current assets | | 89,790 |
| | 32,673 |
|
Property and equipment, net | | 4,034 |
| | 2,264 |
|
Long term investments | | 9,922 |
| | — |
|
Other non-current assets | | 130 |
| | 126 |
|
Total assets | | $ | 103,876 |
| | $ | 35,063 |
|
Liabilities and stockholders’ equity | | | | |
Current liabilities: | | | | |
Accounts payable | | $ | 2,452 |
| | $ | 2,386 |
|
Accrued expenses and other current liabilities | | 6,170 |
| | 3,719 |
|
Deferred revenues, current portion | | 37,153 |
| | 2,275 |
|
Total current liabilities | | 45,775 |
| | 8,380 |
|
Deferred revenue, net of current portion | | 46,542 |
| | 1,409 |
|
Other long-term liabilities | | 37 |
| | 47 |
|
Total liabilities | | 92,354 |
| | 9,836 |
|
Stockholders’ equity: | | | | |
Preferred stock, $0.001 par value per share, 10,000,000 shares authorized and 4,963 and 4,963 issued and outstanding at December 31, 2017 and December 31, 2016 | | — |
| | — |
|
Common stock, $0.001 par value per share, 300,000,000 shares authorized and 45,017,062 and 43,058,827 issued and outstanding at December 31, 2017 and December 31, 2016 | | 45 |
| | 43 |
|
Additional paid-in capital | | 136,484 |
| | 129,350 |
|
Accumulated other comprehensive loss | | (4,695 | ) | | (1,501 | ) |
Accumulated deficit | | (120,312 | ) | | (102,665 | ) |
Total stockholders’ equity | | 11,522 |
| | 25,227 |
|
Total liabilities and stockholders’ equity | | $ | 103,876 |
| | $ | 35,063 |
|
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS AND COMPREHENSIVE LOSS
(In thousands, except per share data)
|
| | | | | | | | |
| | Years Ended December 31, |
| | 2017 | | 2016 |
Revenue | | $ | 25,275 |
| | $ | 5,831 |
|
Operating expenses | | | | |
Research and development | | 22,285 |
| | 19,699 |
|
General and administrative | | 17,584 |
| | 8,891 |
|
Total operating expenses | | 39,869 |
| | 28,590 |
|
Loss from operations | | (14,594 | ) | | (22,759 | ) |
Interest income | | 152 |
| | 2 |
|
Other (expense) income, net | | (2,102 | ) | | 120 |
|
Loss before income taxes | | (16,544 | ) | | (22,637 | ) |
Provision for income tax | | 1,103 |
| | 162 |
|
Net loss | | (17,647 | ) | | (22,799 | ) |
| | | | |
Foreign currency translation | | (2,374 | ) |
| (229 | ) |
Unrealized loss on available-for-sale securities | | (820 | ) | | — |
|
Comprehensive loss | | $ | (20,841 | ) | | $ | (23,028 | ) |
Net loss per share | | | | |
Basic and diluted | | $ | (0.40 | ) | | $ | (0.55 | ) |
Weighted average number of common shares outstanding | | | | |
Basic and diluted | | 43,931 |
| | 41,713 |
|
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY
(In thousands)
|
| | | | | | | | | | | | | | | | | | | | | | | | | | | | | | |
| | Convertible series A preferred shares | | Common Shares | | Additional paid-in capital | | Accumulated other comprehensive loss | | Accumulated deficit | | Total equity |
| | No. of shares | | Share capital | | No. of shares | | Share capital | |
Balance as of January 1, 2016 | | — |
| | $ | — |
| | 39,833 |
| | $ | 40 |
| | $ | 112,227 |
| | $ | (1,273 | ) | | $ | (79,866 | ) | | $ | 31,128 |
|
Net loss | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (22,799 | ) | | (22,799 | ) |
Foreign currency translation adjustment | | — |
| | — |
| | — |
| | — |
| | — |
| | (229 | ) | | — |
| | (229 | ) |
Stock based compensation expense | | — |
| | — |
| | — |
| | — |
| | 1,905 |
| | — |
| | — |
| | 1,905 |
|
Issuance of common and preferred stock, net of $1,279 in offering costs | | 5 |
| | — |
| | 3,226 |
| | 3 |
| | 15,218 |
| | — |
| | — |
| | 15,221 |
|
Balance as of December 31, 2016 | | 5 |
| | — |
| | 43,059 |
| | 43 |
| | 129,350 |
| | (1,501 | ) | | (102,665 | ) | | 25,227 |
|
Net loss | | — |
| | — |
| | — |
| | — |
| | — |
| | — |
| | (17,647 | ) | | (17,647 | ) |
Foreign currency translation adjustment | | — |
| | — |
| | — |
| | — |
| | — |
| | (2,374 | ) | | — |
| | (2,374 | ) |
Unrealized losses on investments | | — |
| | — |
| | — |
| | — |
| | — |
| | (820 | ) | | — |
| | (820 | ) |
Stock based compensation expense | | — |
| | — |
| | — |
| | — |
| | 3,046 |
| | — |
| | — |
| | 3,046 |
|
Issuance of common stock resulting from exercise of stock options | | — |
| | — |
| | 453 |
| | — |
| | 781 |
| | — |
| | — |
| | 781 |
|
Issuance of common stock resulting from exercise of warrants | | — |
| | — |
| | 1,505 |
| | 2 |
| | 3,307 |
| | — |
| | — |
| | 3,309 |
|
Balance as of December 31, 2017 | | 5 |
| | $ | — |
| | 45,017 |
| | $ | 45 |
| | $ | 136,484 |
| | $ | (4,695 | ) | | $ | (120,312 | ) | | $ | 11,522 |
|
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(In thousands)
|
| | | | | | | | |
| | Years Ended December 31, |
| | 2017 | | 2016 |
Operating activities: | |
| |
|
Net loss | | $ | (17,647 | ) | | $ | (22,799 | ) |
Adjustments to reconcile net loss to net cash provided by (used in) operating activities: | |
| |
|
Depreciation | | 369 |
| | 361 |
|
Stock-based compensation | | 3,046 |
| | 1,905 |
|
Other non-cash transactions | | 293 |
| | 49 |
|
Foreign currency re-measurement loss/(gain)
|
| 156 |
|
| — |
|
Changes in operating assets and liabilities: | |
| |
|
Accounts receivable | | (14,469 | ) | | (59 | ) |
Prepaid expenses and other assets | | 1,900 |
| | (1,020 | ) |
Deferred revenue | | 74,378 |
| | 3,752 |
|
Accounts payable | | (332 | ) | | 1,360 |
|
Accrued expenses and other current liabilities | | 2,060 |
| | 2,063 |
|
Net cash provided by (used in) operating activities | | 49,754 |
| | (14,388 | ) |
Investing activities: | |
| |
|
Purchase of property and equipment | | (1,950 | ) | | (560 | ) |
Purchase of investments | | (43,925 | ) | | — |
|
Net cash used in investing activities | | (45,875 | ) | | (560 | ) |
Financing activities: | |
| |
|
Proceeds from exercise of options | | 781 |
| | — |
|
Proceeds from exercise of warrants | | 3,309 |
| | — |
|
Issuance of Common and Preferred Stock, net of issuance costs | | — |
| | 15,221 |
|
Net cash provided by financing activities | | 4,090 |
| | 15,221 |
|
Effect of exchange rate change on cash and cash equivalents | | 553 |
| | (267 | ) |
Net increase in cash and cash equivalents | | 8,522 |
| | 6 |
|
Cash and cash equivalents at beginning of year | | 29,356 |
| | 29,350 |
|
Cash and cash equivalents at end of year | | $ | 37,878 |
| | $ | 29,356 |
|
Supplemental cash flow disclosures: | |
| |
|
Cash paid for income taxes | | $ | — |
| | $ | 162 |
|
Net unrealized loss on investments | | $ | 820 |
| | $ | — |
|
Property and equipment included in accounts payable | | $ | 114 |
| | $ | 22 |
|
The accompanying notes are an integral part of these consolidated financial statements.
PIERIS PHARMACEUTICALS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Corporate Information
Pieris Pharmaceuticals, Inc., was founded in May 2013, and acquired all interest in Pieris GmbH (formerly Pieris AG, a German company which was founded in 2001) in December 2014. Pieris Pharmaceuticals, Inc. and its consolidated subsidiaries (collectively “Pieris” or the “Company”) is a clinical-stage biopharmaceutical company that discovers and develops Anticalin based drugs to target validated disease pathways in a unique and transformative way. Pieris' corporate headquarters is located in Boston, MA and its research facility is located in Freising-Weihenstephan, Germany.
The Company’s pipeline includes, among other programs, an immuno-oncology multispecific tailored for the tumor micro-environment, an inhaled Anticalin to treat uncontrolled asthma, and a half-life-optimized Anticalin to treat anemia.
The Company’s core Anticalin technology and platform was developed in Germany, and the Company has partnership arrangements with major multi-national pharmaceutical companies headquartered in the U.S., Europe, Japan, and with regional pharmaceutical companies headquartered in India.
As of December 31, 2017, the Company had cash, cash equivalents and investments of $82.6 million. Including the $47.3 million of net proceeds, after expenses, from the sale of common stock in February 2018, and the $30.0 million upfront fee received in March 2018 in connection with a strategic partnership agreement signed in February 2018 (See Note 13 - Subsequent events), the Company expects that its existing cash, cash equivalents, and investments, are sufficient to support operating expenses and capital expenditure requirements into the middle of 2020.
2. Summary of Significant Accounting Policies
Basis of Consolidation
The accompanying consolidated financial statements of Pieris Pharmaceuticals, Inc. and its wholly owned subsidiaries were prepared in accordance with accounting principles generally accepted in the United States (“U.S. GAAP”). The consolidated financial statements include the accounts of all subsidiaries. All intercompany balances and transactions have been eliminated.
Use of Estimates
The preparation of the financial statements in accordance with U.S. GAAP requires management to make estimates, judgments, and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses and the related disclosures at the date of the financial statements and during the reporting period. Significant estimates are used for, but are not limited to, revenue recognition; deferred tax assets, deferred tax liabilities and valuation allowances; fair value of stock options and various accruals. Management evaluates its estimates on an ongoing basis. Actual results and outcomes could differ materially from management’s estimates, judgments, and assumptions.
Foreign Currency Translation
The financial statements of Pieris’ foreign subsidiaries are translated from local currency into reporting currency, which is U.S. dollars, using the current exchange rate at the balance sheet date for assets and liabilities, and the weighted average exchange rate prevailing during the period for revenues and expenses. The functional currency for Pieris’ foreign subsidiaries is considered to be the local currency for each entity and, accordingly, translation adjustments for these subsidiaries are included in accumulated other comprehensive loss within stockholders’ equity.
Realized and unrealized gains and losses resulting from foreign currency transactions denominated in currencies other than the functional currency are reflected as other (expense) income, net in the consolidated statements of operations. Foreign currency gains and losses on available-for-sale investment transactions are recorded to other comprehensive income on our balance sheet per Financial Accounting Standards Board (“FASB”) Accounting Standards Codification (“ASC”) Topic 830, Foreign Currency Matters.
Cash, Cash Equivalents and Investments
The Company determines the appropriate classification of its investments at the time of purchase. All liquid investments with original maturities of 90 days or less from the purchase date are considered to be cash equivalents. The Company’s current and
non-current investments are comprised of money market, asset backed securities, government treasuries, and corporate bonds that are classified as available-for-sale in accordance with FASB ASC Topic 320, Investments—Debt and Equity Securities. The Company classifies investments available to fund current operations as current assets on its balance sheets. Investments are classified as non-current assets on the balance sheets if (i) the Company has the intent and ability to hold the investments for a period of at least one year and (ii) the contractual maturity date of the investments is greater than one year.
Available-for-sale investments are recorded at fair value, with unrealized gains or losses included in accumulated other comprehensive loss on the Company’s balance sheets. Realized gains and losses are determined using the specific identification method and are included as a component of interest income. There were no realized gains or losses recognized for the years ended December 31, 2017 and 2016, respectively.
The Company reviews investments for other-than-temporary impairment whenever the fair value of an investment is less than the amortized cost and evidence indicates that an investment’s carrying amount is not recoverable within a reasonable period of time. To determine whether an impairment is other-than temporary, the Company considers its intent to sell, or whether it is more likely than not that the Company will be required to sell the investment before recovery of the investment’s amortized cost basis. Evidence considered in this assessment includes reasons for the impairment, the severity and the duration of the impairment, and changes in value subsequent to period end. As of December 31, 2017, there were no investments with a fair value that was significantly lower than the amortized cost basis or any investments that had been in an unrealized loss position for a significant period.
Concentration of Credit Risk and Off-Balance Sheet Risk
The Company has no financial instruments with off‑balance sheet risk such as foreign exchange contracts, option contracts, or other foreign hedging arrangements. Financial instruments that subject Pieris to concentrations of credit risk include cash and cash equivalents, investments, and accounts receivable. The Company’s cash, cash equivalents, and investments are held in accounts with financial institutions that management believes are creditworthy. The Company’s investment policy includes guidelines on the quality of the institutions and financial instruments and defines allowable investments that the Company believes minimizes the exposure to concentration of credit risk. These amounts, at times, may exceed federally insured limits. The Company has not experienced any credit losses in such accounts and does not believe it is exposed to any significant credit risk on these funds. Accounts receivable primarily consist of amounts due under strategic partnership and other license agreements with major multi-national pharmaceutical companies for which the Company does not obtain collateral.
Fair Value Measurement
The Company is required to disclose information on all assets and liabilities reported at fair value that enables an assessment of the inputs used in determining the reported fair values. FASB ASC Topic 820, Fair Value Measurement and Disclosures, (“ASC 820”) established a hierarchy of inputs used in measuring fair value that maximizes the use of observable inputs and minimizes the use of unobservable inputs by requiring that the observable inputs be used when available. Observable inputs are inputs that market participants would use in pricing the financial instrument based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the financial instrument and are developed based on the best information available in the circumstances. The fair value hierarchy applies only to the valuation inputs used in determining the reported or disclosed fair value of the financial instruments and is not a measure of the investment credit quality. Fair value measurements are classified and disclosed in one of the following three categories:
| |
• | Level 1 inputs are quoted prices in active markets for identical assets or liabilities that the reporting entity has the ability to access at the measurement date. |
| |
• | Level 2 utilizes quoted market prices in markets that are not active, broker or dealer quotations, or alternative pricing sources with reasonable levels of price transparency. |
| |
• | Level 3 inputs are unobservable inputs for the asset or liability in which there is little, if any, market activity for the asset or liability at the measurement date. |
To the extent that valuation is based on models or inputs that are less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Financial instruments measured at fair value on a recurring basis include cash equivalents and investments (Note 4).
An entity may elect to measure many financial instruments and certain other items at fair value at specified election dates. Subsequent unrealized gains and losses on items for which the fair value option has been elected will be reported in net loss. The Company did not elect to measure any additional financial instruments or other items at fair value.
Fair Values of Financial Instruments
The fair value of cash, accounts receivable, and accounts payable approximates the carrying value of these financial instruments because of the short-term nature of any maturities. The Company determines the estimated fair values of other financial instruments, using available market information and valuation methodologies, primarily input from independent third party pricing sources.
Accounts Receivable
Accounts receivable are recorded net of allowances for doubtful accounts and represent amounts due from third parties, strategic partners, and other license agreement counterparties. The Company monitors and evaluates collectability of receivables on an ongoing basis and considers whether an allowance for doubtful accounts is necessary. The Company determined that no such reserve is needed as of December 31, 2017 and 2016. Historically, Pieris has not had collectability issues.
Property and Equipment
Property and equipment are recorded at acquisition cost, less accumulated depreciation and impairment. Depreciation on property and equipment is calculated using the straight-line method over the remaining estimated useful lives of the assets. Maintenance and repairs to these assets are charged to expenses as occurred. The estimated useful life of the different groups of property and equipment is as follows:
|
| | |
Asset Classification | | Estimated useful life (in years) |
Leasehold improvements | | shorter of useful life or remaining life of the lease |
Laboratory equipment | | 10 - 14 |
Office and computer equipment | | 3 - 13 |
Impairment of Long-lived Assets
Pieris reviews its long-lived assets to be held and used for impairment whenever events or changes in business circumstances indicate that the carrying amount of the assets may not be fully recoverable. Pieris evaluates the realizability of its long-lived assets based on profitability and cash flow expectations for the related asset. Any write-downs are treated as permanent reductions in the carrying amount of the assets. Pieris believes that, as of each of the balance sheets presented, none of Pieris’ long-lived assets were impaired.
Revenue Recognition
Pieris has entered into several licensing and development agreements with collaboration partners for the development of Anticalin® therapeutics against a variety of targets in diseases and conditions. The terms of these agreements contain multiple elements and deliverables, which may include: (i) licenses, or options to obtain licenses, to Pieris’ Anticalin technology and (ii) research and development activities to be performed on behalf of the collaborative partner. Payments to Pieris under these agreements may include upfront fees (which include license and option fees), payments for research and development activities, payments based upon the achievement of certain milestones and royalties on product sales. There are no performance, cancellation, termination or refund provisions in any of the arrangements that could result in material financial consequences to Pieris. Pieris follows the provisions of the FASB ASC Topic 605-25, Revenue Recognition—Multiple-Element Arrangements ("ASC 605-25") and FASB ASC Topic 605-28, Revenue Recognition—Milestone Method ("ASC 605-28") in accounting for these agreements.
Multiple-Element Arrangements
When evaluating multiple-element arrangements, Pieris identifies the deliverables included within the agreement and evaluates which deliverables represent separate units of accounting based on whether the delivered element has stand-alone value to the customer or if the arrangement includes a general right of return for delivered items.
The consideration received is allocated among the separate units of accounting using the relative selling price method, and the applicable revenue recognition criteria are applied to each of the separate units of accounting. Pieris uses the best estimate of selling price (“BESP”) methodology to estimate the selling price for each deliverable and unit of accounting because Pieris does not have vendor specific objective evidence (“VSOE”) or third-party evidence (“TPE”) of selling price for these deliverables. To determine the estimated selling price of a deliverable, Pieris considers market conditions as well as entity-specific factors, including those factors contemplated in negotiating the agreements, terms of previous collaborative agreements, similar agreements entered into by third parties, market opportunity, estimated development costs, probability of success, and the time needed to commercialize a product candidate pursuant to the license. In validating Pieris’ BESP, Pieris evaluates whether changes in the key assumptions used to determine the BESP will have a significant effect on the allocation of arrangement consideration among multiple deliverables.
Multiple element arrangements, such as license and development arrangements, are analyzed to determine whether the deliverables, which often include licenses and performance obligations such as research and development services and steering committee services, can be separated or whether they must be accounted for as a combined unit of accounting in accordance with U.S. GAAP. The Company recognizes the arrangement consideration allocated to licenses as revenue upon delivery of the license only if the license has stand-alone value. If the license is considered not to have stand-alone value, the license would then be combined with other undelivered elements into a combined unit of accounting and the license payments and payments for performance obligations would be recognized as revenue when the revenue recognition criteria have been satisfied for the last deliverable within the unit of accounting. In the case of combined units of accounting that include delivered licenses and undelivered services to be provided over time, revenue would be recognized over the estimated period during which services will be provided. For units of accounting that include licenses to be delivered upon satisfactory completion of certain research services, revenue is deferred until the license is delivered and the performance obligation is satisfied.
If the Company is involved in a steering committee, as part of a multiple element arrangement, it assesses whether its involvement constitutes a performance obligation or a right to participate. When steering committee services are determined to be performance obligations, the Company determines the fair value to be allocated to this deliverable and recognize the revenue over the expected term of the development period of the products. Otherwise, the fair value for participation is combined with other research services or performance obligations and is recognized over the term which the Company expects to complete its aggregate performance obligations.
The Company recognizes arrangement consideration allocated to each unit of accounting when all of the revenue recognition criteria in ASC 605-25 are satisfied for that particular unit of accounting. For each unit of accounting, the Company must determine the period over which the performance obligations will be performed and revenue will be recognized. If there is no discernible pattern of performance or objectively measurable performance measures do not exist, then the Company recognizes revenue under the arrangement on a straight-line basis over the period the Company is expected to complete its performance obligations. Conversely, if the pattern of performance over which the service is provided to the customer can be determined and objectively measurable performance measures exist, then the Company recognizes revenue under the arrangement using the proportional performance method. Revenue recognized is limited to the lesser of the cumulative amount of payments received or the cumulative amount of revenue earned as of the period ending date.
Significant management judgment is required in determining the level of effort required under an arrangement and the period over which the Company is expected to complete its performance obligations under an arrangement.
The accounting treatment for options granted to collaborators is dependent upon the nature of the option granted to the collaborative partner. Options are considered substantive if, at the inception of an agreement, Pieris is at risk as to whether the collaborative partner will choose to exercise the option(s) to secure additional goods or services. Factors that are considered in evaluating whether options are substantive include the overall objective of the arrangement, benefit the collaborator might obtain from the agreement without exercising the options, cost to exercise the options relative to the total upfront consideration, and additional financial commitments or economic penalties imposed on the collaborator as a result of exercising the options.
In arrangements where options to obtain additional deliverables are considered substantive, Pieris determines whether the optional licenses are priced at a significant and incremental discount. If the prices include a significant and incremental discount, the option is considered a deliverable in the arrangement. However, if not priced at a discount, the option is not considered a deliverable in the arrangement. When a collaborator exercises an option considered to be at a significant and incremental discount to acquire an additional license, the exercise fee that is attributed to the additional license and any incremental discount allocated at inception are recognized in a manner consistent with the treatment of up-front payments for licenses (i.e., license and research services). In the event an option expires un-exercised, any incremental discounts deferred at the inception of the arrangement are recognized into revenue upon expiration. For options that are non-substantive, the
additional licenses to which the options pertain are considered deliverables upon inception of the arrangement; Pieris applies the multiple-element revenue recognition criteria to determine accounting treatment.
Payments or reimbursements resulting from Pieris’ research and development efforts in multi-element arrangements, in which Pieris’ research and development efforts are considered to be a deliverable, are included in allocable consideration and allocated to the units of accounting. These reimbursements are recognized as the services are performed and are presented on a gross basis, so long as there is persuasive evidence of an arrangement, the fee is fixed or determinable, and collection of the related receivable is reasonably assured. Revenue recognized cannot exceed the amount that has been earned and has been billed or is currently billable. Amounts received prior to satisfying the above revenue recognition criteria are recorded as deferred revenue in the accompanying balance sheets.
Milestone Payments and Royalties
At the inception of each agreement that includes milestone payments, Pieris evaluates whether each milestone is substantive and at risk to both parties on the basis of the contingent nature of the milestone. This evaluation includes an assessment of whether: (a) the consideration is commensurate with either (1) the entity’s performance to achieve the milestone, or (2) the enhancement of the value of the delivered item(s) as a result of a specific outcome resulting from the entity’s performance to achieve the milestone, (b) the consideration relates solely to past performance and (c) the consideration is reasonable relative to all of the deliverables and payment terms within the arrangement. Pieris evaluates factors such as the scientific, regulatory, commercial and other risks that must be overcome to achieve the respective milestone, the level of effort and investment required to achieve the respective milestone and whether the milestone consideration is reasonable relative to all deliverables and payment terms in the arrangement in making this assessment.
Pieris aggregates milestones into four categories (i) research milestones, (ii) development milestones, (iii) commercial milestones and (iv) sales milestones. Research milestones are typically achieved upon reaching certain success criteria as defined in each agreement related to developing an Anticalin protein against the specified target. Development milestones are typically reached when a compound reaches a defined phase of clinical research or passes such phase, or upon gaining regulatory approvals. Commercial milestones are typically achieved when an approved pharmaceutical product reaches the status for commercial sale, including regulatory approval. Sales milestones are certain defined levels of net sales by the licensee, such as when a product first achieves global sales or annual sales of a specified amount.
For revenues from research, development, and commercial milestone payments, if the milestones are deemed substantive and the milestone payments are nonrefundable, such amounts are recognized entirely upon successful accomplishment of the milestones, assuming all other revenue recognition criteria are met. Milestones that are not considered substantive are accounted for as contingent revenue and will be recognized when achieved to the extent the Company has no remaining performance obligations under the arrangement. Revenues from sales milestone payments are accounted for as royalties and are recorded as revenue upon achievement of the milestone, assuming all other revenue recognition criteria are met. Royalty payments are recognized in revenues based on the timing of royalty payments earned in accordance with the agreements, which typically is the period when the relevant sales occur, assuming all other revenue recognition criteria are met.
Research and Development
Research and development expenses are charged to the statement of operations as incurred. Research and development expenses are comprised of costs incurred in performing research and development activities, including salaries and benefits, facilities costs, pre-clinical and clinical costs, contract services, consulting, depreciation and amortization expense, and other related costs. Costs associated with acquired technology, in the form of upfront fees or milestone payments, are charged to research and development expense as incurred.
Incremental direct cost
Incremental direct costs incurred related to the acquisition or origination of a customer contract in a transaction that results in the deferral of revenue are recorded to the statement of operations as incurred.
Income Taxes
The Company applies ASC Topic 740 Income Taxes, which established financial accounting and reporting requirements for the effects of income taxes that result from the Company’s activities during the current and preceding years. Deferred tax assets and liabilities are recognized for the future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and operating losses and tax credit carry
forwards. Deferred tax assets and liabilities are measured using enacted statutory tax rates expected to apply to taxable income in the jurisdictions and years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that includes the enactment date. Where the Company determines that it is more likely than not that some portion or all of the deferred tax assets will not be realized in the future, the deferred tax assets are reduced by a valuation allowance. The Company records interest related and penalties related to uncertain tax positions as part of income tax expense.
Stock-based Compensation
Pieris measures share-based payments in accordance with ASC Topic 718, Stock Compensation. Pieris records its stock-based compensation expense over the requisite service period. Determining the appropriate fair value model and related assumptions requires judgment, including estimating share price volatility and expected terms of the awards. For employee options, the fair value measurement date is generally on the date of grant and the related compensation expense is recognized on a straight-line basis over the requisite period of the awards, less expense for actual forfeitures.
The Company uses the Black-Scholes option pricing model to determine the estimated fair value for stock-based awards. Option-pricing models require the input of various subjective assumptions, including the option’s expected life, expected dividend yield, price volatility, risk free interest rate and forfeitures of the underlying stock. Due to the limited operating history of the Company as a public entity and a lack of company specific historical and implied volatility data, the Company has based its estimate of expected volatility on the historical volatility of a group of similar companies that are publicly traded. When selecting these public companies on which it has based its expected stock price volatility, the Company selected companies with comparable characteristics to it, including enterprise value, risk profiles, position within the industry, and with historical
share price information sufficient to meet the expected term of the stock-based awards. The Company computes historical volatility data using the daily closing prices for the selected companies’ shares during the equivalent period of the calculated expected term of the stock-based awards. The Company will continue to apply this process until a sufficient amount of historical information regarding the volatility of its own stock price becomes available.
Due to the lack of Company specific historical option activity, the Company has estimated the expected term of its employee stock options using the “simplified” method, whereby, the expected term equals the arithmetic average of the vesting term and the original contractual term of the option. The expected term for non-employee awards is the remaining contractual term of the option. The risk-free interest rates are based on the U.S. Treasury securities with a maturity date commensurate with the expected term of the associated award. The Company has never paid, and does not expect to pay dividends in the foreseeable future.
The Company early adopted ASU 2016-09 Compensation - Stock Compensation (Topic 718) as of January 1, 2016. In connection with the early adoption, the Company elected an accounting policy to record forfeitures as they occur. There was no material financial statement impact upon adoption. ASU 2016-09 also provides that companies no longer record excess tax benefits or certain tax deficiencies in additional paid-in capital. Instead, all excess tax benefits and tax deficiencies are recorded as income tax expense or benefit in the Company's statement of operations and comprehensive loss. There was no financial statement impact of adopting this provision of ASU 2016-09 as there were no options previously exercised. As such, the adoption of this standard did not have a material impact on the financial statements. For the years ended December 31, 2017 and 2016, the Company did not record an income statement benefit for excess tax benefits as a valuation allowance is also required on these amounts.
Contingencies
Accruals are recorded for loss contingencies when it is probable that a liability has been incurred and the amount of the related loss can be reasonably estimated. Pieris evaluates, on a quarterly basis, developments in legal proceedings and other matters that could cause an increase or decrease in the amount of the liability that has been accrued previously. Considering facts known at the time of the assessment, Pieris determines whether potential losses are considered reasonably possible or probable and whether they are estimable. Based upon this assessment, Pieris carries out an evaluation of disclosure requirements and considers possible accruals in the financial statements.
Segment Reporting
Operating segments are identified as components of an enterprise where separate discrete financial information is available for evaluation by the chief operating decision maker in making decisions on how to allocate resources and assess performance. Pieris operates as a single segment dedicated to the discovery and development of biotechnological applications and the
Company’s chief operating decision maker (“CODM”) makes decisions based on the Company as a whole. The Company has determined that its CODM is its Chief Executive Officer.
Earnings per Share
Basic earnings per share attributable to common stockholders is calculated by dividing net loss attributable to common stockholders by the weighted average shares outstanding during the period, without consideration for common stock equivalents.
Diluted earnings per share attributable to common stockholders is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share attributable to common stockholders' calculation, preferred stock, stock options, unvested restricted stock, and warrants are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share attributable to common stockholders, as their effect would be anti-dilutive for all periods presented. Therefore, basic and diluted net loss per share were the same for all periods presented.
Recent Accounting Pronouncements
Standards not yet adopted
In May 2014, the FASB issued Accounting Standard Update (“ASU”) No. 2014-09, Revenue from Contracts with Customers (Topic 606) (“ASU 2014-09”). Subsequently, the FASB also issued ASU 2015-14, Revenue from Contracts with Customers (Topic 606) , which adjusted the effective date of ASU 2014-09; ASU No. 2016-08, Revenue from Contracts with Customers (Topic 606): Principal versus Agent Considerations (Reporting Revenue Gross versus Net), which amends the principal-versus-agent implementation guidance and illustrations in ASU 2014-09; ASU No. 2016-10, Revenue from Contracts with Customers (Topic 606): Identifying Performance Obligations and Licensing, which clarifies identifying performance obligation and licensing implementation guidance and illustrations in ASU 2014-09; and ASU No. 2016-12, Revenue from Contracts with Customers (Topic 606): Narrow-Scope Improvements and Practical Expedients, which addresses implementation issues and is intended to reduce the cost and complexity of applying the new revenue standard in ASU 2014-09 (collectively, the “Revenue ASUs”).
The Revenue ASUs provide an accounting standard for a single comprehensive model for use in accounting for revenue arising from contracts with customers and supersedes most current revenue recognition guidance. The accounting standard is effective for public emerging growth companies ("EGC"), like Pieris, for interim and annual periods beginning after December 15, 2018, with an option to early adopt for interim and annual periods beginning after December 15, 2017. The guidance permits two methods of adoption: retrospectively to each prior reporting period presented (the full retrospective method), or retrospectively with the cumulative effect of initially applying the guidance recognized at the date of initial application (the modified retrospective method). The Company is in the process of determining the date and method of adoption and the impact of the Revenue Recognition ASUs on its financial statements.
In February 2016, the FASB issued ASU No. 2016-2, Leases (Topic 842) ("ASU 2016-2"). Under the amendments in ASU 2016-2, lessees will be required to recognize (i) a lease liability, which is a lessees obligation to make lease payments arising from a lease, measured on a discounted basis; and (ii) a right-of-use asset, which is an asset that represents the lessee’s right to use, or control the use of, a specified asset for the lease term for all leases (with the exception of short-term leases) at the commencement date. This guidance is effective for fiscal years beginning after December 15, 2019 including interim periods within those fiscal years. Early adoption is permitted. The Company is currently evaluating the potential impact the adoption of this standard will have on its financial statements and related disclosures.
In May 2017, the FASB issued ASU No. 2017-09, Compensation - Stock compensation (Topic 718) (“ASU 2017-09”). The amendments in this update provide guidance about which changes to the terms or conditions of a share-based payment award require an entity to apply modification accounting. This ASU will be effective for the Company’s fiscal year beginning January 1, 2018, and the Company is not expecting a material impact the adoption will have on its financial statements.
Pieris has considered other recent accounting pronouncements and concluded that they are either not applicable to the business, or that the effect is not expected to be material to the unaudited condensed consolidated financial statements as a result of future adoption.
3. Revenue
General
The Company has not generated revenue from product sales. The Company has generated revenue pursuant to (i) license and collaboration agreements, which include upfront payments for licenses or options to obtain licenses, payments for research and development services and milestone payments, and (ii) government grants. During the years ended December 31, 2017 and 2016, respectively, the Company recognized revenues as follows (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
License fees | $ | 11,285 |
| | $ | 2,736 |
|
Research and development services | 1,403 |
| | 1,440 |
|
Milestone payments | 12,573 |
| | 1,655 |
|
Other revenues | 14 |
| | — |
|
Total Revenue | $ | 25,275 |
| | $ | 5,831 |
|
During the years ended December 31, 2017 and 2016, respectively, the Company recognized revenue from the following strategic partnerships and other license agreements (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
AstraZeneca | $ | 19,769 |
| | $ | — |
|
Servier | 1,907 |
| | — |
|
Roche | 3,516 |
| | 4,080 |
|
Other | 83 |
| | 1,751 |
|
Total Revenue | $ | 25,275 |
| | $ | 5,831 |
|
Under the Company´s existing strategic partnerships and other license agreements, Pieris could receive the following potential milestone payments (in millions):
|
| | | | | | | |
| Research, Development & Regulatory Milestones | | Sales Milestones |
AstraZeneca | $ | 1,111 |
| | $ | 960 |
|
Servier | 1,040 |
| | 942 |
|
Roche | 290 |
| | 123 |
|
Other | 68 |
| | — |
|
Total potential milestone payments | $ | 2,509 |
| | $ | 2,025 |
|
Strategic Partnerships and Other License Agreements
AstraZeneca AB
On May 2, 2017, Pieris entered into a License and Collaboration Agreement (“AstraZeneca Collaboration Agreement”), and a Non-Exclusive Anticalin Platform Technology License Agreement (the “License Agreement” and together with the AstraZeneca Collaboration Agreement, the “AstraZeneca Agreements”) with AstraZeneca AB (“AstraZeneca”), which became effective on June 10, 2017, following expiration of the waiting period under the Hart-Scott-Rodino Antitrust Improvements Act of 1976. Under the AstraZeneca Agreements the parties will advance several novel inhaled Anticalin proteins.
In addition to the Company’s lead inhaled drug candidate, PRS-060 (the “AstraZeneca Lead Product”), Pieris and AstraZeneca will also collaborate to progress four additional novel Anticalin proteins against undisclosed targets for respiratory diseases (the “AstraZeneca Collaboration Products” and together with the AstraZeneca Lead Product, the “AstraZeneca Products”). Pieris is responsible for advancing the AstraZeneca Lead Product through the Phase I trial, with the associated costs funded by AstraZeneca. The parties will collaborate thereafter to conduct a Phase IIa clinical trial in asthma patients, with AstraZeneca continuing to fund development costs. After completion of the Phase IIa trial, Pieris has the option to co-develop the AstraZeneca Lead Product and also has the option to co-commercialize the AstraZeneca Lead Product in the United States. For the four AstraZeneca Collaboration Products, Pieris will be responsible for the initial discovery of the novel Anticalin proteins, after which AstraZeneca will take the lead on continued development. Pieris has the option to co-develop two of these four
AstraZeneca Collaboration Products beginning at a pre-defined preclinical stage and would also have the option to co-commercialize these two programs in the United States, while AstraZeneca will be responsible for development and commercialization of the other programs worldwide.
The term of the AstraZeneca Agreement ends upon the expiration of all of AstraZeneca’s payment obligations under such AstraZeneca Agreement. The AstraZeneca Collaboration Agreement may be terminated by AstraZeneca in its entirety for convenience beginning 12 months after its effective date upon 90 days’ notice or, if Pieris has obtained marketing approval for the marketing and sale of a product, a 180 days’ notice. Each program may be terminated at AstraZeneca’s option, and if any program is terminated by AstraZeneca, Pieris will have full rights to such program. The AstraZeneca Collaboration Agreement may also be terminated by AstraZeneca or Pieris for material breach upon 180 days’ notice of a material breach (or 30 days with respect to payment breach), provided that the applicable party has not cured such breach by the permitted cure period (including an additional 180 days if the breach is not susceptible to cure during the initial 180-day period) and dispute resolution procedures specified in the applicable AstraZeneca Agreement have been followed. The AstraZeneca Collaboration Agreement may also be terminated due to the other party’s insolvency and may in certain instances be terminated on a product-by-product and/or country-by-country basis. Each party may also terminate the agreement if the other party challenges the validity of patents related to certain intellectual property licensed under the AstraZeneca Agreement, subject to certain exceptions for infringement suits, acquisitions and newly-acquired licenses. The License Agreement will terminate upon termination of the AstraZeneca Collaboration Agreement, on a product-by-product and/or country-by-country basis.
At inception, AstraZeneca is granted the following licenses: (i) research and development license for the AstraZeneca Lead Product, (ii) commercial license for the AstraZeneca Lead Product, (iii) individual research licenses for each of the four AstraZeneca Collaboration Products, (iv) individual commercial licenses for each of the four AstraZeneca Collaboration Products, and (v) individual non-exclusive platform technology licenses for the AstraZeneca Lead Product and the four AstraZeneca Collaboration Products. AstraZeneca will be granted individual development licenses for each of the four AstraZeneca Collaboration Products upon completion of the initial discovery of Anticalin proteins.
The collaboration will be managed on an overall basis by a Joint Steering Committee (“JSC”) formed by an equal number of representatives from the Company and AstraZeneca. In addition to the JSC, the AstraZeneca Collaboration Agreement also requires each party to designate an Alliance Manager to facilitate communication and coordination of the Parties activities under that AstraZeneca Agreement, as well as requires participation of both parties on: (i) a Joint Development Committee (“JDC”) and (ii) a Commercialization Committee. The responsibilities of these committees vary, depending on the stage of development and commercialization of each Product.
Under the AstraZeneca Agreements, the Company received an upfront, non-refundable payment of $45.0 million. In addition, the Company will receive payments to conduct a Phase I trial for the AstraZeneca Lead Product. The Company is also eligible to receive research, development, commercial, sales milestone payments, and royalty payments. The Company may receive tiered royalties on sales of potential products commercialized by AstraZeneca and for co-developed products, gross margin share on worldwide sales equal dependent on Pieris’ level of committed investment.
The Company accounted for the AstraZeneca Agreements as a multiple element arrangement under ASC 605-25. The arrangement with AstraZeneca contains the following initial deliverables: (i) five non-exclusive platform technology licenses, (ii) research and development license for the AstraZeneca Lead Product, (iii) commercial license for the AstraZeneca Lead Product, (iv) development and manufacturing services for the AstraZeneca Lead Product, (v) technology transfer services for the AstraZeneca Lead Product, (vi) research services related to the AstraZeneca Lead Product, (vii) participation on each of the committees, (viii) four research licenses for the AstraZeneca Collaboration Products, (ix) four commercial licenses for the AstraZeneca Collaboration Products, and (x) research services for the AstraZeneca Collaboration Products. Additionally, as the development licenses on the four AstraZeneca Collaboration Products may be granted at a discount in the future, the Company determined such discounts be included as an element of the arrangement at inception.
Management considered whether any of the deliverables could be considered separate units of accounting. The Company determined that the licenses granted for the AstraZeneca Lead Product at the inception of the arrangement did not have standalone value from the research services related to the AstraZeneca Lead Product and the licenses granted for the AstraZeneca Collaboration Products did not have standalone value from the research services for the AstraZeneca Collaboration Products, due to the specific nature of the intellectual property and knowledge required to perform the services. The Company also determined that the licenses granted at the inception of the arrangement did have standalone value from the development and manufacturing services for the AstraZeneca Lead Product, but that the participation on the various committees did have standalone value as the development and manufacturing services and committee service could be performed by an outside party. The Company determined that the commercial licenses for the AstraZeneca Collaboration Products granted at the inception of the arrangement did not have standalone value from the development licenses for the
AstraZeneca Collaboration Products as the company would not benefit from the commercial license without the ability to develop each product.
As a result, management concluded that there were eleven units of accounting at the inception of the AstraZeneca Agreements: (i) combined unit of accounting representing a non-exclusive platform technology license, research and development license, and commercial licenses for the AstraZeneca Lead Product and research services for the AstraZeneca Lead Product, (ii) combined unit of accounting representing development and manufacturing services, and technology transfer services for the AstraZeneca Lead Product, (iii) committee participation, (iv-vii) four units of accounting representing a combined non-exclusive platform technology license, research licenses, and research services for each AstraZeneca Collaboration Product, and (viii-xi) four units of accounting representing the combined commercial licenses granted for the AstraZeneca Collaboration Products and corresponding discounts on the development licenses granted for the AstraZeneca Collaboration Products upon the achievement of specified preclinical activities.
The Company determined that neither VSOE nor TPE is available for any of the units of accounting identified at the inception of the arrangement. Accordingly, the selling price of each unit of accounting was developed using management’s BESP. The Company developed the BESP for licenses and corresponding research services by applying a risk adjusted, net present value, estimate of future potential cash flow approach, which included the cost of obtaining research services at arm’s length from a third-party provider, as well as internal full time equivalent costs to support these services. The Company developed the BESP for development and manufacturing services, and technology transfer services for the AstraZeneca Lead Product using estimated internal and external costs to be incurred.
The Company developed the BESP for committee participation by using management’s best estimate of the anticipated participation hours multiplied by a market rate for comparable participants.
The Company developed the BESP for the commercial licenses and discounts granted on the development licenses by probability weighting multiple cash flow scenarios using the income approach.
Allocable arrangement consideration at inception is comprised of the $45.0 million upfront fee and the $8.2 million estimated development and manufacturing services to be reimbursed for the AstraZeneca Lead Product. The aggregate allocable consideration of $53.2 million is allocated among the separate units of accounting using the relative selling price method.
The amounts allocated to the unit of accounting for the AstraZeneca Lead Product and four units of accounting for the four AstraZeneca Collaboration Products will be recognized on a proportional performance basis as the activities are conducted over the life of the arrangement. The amounts allocated to the development and manufacturing services, and technology transfer services for the AstraZeneca Lead Product will be recognized on a proportional performance basis over the estimated term of development through Phase IIa trial. The amounts allocated to the participation on each of the committees will be recognized on a straight-line basis over the expected term of development of the AstraZeneca Lead Product and the AstraZeneca Collaboration Products. The term of performance at the inception of the arrangement is approximately five years. The amounts allocated to the commercial licenses and discounts on the development licenses granted in the future for the AstraZeneca Collaboration Products will be recognized upon delivery of each of the development licenses assuming all other revenue recognition criteria have been met. Additionally, the Company evaluated payments required to be made between both parties as a result of the shared development costs of the AstraZeneca Lead Product and the two AstraZeneca Collaboration Products for which Pieris has a co-development option. The Company will classify payments made as a reduction of revenue and will classify payments received as revenue, in the period they are earned.
Under the AstraZeneca Agreements the Company is eligible to receive various research, development, commercial, and sales milestones. Management determined certain of the research, development, and commercial milestones that may be received under the AstraZeneca Agreements are substantive when the Company is involved in the development and commercialization of the applicable AstraZeneca Products. Payment related to achievement of such milestones, if any, will be recognized as revenue when the milestone is achieved. Total potential substantive development milestones range from $72.2 million to $611.4 million, dependent on the Company’s decision, on a product-by-product basis, whether to co-develop the AstraZeneca Lead Product and AstraZeneca Collaboration Products. Research, development, and commercial, and sales milestones are deemed non-substantive if they are based solely on the performance of another party. Non-substantive milestones will be treated as contingent revenue and will be recognized when achieved to the extent the Company has no remaining performance obligations under the arrangement. Total potential non-substantive research, development, and commercial milestones range from $366.2 million to $1.1 billion. The Company may receive lower research, development, and commercial, milestones if the Company chooses to co-develop the AstraZeneca Lead Product and/or AstraZeneca Collaboration Products, depending on the level of co-development investment. Total potential sales milestones are up to $1.0 billion and will be recognized when earned, assuming all other revenue recognition revenue criteria have been met.
The Company will recognize royalty and gross margin share revenue in the period of sale of the related AstraZeneca Product, based on the underlying contract terms, provided that the reported sales are reliably measurable and the Company has no remaining performance obligations, assuming all other revenue recognition criteria are met.
The successful initiation of a Phase I study during the year ended December 31, 2017 resulted in a milestone achievement of $12.6 million, which was recorded as revenue in the respective period. As of December 31, 2017, there is $28.6 million and $16.3 million of deferred revenue and non-current deferred revenue, respectively, related to the AstraZeneca Agreements.
ASKA Pharmaceutical Co. Ltd.
On February 27, 2017 the Company entered into an Exclusive Option Agreement (the “Aska Agreement”) with ASKA Pharmaceutical Co., Ltd. (“Aska”) to grant Aska an option to acquire (1) a non-exclusive license to certain intellectual property rights associated with the Pieris’ Anticalin platform (“Licensed Platform IP”) and (2) an exclusive license to certain intellectual property rights specifically related to Pieris’ PRS-080 Anticalin protein (“Licensed Product IP”) in order to develop, manufacture, import, sale, export, and offer for sale and export any pharmaceutical formulation containing PRS-080, the Company’s PEGylated Anticalin protein targeting hepcidin (“Licensed Product”) in Japan and certain other Asian territories (“Licensed Territory”).
Pieris is obliged to use commercially reasonable efforts to complete the Phase IIa Study for PRS-080 and to submit to Aska, in writing, the final results of the study when available. Upon receipt, Aska will have 60 days to evaluate the results of the Phase IIa study (“Evaluation Period”). Aska agreed to notify Pieris, in writing, of its decision to exercise its option to acquire rights to the Licensed Product. If the Phase IIa Study meets the applicable success criteria and Aska fails to provide notification that it will exercise its option, Aska shall pay the Company an additional fee within thirty days of the end of the Evaluation Period (the “Break-Up Fee”). If Aska exercises the option, Aska and the Company will enter into a separate definitive arrangement governing the future development and commercialization activities.
The Company determined that the completed Phase IIa study represents the single deliverable (and the sole unit of account) under the Aska Arrangement for which an upfront payment of $2.75 million was received from Aska. While the completion of the Phase IIa trial requires the completion of a number of actions, the Company determined that the significance in value of the finalization of the data and evaluation of results of the Phase IIa study is the point at which revenue would be recognized. Therefore, no revenue was recognized in connection with this arrangement for the years ended December 31, 2017 and 2016, respectively. As of December 31, 2017, there is $3.1 million of current deferred revenue related to the Company’s option agreement with Aska.
Les Laboratoires Servier and Institut de Recherches Internationales Servier
On January 4, 2017, Pieris entered into a License and Collaboration Agreement (“Servier Collaboration Agreement”), and Non-Exclusive Anticalin Platform Technology Agreement (the “License Agreement” and together with the Servier Collaboration Agreement, the “Servier Agreements”) with Les Laboratoires Servier and Institut de Recherches Internationales Servier (collectively “Servier”) pursuant to which Pieris and Servier will initially pursue five bispecific therapeutic programs, led by the PRS-332 program (the “Lead Product”), a PD-1-targeting bispecific checkpoint inhibitor. Pieris and Servier will jointly develop PRS-332 and split commercial rights geographically, with Pieris retaining all commercial rights in the United States and Servier having commercial rights in the rest of the world. Each party is responsible for an agreed upon percentage of shared costs, as set forth in the budget for the joint development plan, and as further discussed below.
Four additional committed programs have been defined, which may combine antibodies from the Servier portfolio with one or more Anticalin proteins based on Pieris’ proprietary platform to generate innovative immuno-oncology bispecific drug candidates (“Collaboration Products”). The Servier Collaboration Agreement may be expanded by up to three additional therapeutic programs. Pieris has the option to co-develop and retain commercial rights in the United States for up to three programs beyond the Lead Product (“Co-Development Collaboration Products”), while Servier will be responsible for development and commercialization of the other programs worldwide (“Servier Worldwide Collaboration Products”). Each party is responsible for an agreed upon percentage of shared costs, as set forth in the budget for the collaboration plan, and further discussed below.
Co-Development Collaboration Products may be jointly developed, according to a collaboration plan, through marketing approval from the U.S. Food and Drug Administration (“FDA”) or European Medicines Agency (“EMA”). Servier Worldwide Collaboration Products may be jointly developed, according to a collaboration plan, through specified preclinical activities, at which point Servier becomes responsible for further development of the collaboration product.
At inception, Servier was granted the following licenses: (i) development license for the Lead Product, (ii) commercial license for the Lead Product, (iii) individual research licenses for each of the four Collaboration Products, and (iv) individual non-exclusive platform technology licenses for each of the Lead Product and four Collaboration Products. Upon achievement of certain development activities, specified by the collaboration for each Servier Collaboration Agreement, Servier will be granted a development license and a commercial license. For the Lead Product and Co-Development Collaboration Products, the licenses granted are with respect to the entire world except for the United States. For Servier Worldwide Collaboration Products, the licenses granted are with respect to the entire world.
The Servier Agreements will be managed on an overall basis by a joint executive committee (“JEC”) formed by an equal number of members from the Company and Servier. Decisions by the JEC will be made by consensus, however, in the event of a disagreement, each party will have final-decision making authority as it relates to the applicable territory in which such party has commercialization rights for the applicable product. In addition to the JEC, the Collaboration Agreement requires the participation of both parties on: (i) a JSC, (ii) a JDC, (iii) a Joint Intellectual Property Committee (“JIPC”), and (iv) a Joint Research Committee (“JRC”). The responsibilities of these committees vary, depending on the stage of development and commercialization of the Lead Product and each of the Collaboration Products.
For the Lead Product and Co-Development Collaboration Products, Pieris and Servier are responsible for an agreed upon percent of the shared costs required to develop the products through commercialization. In the event that Pieris fails to exercise their option to co-develop the Co-Development Collaboration Products, Servier has the right to continue with the development and will be responsible for all costs required to develop the products through commercialization.
Under the Servier Agreements, the Company received an upfront, non-refundable payment of €30.0 million (approximately $32.0 million). In addition, the Company is eligible to receive research, development, commercial, and sales milestone payments as well as tiered royalties up to low double digits on the sales of commercialized products in the Servier territories.
The initial research collaboration term, as it relates to the Lead Product and Collaboration Products, shall continue for three years from the effective date, and may be mutually extended for two one-year terms consecutively applied. The term of the Servier Agreements ends upon the expiration of all Servier’s payment obligations under the respective Servier Agreements.
The term of the individual Servier Agreements end upon the expiration of all of Servier’s payment obligations under such Servier Collaboration Agreement. The Servier Collaboration Agreements may be terminated by Servier for convenience beginning 12 months after their effective date upon 180 days’ notice. The Servier Collaboration Agreements may also be terminated by Servier or Pieris for material breach upon 90 days’ or 120 days’ notice of a material breach, with respect to the Servier Collaboration Agreement and License Agreement, respectively, provided that the applicable party has not cured such breach by the applicable 90-day or 120-day permitted cure period, and dispute resolution procedures specified in the applicable Servier Collaboration Agreement have been followed. The Servier Collaboration Agreements may also be terminated due to the other party’s insolvency or for a safety issue and may in certain instances be terminated on a product-by-product and/or country-by-country basis. The License Agreement will terminate upon termination of the Servier Collaboration Agreement, on a product-by-product and/or country-by-country basis.
The Company accounted for the Servier Agreements as a multiple element arrangement under ASC 605-25. The arrangement with Servier contains the following initial deliverables: (i) five non-exclusive platform technology licenses, (ii) development license for the Lead Product, (iii) commercial license for the Lead Product, (iv) research and development services for the Lead Product, (v) participation on each of the committees, (vi) four research licenses for Collaboration Products, and (vii) research and development services for the Collaboration Products. Additionally, as the development and commercial licenses on the four Collaboration Products may be granted at discount in the future, the Company determined such discounts be included as an element of the arrangement at inception.
Management considered whether any of the deliverables could be considered separate units of accounting. The Company determined the licenses granted, at arrangement inception, did not have standalone value from the research and development services to be provided for the Lead Product and Collaboration Products, over the term of the Servier Agreements, due to the specific nature of the intellectual property and knowledge required to perform the research and development services. The Company determined that the participation on the various committees did have standalone value from the delivered licenses as the services could be performed by an outside party.
As a result, management concluded there are ten units of accounting at inception of the agreement: (i) combined unit of accounting representing a non-exclusive platform technology license, commercial license, development license and research and development services for the Lead Product, (ii) four units of accounting each representing a combined non-exclusive platform technology license, research license, and research and development services for each Collaboration Product (iii) one
unit of accounting representing the participation of the various governance committees, and (iv) four units of accounting representing the discounts on the development and commercial licenses granted for the Collaboration Products upon the achievement of specified preclinical activities.
The Company determined that neither VSOE nor TPE is available for any of the units of accounting identified at arrangement inception. Accordingly, the selling price of each unit of accounting was developed using BESP. The Company developed its best estimate of selling price for licenses by applying a risk adjusted, net present value, estimate of future potential cash flows approach, which included the cost of obtaining research and development services at arm’s length from a third-party provider, as well as internal full time equivalent costs to support these services.
The Company developed the BESP for committee participation by using management’s best estimate of the anticipated participation hours multiplied by a market rate for comparable participants.
The Company developed the BESP for the discounts granted on the licenses by probability weighting multiple cash flow scenarios using the income approach.
Allocable arrangement consideration at inception is comprised of the upfront fee of €30.0 million (approximately $32.0 million) and was allocated among the separate units of accounting using the relative selling price method.
The amounts allocated to the combined unit of accounting for the Lead Product and four units of accounting for the four Collaboration Products will be recognized on a proportional performance basis as the activities are conducted over the life of the arrangement. The term of the performance at inception of the agreement for the Lead Product and each of the Co-Developed Collaboration Products may be through approval of certain regulatory bodies; a period which could be many years. The term of the performance at inception of the agreement for each of the other two Servier Worldwide Collaboration Products is approximately two to three years. The amounts allocated to the participation on each of the committees will be recognized ratably over the anticipated performance period over the entirety of the arrangement with Servier. The amounts allocated to the discounts of the development and commercial licenses granted in the future will be recognized upon delivery of each of the licenses assuming no other performance obligations.
Additionally, the Company evaluated payments required to be made between both parties as a result of the shared development costs of the Lead Product and Co-Development Collaboration Products. The Company will classify payments made as a reduction of revenue and will classify payments received as revenue, in the period they are earned.
Under the Servier Agreements the Company is eligible to receive various research, development, commercial, and sales milestones. Management determined certain research, development and commercial milestones, which may be received under the Servier Agreements, are substantive when the Company is involved in the development and commercialization of the applicable product. Payments related to the achievement of such milestones, if any, will be recognized as revenue when the milestone is achieved. Total potential substantive research, development and commercial milestones are up to €163.0 million. Research, development, and commercial milestones are deemed non-substantive if they are based solely on the performance of another party. Non-substantive milestones will be treated as contingent revenue and will be recognized when achieved, to the extent the Company has no remaining performance obligations under the arrangement. Milestone payments earned upon the achievement of sales events will be recognized when earned.
The Company will recognize royalty revenue in the period of sale for the related product(s), based on the underlying contract terms, provided that the reported sales are reliably measurable and the Company has no remaining performance obligations, assuming all other revenue recognition criteria are met.
As of December 31, 2017, there is $ 4.1 million and $30.2 million of deferred revenue and non-current deferred revenue, respectively, related to the Servier Agreements.
F. Hoffmann-La Roche Ltd and Hoffmann- La Roche Inc.
In December 2015, the Company entered into a Research Collaboration and License Agreement (the “Roche Agreement”) with F. Hoffmann- La Roche Ltd. and Hoffmann- La Roche Inc., (“Roche”), for the research, development, and commercialization of Anticalin-based drug candidates against a predefined, undisclosed target in cancer immune therapy. The parties will jointly pursue a preclinical research program, targeting the identification and generation of Anticalin proteins that bind to a specific target, for an expected period of 20 months, which may be extended by Roche for up to an additional 12 months. Roche has the ability to continue exclusivity rights for up to an additional 5 years. Both Roche and the Company will participate in a joint research committee in connection with this agreement. Following the research program, Roche will be responsible for
subsequent pre-clinical and clinical development of any product developed through the research plan and will have worldwide commercialization rights to any such product.
Effective September 28, 2017, Roche exercised their option to extend the initial period for the research program until April 30, 2018. Effective November 20, 2017, a second amendment to the Roche Agreement provided Roche the option to extend this period until up to August 31, 2018, which it did on February 26, 2018.
The Company received $6.5 million upfront upon entering the Roche Agreement. Additionally, Roche will pay Pieris for research services provided by Pieris in conjunction with the research program. Roche will also pay Pieris for certain milestones relating to development, regulatory, and sales milestones, as they are achieved. Revenue recognition is associated with the portion of the research services performed during the periods as well as the value of research services provided by Pieris in connection with the ongoing research program. As of December 31, 2017, $1.5 million of deferred revenue remains related to the Roche Agreement.
The Company identified the research and commercial licenses, performance of R&D services, and participation in the joint research committee as deliverables under the Roche Agreement. For revenue recognition purposes, management determined that there are two units of accounting at the inception of the agreement representing (i) the research and commercial licenses and the performance of R&D services which do not have standalone value, and (ii) the participation in the joint research committee. The upfront consideration received was allocated to the units of accounting and is being recognized on a proportional performance basis as the activities are conducted over the life of the Roche Agreement.
Management determined that the development and regulatory milestones are not substantive because they do not relate solely to past performance of the Company and the Company’s involvement in the achievement is limited to progress reports and other updates. Non-substantive milestones will be recognized when achieved to the extent the Company has no remaining performance obligations under the arrangement.
Other Collaborations
The Company has entered into several other research and collaboration agreements for which the Company could achieve future milestone payments. For revenue recognition purposes, management determined these milestones to be substantive in accordance with applicable accounting guidance related to milestone revenue. Substantive uncertainty existed at the inception of the arrangements as to whether the milestones would be achieved because of the numerous variables, such as the high rate of failure inherent in research and development activities and the uncertainty involved with obtaining regulatory approval. No milestones or other revenues related to these agreements were recognized during the year ended December 31, 2017. For the year ended December 31, 2016, the Company recognized a milestone payment under one of these other agreements, which is based on the initiation of a toxicity study in non-human primates.
4. Cash, cash equivalents and investments
As of December 31, 2017, cash, cash equivalents and investment comprised funds in depository, money market accounts, U.S. treasury securities, asset backed securities, and corporate bonds. None of these cash equivalents and investments existed as of December 31, 2016. The following table presents the cash equivalents and investments carried at fair value in accordance with the hierarchy defined in Note 2 (in thousands):
|
| | | | | | | | | | | | |
| Total | Quoted prices in active markets (Level 1) | Significant other observable inputs (Level 2) | Significant unobservable inputs (Level 3) |
December 31, 2017 | | | | |
Money market funds, included in cash equivalents | $ | 4,583 |
| $ | 4,583 |
|
|
| $ | — |
|
Corporate bonds, included in cash equivalents | 13,595 |
|
|
| 13,595 |
| — |
|
Investments - U.S. treasuries | 4,172 |
| 4,172 |
|
|
| — |
|
Investments - Asset-backed securities | 6,384 |
|
|
| 6,384 |
| — |
|
Investments - Corporate bonds | 34,117 |
|
|
| 34,117 |
| — |
|
Total | $ | 62,851 |
| $ | 8,755 |
| $ | 54,096 |
| $ | — |
|
Cash equivalents and marketable securities have been initially valued at the transaction price and subsequently valued, at the end of each reporting period, utilizing third party pricing services or other market observable data. The pricing services utilize
industry standard valuation models, including both income and market based approaches and observable market inputs to determine value. The Company validates the prices provided by its third-party pricing services by reviewing their pricing methods and obtaining market values from other pricing sources, as needed. After completing its validation procedures, the Company did not adjust any fair value measurements provided by the pricing services as of December 31, 2017.
Cash equivalents and investments at December 31, 2017 consist of the following (in thousands):
|
| | | | | | | | | | | | | |
| Contractual maturity
(in days) | Amortized Cost | Unrealized gains | Unrealized losses | Fair Value |
Investments | | | | | |
U.S. treasuries | 212-340 | $ | 4,287 |
| $ | — |
| $ | (115 | ) | $ | 4,172 |
|
Asset-backed securities | 197-365 | 2,709 |
| — |
| (14 | ) | 2,695 |
|
Asset-backed securities | greater than 365 | 3,798 |
| — |
| (110 | ) | 3,688 |
|
Corporate bonds | 91-365 | 28,356 |
| — |
| (473 | ) | 27,882 |
|
Corporate bonds | greater than 365 | 6,344 |
| 1 |
| (108 | ) | 6,236 |
|
Total | | $ | 45,494 |
| $ | 1 |
| $ | (820 | ) | $ | 44,673 |
|
5. Property and equipment, net
Property and equipment are summarized as follows (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
Laboratory equipment | $ | 6,101 |
| | $ | 3,869 |
|
Office and computer equipment | 494 |
| | 499 |
|
Leasehold improvements | 318 |
| | 321 |
|
Property and equipment at cost | 6,913 |
| | 4,689 |
|
Accumulated depreciation | (2,879 | ) | | (2,425 | ) |
Property and equipment, net | $ | 4,034 |
| | $ | 2,264 |
|
Depreciation expense was $0.4 million and $0.4 million for the years ended December 31, 2017 and 2016, respectively. There were no other changes in accumulated depreciation other than the foreign currency impact.
6. Accrued Expenses
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
Compensation expense | $ | 2,325 |
| | $ | 1,198 |
|
Professional fees | 1,322 |
| | 624 |
|
Research and development fees | 791 |
| | 1,284 |
|
Audit and tax fees | 424 |
| | 455 |
|
Other current liabilities | 1,308 |
| | 158 |
|
Total | $ | 6,170 |
| | $ | 3,719 |
|
7. Income Taxes
The Company reported a loss before income taxes consisting of the following (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
Domestic | $ | (13,840 | ) | | $ | (8,725 | ) |
Foreign | (2,704 | ) | | (13,913 | ) |
Loss before income taxes | $ | (16,544 | ) | | $ | (22,637 | ) |
The components of the provision (benefit) for income taxes are as follows (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
Current: | | | |
Federal | $ | — |
| | $ | — |
|
State | — |
| | — |
|
Foreign | 1,103 |
| | 162 |
|
Total current | 1,103 |
| | 162 |
|
Deferred: | | | |
Federal | — |
| | — |
|
State | — |
| | — |
|
Foreign | — |
| | — |
|
Total deferred | — |
| | — |
|
Provision (benefit) for income taxes | $ | 1,103 |
| | $ | 162 |
|
The reconciliation of the federal statutory rate to Pieris’ effective tax rate is as follows:
|
| | | | | |
| 2017 | | 2016 |
Federal income tax rate | 34.0 | % | | 34.0 | % |
Tax Reform - Change in enacted rate | (22.2 | ) | | |
Foreign rate differential | (1.2 | ) | | (2.9 | ) |
State tax, net of federal benefit | 2.4 |
| | 0.9 |
|
Permanent items | — |
| | (2.3 | ) |
Other | 1.8 |
| | (0.9 | ) |
Withholding tax | — |
| | (0.7 | ) |
Change in valuation allowance | (21.5 | ) | | (28.8 | ) |
Effective income tax rate | (6.7 | )% | | (0.7 | )% |
The components of deferred tax assets and liabilities related to net tax effects of temporary differences between the carrying amount of assets and liabilities for financial reporting purposes and the amounts used for income taxes purposes were as follows (in thousands):
|
| | | | | | | |
| Years Ended December 31, |
| 2017 | | 2016 |
Deferred tax assets: | | | |
Net operating loss carryforwards | $ | 22,170 |
| | $ | 18,498 |
|
Share based awards compensation | 1,280 |
| | 1,080 |
|
Accrued compensation/other | 296 |
|
| 239 |
|
Deferred Revenue | 1,504 |
| | — |
|
Total deferred tax assets | 25,250 |
| | 19,817 |
|
Less: valuation allowance: | (25,250 | ) | | (19,817 | ) |
Net deferred tax asset | $ | — |
| | $ | — |
|
The Company operates in multiple jurisdictions. Accordingly, the Company files United States federal and state income tax returns as well as returns in multiple foreign jurisdictions. In assessing the realizability of deferred tax assets, management considers whether it is more likely than not that some portion or all deferred tax assets will not be realized. The ultimate realization of deferred tax assets is dependent upon the generation of future taxable income during the periods in which those temporary differences become deductible. Management considers the scheduled reversal of deferred tax liabilities, projected future taxable income, and tax-planning strategies in making this assessment. Management believes it is more likely than not that the results of future operations will not generate sufficient taxable income in the U.S. or in its foreign jurisdictions to realize the full benefits of its deferred tax assets. As of December 31, 2017, we continue to maintain a full valuation allowance against all net deferred tax assets.
The increase in the valuation allowance of deferred tax assets of $5.5 million was primarily influenced by the operating losses generated in current tax year, and the impact of deferred revenue.
As of December 31, 2017, the Company had net operating loss carryforwards for United States federal income tax purposes of $24.5 million and net operating loss carryforwards for state income tax purposes of $21.3 million. These tax loss carryforwards expire through 2037. In the United States, utilization of the NOL carryforwards may be subject to a substantial annual limitation under Section 382 of the Internal Revenue Code of 1986 and similar state provisions due to ownership change limitations that have occurred previously or that could occur in the future. These ownership changes may limit the amount of NOL carryforwards that can be utilized annually to offset future taxable income and tax, respectively. The Company has not currently completed a study to assess whether an ownership change has occurred, or whether there have been multiple ownership changes since the Acquisition.
As of December 31, 2017, the Company had German corporate income tax and trade tax net operating loss carryforwards of approximately $46.1 million and $45.6 million respectively. Under current German laws, tax loss carryforwards may only be used to offset any relevant later assessment period (calendar year) $1.2 million plus 60% of the exceeding taxable income and trade profit of such period. In addition, certain transactions, including transfers of shares or interest in the loss holding entity, may result in the partial or total forfeiture of tax losses existing at that date. Partial or total forfeiture of tax losses may further occur in corporate reorganizations of the loss holding entity. Tax years ended December 31, 2013 or later remain subject to examination by the German tax authorities.
The Company accounts for uncertain tax positions pursuant to ASC 740 which prescribes a recognition threshold and measurement process for financial statement recognition of uncertain tax positions taken or expected to be taken in a tax return. If the tax position meets this threshold, the benefit to be recognized is measured at the largest amount of benefit that is more likely than not (determined by cumulative probability) of being realized upon ultimate settlement with the taxing authority. The Company recorded an uncertain tax position related to a prior year position, that if successfully challenged by tax authorities could result in the loss of certain tax attributes. The balance of uncertain tax positions will remain until such time that settlement is reached with the relevant tax authorities or should the statute of limitations expire. The Company recognizes interest and penalties, if any, related to uncertain tax positions in income tax expense. No interest and penalties related to uncertain tax positions were accrued at December 31, 2017 and December 31, 2016.
The following table sets forth a reconciliation of the beginning and ending amounts of unrecognized tax benefits, excluding the impact of interest and penalties, for the years ended December 31, 2017 and 2016 (in thousands):
|
| | | |
Unrecognized tax benefits at December 31, 2016 | $ | 5,655 |
|
Currency translation adjustment | 796 |
|
Unrecognized tax benefits at December 31, 2017 | $ | 6,451 |
|
The Company does not expect unrecognized tax benefits to change significantly over the next twelve months. The full amount of unrecognized tax benefits would impact the effective rate, subject to valuation allowance considerations, if recognized.
Enacted Tax Legislation
The Company does not expect unrecognized tax benefits to change significantly over the next twelve months. The full amount of unrecognized tax benefits would impact the effective rate, subject to valuation allowance considerations, if recognized. On December 22, 2017, the Tax Cuts and Jobs Act of 2017 (the “Act”) was signed into law making significant changes to the Internal Revenue Code. Changes include, but are not limited to, a corporate tax rate decrease from 35% to 21% effective for tax years beginning after December 31, 2017, the transition of U.S international taxation from a worldwide tax system to a territorial system, and a one-time transition tax on the mandatory deemed repatriation of cumulative foreign earnings as of December 31, 2017. ASC 740 requires the tax effects of changes in tax laws must be recognized in the period in which the law is enacted, or December 22, 2017 for the Act. ASC 740 also requires deferred tax assets and liabilities to be measured at the enacted tax rate expected to apply when temporary differences are to be realized or settled. Thus, at the date of enactment, the Company’s deferred taxes were re-measured utilizing the new federal income tax rate of 21%. On December 22, 2017, the SEC staff issued Staff Accounting Bulletin No. 118, Income Tax Accounting Implications of the Tax Cuts and Jobs Act, or SAB 118, which allows the recording of provisional amounts during a measurement period not to extend beyond one year of the enactment date. In accordance with SAB 118, we have determined that our deferred tax asset value and associated valuation allowance reduction of $3.7 million is a provisional amount and a reasonable estimate at December 31, 2017. The final impact may differ from this provisional amount due to, among other things, changes in interpretations and assumptions we have made thus far and the issuance of additional regulatory or other guidance. We expect to complete the final impact within the measurement period. The Company has quantified the impact of the rate reduction from 34% to 21% in its balance sheet. No other aspect of the Act had a material effect on the Company’s consolidated financial statements or related footnote disclosure.
Australian Tax Credits
From time to time, the Company's Australian subsidiary has applied for and received cash tax credit, irrespective of a tax liability, based on certain qualifying activities. The Australian research and development Tax Incentive credit is a self-assessed, entitlement program that provides a credit for eligible research and development entities engaging in qualifying research and development activities. The level of credit is based upon a percentage of the research and development entity’s aggregated turnover for the income tax year, subject to income limitations and provided that the research and development entity is not controlled by an exempt entity or combination of exempt entities. Pieris records the Australian research and development tax credit as an offset to research and development expenses in the consolidated statements of operations, which follows the income statement classification of the original expenses. For the year ended December 31, 2016, the Company recorded $1.5 million as an expense offset, along with a receivable of $1.5 million at December 31, 2016. The Company did not apply for this tax incentive credit in 2017.
8. Stockholders’ equity
Common Stock
During the year ended December 31, 2017, the Company issued 453,209 net shares of common stock upon exercise of stock options. Stock options to purchase 182,471 shares of common stock were net exercised resulting in the net issuance of 83,491 shares of common stock and stock options to purchase 369,718 shares of common stock were exercised for cash, providing cash proceeds of $0.8 million. No stock options were exercised during the 2016 period.
During the year ended December 31, 2017, the Company issued 1,505,026 shares of common stock due to warrant exercises. Net exercise of 89,330 shares of common stock underlying the warrants resulted in the issuance of 49,127 shares of common stock. Additionally, 1,455,899 were exercised resulting in cash proceeds of $3.3 million. No warrants were exercised during the 2016 period.
Each share of the Company’s common stock is entitled to one vote and all shares rank equally as to voting and other matters.
Dividends may be declared and paid on the common stock from funds legally available therefor, if, as and when determined by the Board of Directors.
Private Placement and Preferred Stock
In June 2016, the Company entered into a securities purchase agreement (the “Securities Purchase Agreement”) for a private placement of the Company’s securities with a select group of institutional investors (the “2016 PIPE”). The 2016 PIPE sale transaction, by the Company, consisted of 8,188,804 units at a price of $2.015 per unit for gross proceeds, to the Company, of approximately $16.5 million. After deducting for placement agent fees and offering expenses, the aggregate net proceeds from the private placement was approximately $15.3 million.
Each unit consisted of (i) one share of the Company’s Common Stock or non-voting series A convertible preferred stock (the “Series A Convertible Preferred Stock”) which are convertible into one-thousand shares of common stock, (ii) one warrant to purchase 0.4 shares of Common Stock at an exercise price of $2.00 per share and (iii) one warrant to purchase 0.2 shares of Common Stock at an exercise price of $3.00 per share. The warrants will be exercisable for a period of five years from the date of issuance. Each share of Series A Convertible Preferred Stock was issued at a price of $2.015 per share, and is convertible into 1,000 shares of common stock, provided the holder and/or its affiliates do not own greater than 9.99% of the total number of Pieris common stock then outstanding. The Series A Convertible Preferred Stock has a par value of $0.001 per share, has no registration or voting rights, and holders are entitled to receive dividends on a pari passu basis with the Company´s common stock, when, and if declared. In event of a true liquidation or winding down of the business, holders of Series A Convertible Preferred Stock will be paid prior to the holders of Common Stock. In connection with the 2016 PIPE, the Company issued 3,225,804 shares of Common Stock and 4,963 shares of Series A Convertible Preferred Stock to the 2016 PIPE investors.
The Company has 4,963 shares of preferred stock both issued and outstanding during the years ended December 31, 2017 and 2016, respectively.
9. Net Loss per Share
Basic net loss per share is calculated by dividing net income (loss) by the weighted average shares outstanding during the period, without consideration for common stock equivalents. Diluted net loss per share is calculated by adjusting weighted average shares outstanding for the dilutive effect of common stock equivalents outstanding for the period, determined using the treasury-stock and if-converted methods. For purposes of the diluted net loss per share calculation, preferred stock, stock
options, and warrants are considered to be common stock equivalents but have been excluded from the calculation of diluted net loss per share, as their effect would be anti-dilutive for all periods presented. Therefore, basic and diluted net loss per share were the same for all periods presented.
For the years ended December 31, 2017 and 2016, and as calculated using the treasury stock method, approximately 14.4 million and 8.2 million of weighted average shares, respectively, were excluded from the calculation of diluted weighted average shares outstanding as their effect was antidilutive.
10. Stock and employee benefit plans
In December 2014, the Company adopted the 2014 Employee, Director and Consultant Equity Incentive Plan, (the “2014 Plan”) which provides for the grant of stock options to certain designated employees of the Company, non-employee directors of the Company and certain other persons performing significant services for the Company as designated by the Compensation Committee of the Board of Directors. The 2014 Plan was terminated on June 28, 2016 when the Company adopted the 2016 Employee, Director and Consultant Equity Incentive Plan, (the “2016 Plan”). Therefore, no options were granted under the 2014 Plan and no options are available for future grant after this date.
The 2016 Plan provides for the grant of stock options, restricted and unrestricted stock awards, and other stock-based awards to employees of the Company, non-employee directors of the Company, and certain other consultants performing services for the Company as designated by either the Board of Directors or the Compensation Committee of the Board of Directors. The 2016 Plan authorizes the issuance of up to 3,750,000 shares of common stock plus awards canceled or expired under the 2014 Plan, if any. Shares which are vested or continue to vest can still be exercised under the 2014 Plan. The 2016 Plan does not provide for an “evergreen” provision. The vesting periods of equity incentives issued under the 2016 Plan are determined by the Compensation Committee of the Company´s Board of Directors, with stock options generally vesting over a four-year period.
The Company’s stock options have a maximum term of 10 years from the date of grant. Stock options granted under the 2014 Plan or 2016 Plan may be either incentive stock options or nonqualified stock options. The exercise price of stock options granted under the 2014 Plan and 2016 Plan must be at least equal to the fair market value of the common stock on the date of grant. The Company’s general policy is to issue common shares upon the exercise of stock options.
The Company estimates the fair value of each employee stock award on the grant date using the Black-Scholes option-pricing model based on the following assumptions:
|
| | | | |
| | Years Ended December 31, |
| | 2017 | | 2016 |
Risk free interest rate | | 1.77% - 2.22% | | 1.13% - 2.08% |
Expected term | | 5.00 - 5.73 years | | 5.00 - 5.73 years |
Dividend yield | | — | | — |
Expected volatility | | 75.1% - 78.9% | | 74.9% - 76.0% |
The weighted-average fair value of the 1,852,682 and 1,873,047 options granted during the years ended December 31, 2017 and 2016 was $2.47 and $1.00, respectively.
Total stock-based compensation expense is recorded in operating expenses based upon the functional responsibilities of the individuals holding the respective options as follows (in thousands):
|
| | | | | | | | |
| | Years Ended December 31, |
| | 2017 | | 2016 |
Research and development | | $ | 844 |
| | $ | 599 |
|
General and administrative | | 2,202 |
| | 1,306 |
|
Total stock-based compensation | | $ | 3,046 |
| | $ | 1,905 |
|
As of December 31, 2017, there were 1,612,514 shares available for future grant under the 2016 Plan, including 194,542 shares which were forfeited under the 2014 Plan.
The following table summarizes stock option activity for employees and nonemployees (shares in thousands):
|
| | | | | | | | | | | | |
| Number of Options | | Weighted- Average Exercise Price | | Weighted- Average Remaining Contractual Life | | Aggregate Intrinsic Value (in thousands) |
Outstanding, December 31, 2016 | 4,423 |
| | $ | 1.99 |
| | | | $ | — |
|
Granted | 1,853 |
| | 3.23 |
| | | | 4 |
|
Exercised | (552 | ) | | 2.34 |
| | | | 886 |
|
Canceled | (280 | ) | | 2.78 |
| | | | 277 |
|
Outstanding, December 31, 2017 | 5,444 |
| | $ | 2.33 |
| | 8.07 years | | $ | 28,395 |
|
Vested or expected to vest, December 31, 2017 | 5,444 |
| | $ | 2.33 |
| | 8.07 years | | $ | 28,395 |
|
Exercisable, December 31, 2017 | 3,067 |
| | $ | 1.96 |
| | 7.32 years | | $ | 17,141 |
|
Periodically, the Company grants options outside of the 2014 and 2016 Plans as inducement options, which are material awards to the executive officers or other personnel entering senior leadership roles with the Company. These awards are excluded from the table above. The following table summarizes stock option activity for these inducement options (in thousands):
|
| | | | | | | | | | | | |
| Number of Options | | Weighted- Average Exercise Price | | Weighted- Average Remaining Contractual Life | | Aggregate Intrinsic Value (in thousands) |
Outstanding, December 31, 2016 | 1,000 |
| | $ | 2.41 |
| | | | $ | — |
|
Granted | 625 |
| | $ | 5.24 |
| | | | $ | — |
|
Canceled | (94 | ) | | $ | 1.45 |
| | | | $ | 572 |
|
Outstanding, December 31, 2017 | 1,531 |
| | $ | 3.62 |
| | 8.79 years | | $ | 6,015 |
|
Vested or expected to vest | 1,531 |
| | $ | 3.62 |
| | 8.79 years | | $ | 6,015 |
|
Exercisable, December 31, 2017 | 688 |
| | $ | 2.23 |
| | 8.39 years | | $ | 3,657 |
|
The compensation expense with all inducement options was $0.8 million of which $0.3 million is included in research and development expense and $0.5 million is included in general and administration expense for the year ended December 31, 2017. The compensation expense for inducement options for the year ended December 31, 2016 was $0.3 million and is included in research and development expense.
As of December 31, 2017, the total unrecognized compensation cost related to non-vested awards was $6.4 million of which $2.4 million are for inducement options. The Company expects to recognize the compensation cost over a remaining weighted-average period of 3.28 years.
401(k) Savings plan
In 2015, the Company established a defined-contribution savings plan under Section 401(k) of the Internal Revenue Code (the “401(k) Plan”). The 401(k) Plan covers all U.S. based employees who meet defined minimum age and service requirements, and allows participants to defer a portion of their annual compensation on a pretax basis. The Company made matching contributions to participants in this plan which totaled $45 thousand and $32 thousand for the years ended December 31, 2017 and 2016, respectively.
11. License and Transfer Agreement
In April 2016, the Company entered into a license and transfer agreement (the “Original Agreement”) with Enumeral Biomedical Holdings, Inc. (“Enumeral”), pursuant to which the Company acquired a non-exclusive worldwide license to use specified patent rights and know-how owned by Enumeral to research, develop and market fusion proteins. As contemplated by the terms of the Original Agreement, the Company entered into a definitive license and transfer agreement (the “Definitive Agreement”) with Enumeral on June 6, 2016, to expand the scope of the Company’s option to license additional antibodies from Enumeral. Under the Definitive Agreement, Enumeral granted Pieris options to license two additional undisclosed Enumeral antibodies (each, a “Subsequent Option”). The Subsequent Options expired unexercised on May 31, 2017.
Under the terms of the Original Agreement, the Company agreed to pay Enumeral an upfront license fee of $250 thousand upon signing in April 2016 and subsequently elected to pay a $750 thousand maintenance fee in May 2016. All amounts paid pursuant to the Original Agreement were recorded as research and development expense in the period they were incurred. No license or maintenance fees were incurred for the year ended December 31, 2017.
On January 29, 2018, Enumeral filed a voluntary petition for relief under Chapter 11 of the United States Bankruptcy Code in the Bankruptcy Court for the District of Massachusetts (the “Bankruptcy Court”). Enumeral continues to operate its business as a debtor in possession under the jurisdiction of the Bankruptcy Court.
Prior to and in contemplation of its bankruptcy filing, on January 26, 2018, Enumeral entered into an Asset Purchase Agreement (the “Purchase Agreement”) with XOMA US LLC. The Purchase Agreement contemplates that Enumeral will sell specified assets of its PD-1 antibody program, including without limitation, all of its rights under the Definitive License Agreement (collectively, the “PD-1 Assets”). The Bankruptcy Court approved a process pursuant to which Enumeral will solicit higher or better offers for the PD-1 Assets and conduct an auction process to determine the ultimate purchaser. The process contemplates that the winning bidder will purchase the PD-I Assets, including the Definitive License Agreement, with the sale contemplated to close in early April 2018. We will have an opportunity to vet the proposed purchaser to ensure that they (i) can continue to perform Enumeral’s obligations under the Definitive License Agreement, and (ii) will not interfere with our use of the rights we currently enjoy under the Definitive License Agreement.
12. Commitments and Contingencies
Licensing Commitments
The Company has license agreements with two parties under which the Company is obliged to pay annual license fees. One agreement requires annual license payments for Strep-tag technology that represents tool technologies used for research purposes only. This agreement expires in 2024. Another license agreement also requires a minimum annual license fee for an exclusive license to certain patents related to the Company's core Anticalin technology platform. This agreement expires upon expiration of the last patent subject to that agreement. The minimum annual license fee commitments under the two agreements as of December 31, 2017 (in thousands):
|
| | | |
| License payments |
2018 | $ | 96 |
|
2019 | 96 |
|
2020 | 96 |
|
2021 | 96 |
|
2022 | 96 |
|
Thereafter | 372 |
|
Total minimum license payments | $ | 852 |
|
The Company is also obliged to pay low single digit royalties, including annual minimum royalties, on sales of products that utilize its core Anticalin technology platform. Should the Company grant licenses or sublicenses to those patents to third parties, it will be obliged to share a percentage of resulting revenue with the licensee, which percentage of resulting revenue is creditable against annual license payments. In addition, payment obligations are reduced by the Company's proportionate contribution to a joint invention. Payment obligations terminate on expiration or annulment of the last patent covered by the agreement.
Leases
The Company leases office and laboratory space in Freising, Germany as well as office space in Boston, Massachusetts. The German lease agreement has a defined termination date, which is eight months after the end of each quarter, with unlimited renewal periods at the Company's discretion. In June 2016, the Company entered into a second lease agreement for additional office space in the same building, which has a fixed term of one year along with unlimited renewal periods. As Pieris did not cancel this lease agreement by June 2017, the term of the lease extended by one year until June 2019. In August 2015, the Company entered into an Agreement of Sublease to lease approximately 3,950 square feet in Boston, MA. The term of the lease expires in February 2022.
The Company records rent expense on a straight-line basis over the lease term period. For the years ended December 31, 2017 and 2016 respectively, the Company recognized rent expense under its existing lease agreements in an amount of $0.5 million and $0.5 million for the years ended December 31, 2017 and 2016, respectively.
The Company’s contractual commitments of the non-cancellable portion under these operating leases as of December 31, 2017 are as follows (in thousands):
|
| | | |
| Total |
2018 | $ | 456 |
|
2019 | 218 |
|
2020 | 200 |
|
2021 | 204 |
|
2022 | 34 |
|
Total minimum lease payments | $ | 1,112 |
|
13. Subsequent Events
License and Collaboration Agreement and Non-Exclusive Anticalin Platform Technology License Agreement with Seattle Genetics
On February 8, 2018, the Company, together with Pieris GmbH, entered into a License and Collaboration Agreement and a Non-Exclusive Anticalin Platform Technology License Agreement (collectively the “Seattle Genetics Agreements”) with Seattle Genetics, pursuant to which the parties will develop multiple targeted bispecific immuno-oncology treatments for solid tumors and blood cancers.
Under the terms of the Seattle Genetics Agreements, Seattle Genetics paid Pieris a $30 million upfront fee, tiered royalties on net sales up to the low double-digits, and will pay up to $1.2 billion in total success-based payments across three product candidates. The companies will pursue multiple antibody-Anticalin fusion proteins during a research phase, and Seattle Genetics has the option to select up to three therapeutic programs for further development. Prior to the initiation of a pivotal trial, Pieris may opt into global co-development and U.S. commercialization of the second program and share in global costs and profits on a 50/50 basis. Seattle Genetics will solely develop, fund and commercialize the other two programs. Seattle Genetics may also decide to select additional candidates from the initial research phase for further development in return for the payment to Pieris of additional fees, milestone payments, and royalties.
The term of each of the Seattle Genetics Agreements ends upon the expiration of all of Seattle Genetics’ payment obligations under such Agreement. The License and Collaboration Agreement may be terminated by Seattle Genetics in its entirety for convenience beginning 12 months after its effective date upon 90 days notice or, for any program where a pivotal study has been initiated, upon 180 days notice. Any program may be terminated at Seattle Genetics’ option. If any program is terminated by Seattle Genetics after a pre-defined pre-clinical stage, Pieris will have full rights to continue such program. If any program is terminated by Seattle Genetics prior to such pre-defined pre-clinical stage, Pieris will have the right to continue to develop such program, but will be obligated to offer a co-development option to Seattle Genetics for such program. The License and Collaboration Agreement may also be terminated by Seattle Genetics or Pieris for an uncured material breach by the other party upon 90 days notice, subject to extension for an additional 90 days if the material breach relates to diligence obligations and subject, in all cases, to dispute resolution procedures. The License and Collaboration Agreement may also be terminated due to the other party’s insolvency and may in certain instances, including for reasons of safety, be terminated on a product-by-product basis. Each party may also terminate the Seattle Genetics Agreements if the other party challenges the validity of any patents licensed under the Seattle Genetics Agreements, subject to certain exceptions. The Non-Exclusive Anticalin Platform Technology License Agreement will terminate upon termination of the License and Collaboration Agreement, whether in its entirety or on a product-by-product basis.
Other License Agreement Updates
Our collaboration with Roche
On February 26, 2018, pursuant to the Roche Agreement, as amended, Roche provided notice to Pieris of its extension of the research term of the Roche Agreement to continue until August 31, 2018.
Our collaboration with Daiichi Sankyo
Daiichi Sankyo Company Limited (“Daiichi Sankyo” or "Daiichi") partnered with Pieris to research, develop and commercialize two Anticalin therapeutics pursuant to a May 31, 2011 Collaboration Research and Technology Licensing Agreement (the “DS Collaboration Agreement”).
On May 8, 2017, Daiichi Sankyo discontinued the development of one of those two therapeutics, a PSCK9 program (DS-9001), after having advanced this program to first-in-human studies for strategic reasons. Consequentially, this Anticalin program, including respective intellectual property rights, were transferred back to Pieris.
The second of these two therapeutic programs pursued under the DS Collaboration Agreement is an Anticalin protein directed to calcitonin gene-related peptide (“CGRP”) for therapeutic use in the treatment of migraine headaches. The CGRP program is in the IND-enabling stage and Pieris previously reported Daiichi Sankyo’s decision to initiate a GLP toxicity study in connection with this program.
Due to Daiichi Sankyo’s strategic prioritization and commercial reasons related to competing CGRP inhibitors in advanced stages of development, Daiichi Sankyo provided notice to Pieris on March 1, 2018 of its termination of the CGRP program. Pre-clinical data regarding this CGRP antagonizing Anticalin protein indicate that it has good drug-like properties, including strong target affinity, the ability to neutralize the biological activity of CGRP in vitro, and the ability to inhibit vasodilation, a cause of migraine pain, in rat skin following subcutaneous administration of the antagonist. In connection with the termination of the development of the CGRP program, and under the terms of the DS Collaboration Agreement, Daiichi Sankyo is obligated to carry out certain activities to facilitate transfer of activities, regulatory filings, materials, data, agreements and other matters to Pieris in connection with the return to Pieris of the development program related to the CGRP program. Pieris intends to diligently review the data associated with the program and consider its strategic options thereafter.
The foregoing summary of the terms relating to the DS Collaboration Agreement, including with respect to Daiichi Sankyo’s termination of such DS Collaboration Agreement with respect to the CGRP program, is qualified in its entirety by the terms of the DS Collaboration Agreement, which were filed with the SEC as exhibit 10.7 to the Form 10-K filed on March 30, 2017. The contents of such exhibit are hereby incorporated by reference.
As a result of the termination of the DS Collaboration Agreement, the Company is no longer eligible for future research, development and regulatory milestones.
Underwritten Public Offering
In February 2018, the Company completed an underwritten public offering of its common stock in which it sold 6,325,000 shares of Common Stock, including the exercise in full by the underwriters of their option to purchase an additional 825,000 shares of Common Stock, to the public at a price of $8.00 per share. The offering was completed under the shelf registration statement that was filed on Form S-3 and declared effective by the SEC on August 3, 2016. Net proceeds of the underwritten public offering, after deducting the underwriting discounts and commissions, were $47.6 million, excluding offering expenses of approximately $0.3 million incurred by the Company.